![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
40 Cards in this Set
- Front
- Back
|
Infra-red radiation is part of what spectrum? |
The electro-magnetic spectrum |
|
|
Name other names used for infra-red radiation |
Heat radiation or Thermal radiation |
|
|
How does infra-red radiation heat up objects? |
It makes the molecules move faster |
|
|
Can humans see infra-red radiation? |
No, digital cameras can though. |
|
|
All objects can ....................... or ...................... radiation. |
emit and absorb |
|
|
What can pick up radiation emitted by hot objects? |
Infra red cameras |
|
|
The ............. the object, the more radiation it emits. |
Hotter |
|
|
what type of camera can show hot spots? |
Heat sensitive camera |
|
|
Which colours absorb thermal radiation the best? |
Dark, especially black colours |
|
|
Which colours reflect thermal radiation the best? |
White and silver |
|
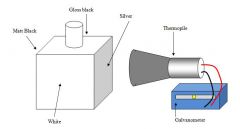
What is this experiment called? |
Leslie's Cube |
|
|
Explain the steps involved in the Leslie's cube experiment |
Hot water is poured into the cube and heat passes from the sides by radiationwhich is picked up by a detector called athermopile.
The thermopile is connected to a galvanometer, a very sensitive ammeter which can detect tiny currents.
The more infra red that is given out, the further the galvanometer moves across. |
|
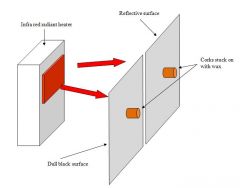
Explain which cork will slip first |
The cork on the dull black surface will slip down first.
This is because the infra-red is absorbed by the dull black surface.
The other cork will not move, because the infra-red is reflected back. |
|
|
What are the two factors that can change the amount of thermal radiation an object emits and absorbs? |
Shape and colour |
|
|
Which absorbs more radiation Dull black surfaces or white shiny surfaces? |
Dull black surfaces |
|
|
White surfaces ............. radiation. |
reflect |
|
|
Houses in hot countries are often pained white for what reason? |
To reflect the thermal radiation from the sun, this helps keep the house cool |
|
|
A fell-runner suffering from hypothermia (has got too cold) is wrapped up in a space blanket which is made of what? Why? |
highly reflective foil, to reflect all their heat back onto them |
|
|
Thermal radiation is an important factor in what environmental change? |
global warming |
|
|
What is Kinetic theory about? |
How molecules move about and change state from solid to liquid to gas and back. |
|
|
On planets that are close to their stars, what state is water in |
a gas, as it has evaporated because it is so hot |
|
|
what is this state of matter? How can you tell? How does it change state from this? |
A solid, it has a long range order. The molecules cannot move about.
However the bonds in a molecule can vibrate. They vibrate more if the temperature is higher. If the molecules vibrate enough, the bonds between molecules break, and the long-range order breaks down. We say that the substance is melting. |
|
|
what is this state of matter? How can you tell? How does it change state from this? |
Liquid, small groups of molecules can move past each other. A liquid adopts the shape of its container. If there is no container, it will spread out into a thin layer.
The liquid evaporates quickly. If you spill some ethanol on the bench, it disappears quickly. If we continue to put energy into the liquid, the molecules move about faster.
Also intermolecular bonds break more, and single molecules are given off as a gas. All liquids have a boiling point at which the liquid becomes a gas. |
|
|
what is this state of matter? How can you tell? How does it change state from this? |
Gas, the molecules are now on their own. The space between molecules is large, and the molecules are free to move about.
They collide against the walls of the container. The force from these collisions is called the pressure. Since there is a lot of space between molecules, the gas can be squashed together, or compressed. If we compress the gas enough, we can actually make it liquid again.
|
|
|
What is sublimation? |
Some materials, for example carbon dioxide, can change state from solid to gas without passing a liquid phase. |
|
|
explain the changes of state graph... |
The flat parts on the graph are made when the temperature stays the same. The temperature stays the same because all the kinetic energy is being used to brake the bonds instead of to raise the temperature. while the particles are being separated it doesn't have enough heat energy to warm raise the temperature. The same applies then the bonds are being re-made because it also takes energy to join the back together, condensing and freezing |
|
|
What temperature does water freeze at? |
0 degrees celsius |
|
|
What does the boiling point of water depend on? |
The temperature
The amount of pressure |
|
|
When water is at 0oc what is it often referred to as? Why? |
The triple point because at that temperature, it can exist in all three states, solid, liquid, or gas. |
|
|
Temperature |
-the measure of how hot something is, measured in degrees Celsius |
|
|
Heat |
a flow of energy from a hot object to a cold object. Heat is measured in joules (J). |
|
|
Heat always flows from what hot to cold, cold to hot or either? |
Hot to cold |
|
|
The greater the temperature difference between the hot object and the cold object... |
...the greater is the flow of heat. |
|
|
Temperature represents what? |
internal energy, the energy that is the result of molecules vibrating. |
|
|
what are the three methods for heat to flow |
Conduction Convection Radiation |
|
|
Metals are___________conductors of heat |
good |
|
|
Non-metals are generally_______ conductors of heat |
bad |
|
|
Liquids and gases are_______conductors of heat |
Bad |
|
|
A bad conductor of heat is called what? |
an insulator |
|
|
Why does it keep you warm? |
The fibres criss-cross to make small pockets of air.
Air is a very poor conductor of heat. |

