![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
43 Cards in this Set
- Front
- Back
|
What are Photoautotrophs and Chemoheterotrophs ? |
Photoautotrophs: use sunlight to reduce CO2 (trees/plants) into sugars Chemoheterotrophs: oxidize sugars to make ATP (animals) to run other reactions |
|
|
What is cellular respiration ? |
Cellular respiration produces ATP frommolecules with high potential energy. The energy released from sugar processing isused to transfer a phosphate onto ADP to make ATP. |
|
|
What is the potential energy and chemical energy? |
Potential energy is stored energy. Potential energy is stored energy or energy atrest (ie. spring loaded mouse trap). Chemical energy is storedin the position of electrons. |
|
|
Recall: Carbohydrates and Energy Storage |
* Carbohydrates store and provide chemical energyin cells in the form of C–H bonds. * Carbohydrates - more free energy thanCO2; their electrons in C–H bonds andC–C bonds are shared more equally and held less tightly than the electrons in C–O bonds * Fatty acids: have even more C–H bonds = more free energy |
|
|
Recall: Exchange of potential & kinetic energy |
Energy is constantly being exchanged between these twoforms. Kinetic energy is energy of motion or energy inuse (rolling car, bowling ball, heat, light) Potential energy is stored energy. i.e. when a rock tumbles down a waterfall, its potential energy is converted intokinetic energy. |
|
|
Law of conservation of energy |
The law of conservation of energy says that energy cannot be created or destroyed, but canchange from one form to another,i.e. a water molecule is sitting at the top of a waterfall has a defined amount of potential energy. As it falls some of this stored energy is converted to kinetic energy (energy of motion). |
|
|
The 2nd Law of energy |
1. energy cannot be changed fromone form to another without a loss of usable energy (most of the energy lost is heat). 2. Another way to state the second energy law is that every energy transformation leads to more disorder or randomness (the degree of disorder or disorganization is called "entropy"). |
|
|
What Makes a Chemical Reaction Spontaneous? |
* Chemical reactions are spontaneous, if they proceed ontheir own, without any continuous external influence * spontaneity of a reaction is determined by two factors: 1. The amount of potential energy: the products of the spontaneous reaction have less potential energy than the reactants 2. The degree of order: The products are less ordered than the reactants |
|
|
Exergonic and endergonic reactions |
Exergonic reaction:energy released (ex=out) Endergonic reaction:energy required (endo=in) |
|
|
The second law of thermodynamics |
The second law of thermodynamics states that entropy (the amount of disorder in a group ofmolecules) always increases. In other words, chemical reactions result in products with less ordered (usable)energy. In general: Physical + chemical processes result in lower energy + more disorder |
|
|
What is "Gibbs Free-Energy Change" ? |
(ΔG) determines whether areaction is spontaneous or requires energy – represented by the letter "G" - defined as the energy of a system that is free to dowork - ΔG < 0 is an exergonic spontaneous reaction - ΔG > 0 is an endergonic reaction that requires energyinput - ΔG = 0 is a reaction that is at equilibrium |
|
|
What are exergonic and endergonic reactions in metabolism? |
EXERGONIC reaction (downhill) results in * net release of free energy * is spontaneous * ΔG is negative ENDERGONIC reaction (uphill) * absorbs energy from its surroundings * non-spontaneous * ΔG is positive: energy is required to drive the reaction |
|
|
What is ATP ? |
* ATP (adenosine triphosphate) is the cellular currency for energy - it provides the fuel for most cellular activities. * ATP has high potential energy and allows cells to do work. * ATP works by phosphorylating (transferring a phosphate group) target molecules. * Carbohydrates and fats are high energy storage moleculesthat, when "burned", are used to generate ATP |
|
|
2 Forms of cellular metabolism? |
Catabolism = breaking down Anabolism= building up Anabolic reactions need ATP |
|
|
How does ATP power cellular work? |
* ATP couples exergonic and endergonic reactions cells manage energy resources by "Energy coupling" = * use of an exergonic process to power an endergonic one * cells need energy for: Transport: pump substance across membrane against a gradient Chemical: pushing of endergonic reactions that require energy Mechanical: muscle contraction/ beating of cilia,etc. |
|
|
Structure and Function of ATP |
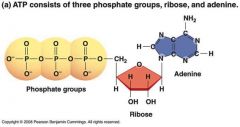
* ATP: 3 phosphate groups, ribose and adenine * high potential energy because thefour - charges in its 3 phosphate groups (O-) repel each other (negative charges repel) |
|
|
Energy Stored in Glucose Is Transferred to ATP |
cell needs energy > carbohydrates participate in exergonic reaction that produces ATP: CH2O + O2 + ADP + Pi → CO2 + H2O + ATP The free energy in ATP is used to drive endergonic reactions and perform cell work. |
|
|
Why does ATP hydrolysis release energy? |
When ATP is hydrolyzed to ADP and inorganicphosphate, there is a large change in free energy Hydrolysis of ATP = Exergonic = energy released ATP → ADP + Pi + Energy Higher ΔG (free energy) → Lower ΔG (higher free energy) (lower energy=more stable) |
|
|
Exergonic vs. endergonic: differences |
Exergonic: Products have less energy Energy released Spontaneous Entrophy (=disorder) increases Endergonic: products have more energy than reactants Energy required Not spontaneous Entrophy (=disorder) decreases |
|
|
What are coupled reactions? |
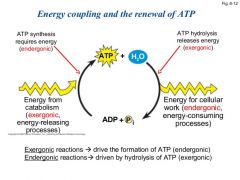
Coupled reactions occur in the same place at the sametime. One reaction provides the energy for a secondreaction that requires energy. |
|
|
ATP Coupling Helps many Cellular Processes |
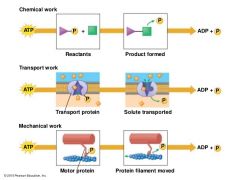
chemical work: reactants + ATP → product formed+ ADP + P Transport work: Transport protein +ATP → solute tranported +ADP + P Mechanical work: motor protein +ATP → protein filament moved + ADP + P |
|
|
What Is a Redox Reaction? |
* Reduction–oxidation reactions (redox reactions) arechemical reactions that involve electron transfer. * When an atom or molecule gains an electron, it is reduced. * When an atom or molecule loses an electron, it is oxidized * Oxidation and reduction events are always coupled — if oneatom loses an electron, another has to gain it.– Electron donors are always paired with electron acceptors. |
|
|
Oxidation is the loss of electrons Reduction is the gain of electrons |
atom is oxidized = has given up some of those negativelycharged electrons = will increaseits overall charge electrons are negatively charged, so an increase inelectrons = a decrease in overall charge (thecompound becomes more negatively charged) = Hence,reduced. |
|
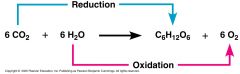
Photosynthesis |
* Co2 becomes reduced (gain of electrons) to sugar water molecules are oxidized (loss of electrons) to O2 Thus, C atoms are reduced + O atoms are oxidized. * This changes chemical energy. * Electrons in the productsare held less tightly. Lessstable, so more potentialenergy. Lower entropy[more order=usable] * Photosynthesis is endergonic: light energy is captured, converted into chemical energy, then the energy is stored in chemical bonds of sugar |
|
|
What Happens When Glucose Is Oxidized? |

Glucose oxygen→ CO2 + water C6H12O6 + 6 O2 → 6 CO2 + 6 H2O + energy Much of the change in free energy is used to synthesize ATP from ADP and Pi. |
|
|
Electrons Are Usually Accompanied by Protons |
Oxidized molecules have lower potential energy They often lose a proton (H+) along with an electron and have many or more C-O bonds,which are stronger & harder to break apart. Follow the hydrogen atoms in redox reactions |
|
|
Nicotinamide adenine dinucleotide (NAD) as an electron carrier |
When NADH is reduced, it readily donates electrons to other molecules and is thus called an electroncarrier and has “reducing power.” |
|
|
Breathing vs. Cellular Respiration |
Breathing supplies oxygen to our cells and removes Co2 Cellular respiration is a series of chemical reactions that oxidize glucose into Co2 and H2O, generating ATP |
|
|
Cellular Respiration: Why do it ? |
It’s like harvesting glucose into smallerenergy packets (like ATP) C6H12O6 one glucose molecule → several ATPs $100 bill → several loonies ($1), plus change to buy gum! |
|
|
Overview of Cellular Respiration |
* All organisms use glucose to build fats, carbohydrates, and other compounds; * cells recover glucose by breaking downthese molecules. * Glucose is used to make ATP through either cellular respiration (produces ATP from a molecule withhigh potential energy, usually glucose), or fermentation. |
|
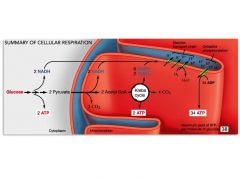
The 4 Steps of Cellular Respiration |
1. Glycolysis – glucose is broken down to pyruvate. 2. Pyruvate processing – pyruvate is oxidized to form acetylCoA. 3. Citric acid cycle – acetyl CoA is oxidized to CO2. 4. Electron transport and chemiosmosis – compounds thatwere reduced in steps 1–3 are oxidized in reactions leading to ATP production |
|
|
The Steps of Cellular Respiration |
Note Three Things About Equation: 1. Bonds in Glucose (glc) Being Broken/Rearranged 2. Glucose is being COMPLETELY oxidized → CO2 3. Oxygen is being reduced → H2O |
|
|
1. Glycolysis: Processing Glucose to Pyruvate (occurs in the cytoplasm) |
Glycolysis ("sugar splitting"), a series of 10 chemical reactions, is the first stepin glucose oxidation. glycolysis= glucose → two 3-carbonmolecules of pyruvate → potential energy released → used to phosphorylate ADP to form ATP |
|
|
2. Pyruvate Processing |
Pyruvate processing is the second step in glucose oxidation. Itis catalyzed by an enzyme. In the presence of O2, pyruvate undergoes a series of reactions that results in the product acetylCoA. |
|
|
3. the citric acid cycle = Krebs cycle (takes place in Mitochondrial Matrix) |
During the third step of glucose oxidation, the acetyl CoAproduced by pyruvate processing enters the citric acidcycle, Each acetyl CoA is oxidized to two molecules of CO2 Some of the potential energy released is used to 1. Reduce NAD+ to NADH. 2. Reduce (FAD) to FADH2(another electron carrier). 3. Phosphorylate GDP to form GTP (later converted toATP) |
|
|
Summary of the products of the citric acid cycle |
from1 acetyl-CoA molecule the citric acid cycle generates: •The acetyl-CoA has been oxidized to 2 molecules of Co2. •3 molecules of NAD were reduced to NADH. •1 molecule of FAD was reduced to FADH2. •1 molecule of GTP (the equivalent of ATP) was produced |
|
|
Summary of glucose oxidization |
For each glucose molecule that is oxidized to 6 CO2, the cell reduces 10 molecules of NAD+ to NADH and 2 molecules of FAD to FADH2, and produces 4molecules of ATP by substrate-levelphosphorylation. |
|
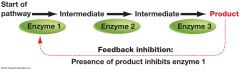
what is Feedback Inhibition ? |
Feedback inhibition: when an enzyme in a pathway is inhibited by the product of that pathway Feedback Inhibition Regulates Glycolysis: During glycolysis, high levels of ATP inhibit an enzyme which catalyzes one of the earlyreactions (glycolysis step 3). Cells that are able to stop glycolytic reactions when ATP is abundant can conserve their stores of glucose for times when ATP is scarce. |
|
|
The Citric Acid Cycle Regulation and Summary |
* The citric acid cycle can be turned off at multiple points, via several different mechanisms of feedback inhibition. * To summarize, the citric acid cycle starts with acetyl CoA and ends with CO2. * The potential energy that is released is used to produce NADH, FADH2, and ATP. * When energy supplies are high, the cycle slows down. |
|
|
Free Energy Changes, NADH, and FADH2 |
* For each glucose molecule that is oxidized to 6 CO2, the cell reduces 10 molecules of NAD+ to NADH and 2 molecules of FAD to FADH2, and produces 4 molecules of ATP by substrate-level phosphorylation. * The ATP can be used directly for cellular work. * However, most of glucose's original energy is contained in the electrons transferred to NADH and FADH2, which then carry them to oxygen, the final electron acceptor. |
|
|
After glycolysis and the citric acid cycle are complete, where is most of the energy that was once contained in the bonds of glucose? a. converted by the cell into ATP via substrate-level phosphorylation b. in NADH and FADH2 that are carrying energy-rich electrons c. in the CO2 that has been released d. converted by the cell into ATP via oxidative phosphorylation |
b. in NADH and FADH2 that are carrying energy-rich electrons |
|
|
The Electron Transport Chain |
* During the fourth step in cellular respiration, the high potential energy of the electrons carried by NADH and FADH2 is gradually decreased as they move through a series of redox reactions. * The proteins involved in these reactions make up what is called an electron transport chain (ETC). * O2 is the final electron acceptor. The transfer of electrons along with protons to oxygen forms water. |
|
|
What is Oxidative phosphorylation ? |
Oxidative phosphorylation is the process in which ATP is formed as a result of the transfer of electrons from NADH or FADH2 to O2 by a series of electron carriers. This process, which takes place in mitochondria, is the major source of ATP in aerobic organisms. |

