![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
15 Cards in this Set
- Front
- Back
|
How do we name alcohols |
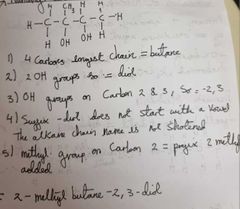
Identify the longest carbon chain Identify how many OH groups there are Identify which carbon the OH group(s) are on If the suffix starts with a vowel the alkane chain name is shortened Identify where the other groups are if there are any |
|
|
What are primary alcohols |
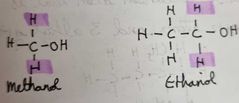
When the OH groups is attached toba carbon atom that is attached to 2 hydrogen atoms and 1 alkyl group |
|
|
What are secondary Alcohols |
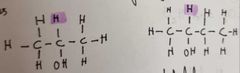
OH group attached to a carbon atom is attached to 1 hydrogen atom and 2 alkyl group |
|
|
What are the physical properties of Alcohols |
Alcohols are less volative, have high MP and greater water solubility than the alkanes with the same number of carbon atoms |
|
|
Why are Alcohols less volatile than alkanes with the same number of C |
Has weak London forces but there will also be much stronger Hydrogen bonds between the polar OH groups Requires greater energy to break than the alkane with the same number of carbons |
|
|
Why are alcohols soluble in water |
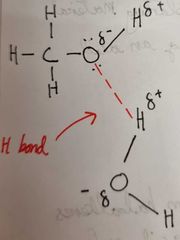
Compounds that can form hydrogen bonds with water are far more water soluble than a compound that can't
Alcohols are completely soluble as hydrogen bonds form between the polar OH group of the Alcohol and water |
|
|
What are tertiary alcohols |
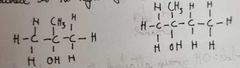
OH group is attached to a carbon atom that I'd attached to no hydrogen atoms and 3 alkyl groups |
|
|
What is produced in the combustion of alcohols |
CO2 and H2O Exothermic reaction Longer the carbon chain the greater energy released |
|
|
What happens in the dehydration of Alcohols |
Water molecule is removed from the starting material Heated under reflux in the presence of an acid catalyst Alkene produced |
|
|
What happens in the substitution reactions of alcohols |
Reacted with hydrogen halides to form haloalkenes Heated under reflux with sulfuric acid and a sodium halide, the hydrogen bromide is formed |
|
|
What happens in the oxidation of alcohols |
Primary and secondary alcohols can be oxidised by an oxidising agent Oxidising mixture = solution of potassium dichromate, acidification with H2SO4 |
|
|
What happens when we oxidise primary alcohols |
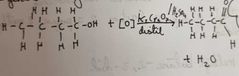
Gentle heating and distil Produces Aldehyde |
|
|
What happens when we further oxidise a primary alcohol |
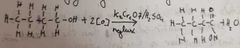
Heated strongly + reflux Produces a carboxylic acid |
|
|
What happens when we oxidise a secondary alcohol |
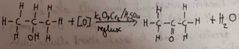
Oxidised to ketones
Heated under reflux Dichromate ions turn colour from orange to green |
|
|
What happens when we oxidise tertiary alcohols |
They don't undergo oxidation reactions Acidification dichromate remains orange when added |

