![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
27 Cards in this Set
- Front
- Back
|
Define equilibrium |
In a closed system The forward and backward reactions Occur at the same rate |
|
|
Functional group: alcohol |
-OH |
|
|
Functional group: carboxylic acid |
-COOH |
|
|
Functional group: esther |
-COO- |
|
|
Number of moles = |
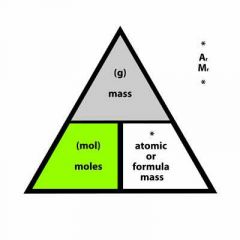
|
|
|
Concentration = |
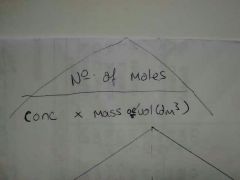
|
|
|
Define electrolyte |
The substance broken down in electrolysis A liquid containing free moving ions, that is broken down by electricity in the process of electrolysis |
|
|
In which form is the electrolyte? |
Molten or in solution, so the ions are free to move |
|
|
When does electrolysis take place? |
When a current is passed through the: Molten ionic compound OR The solution containing ions |
|
|
What are the electrodes made from? |
An inert substance |
|
|
Why are the electrodes made from the type of material that they are made from? |
So they do not react with the products |
|
|
What is produced at the anode (+ve) when an aqueous solution is electrolysed? |
Oxygen gas UNLESS halides are present |
|
|
What is produced at the cathode (-ve) when an aqueous solution is electrolysed? |
Hydrogen gas UNLESS the solution contains metal ions that are LESS reactive that hydrogen |
|
|
What is the ore of aluminium? |
Bauxite (aluminium oxide) |
|
|
What is added to aluminium to make it melt at a lower temperature? |
It is mixed with molten cryolite |
|
|
Why do carbon anodes need to be replaced regularly in the electrolysis of aluminium? |
At high temperatures, the oxygen from the bauxite reacts with the carbon anodes to form carbon dioxide, so they gradually burn away |
|
|
What are the products of the electrolysis of brine? |
Hydrogen gas Chlorine gas Sodium hydroxide solution |
|
|
What are the uses of the products of the electrolysis of brine? |
Hydrogen - margarine, HCl Sodium hydroxide - soap, bleach, paper, control pH (strong alkali) Chlorine - bleach, kill bacteria in water |
|
|
What is electroplating? |
Using electrolysis to place a thin layer of metal onto an object |
|
|
Benefits of electroplating |
Aesthetics Protects a metal from corroding Increases the hardness of a surface Reduce costs by using a thin layer of an expensive metal rather than making the whole object from the pure metal |
|
|
Which electrode is the object being electroplated placed as? |
Cathode (-ve) |
|
|
Describe phytomining |
Plants are grown in soil which contains copper
As the plant takes up nutrients from the soil, copper also builds up in the plant's tissue
The plant is harvested, dried, and burnt in a furnace
Copper is collected from the ash of the plant using scrap iron (displacement) and electrolysis |
|
|
Benefits of phytomining and bioleaching |
Copper can be obtained from low grade ores. Copper-rich ores are less available |
|
|
Disadvantages of phytomining and bioleaching |
They are slow processes |
|
|
Describe bioleaching |
Bacteria feed on low grade copper ores
Through both biological and chemical processes, a solution of copper ions (leachate) is obtained
Scrap iron (displacement) and electrolysis is used to extract the copper from solution |
|
|
What is the catalyst for the haber process? |
Iron |
|
|
What is the catalyst for cracking? |
Zeolite or porcelain |

