![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
179 Cards in this Set
- Front
- Back
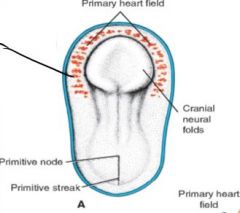
|
Blood islands: develop into horseshoe shaped endothelial tube (primitive heart)
Primary heart field develops into pericardial cavity |
|
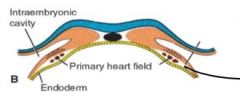
|
Top = splanchnic mesoderm layer (where blood islands form)
Bottom = primary heart field blood islands blue = ectoderm Orange = mesoderm yellow = endoderm |
|
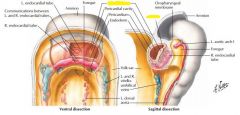
|
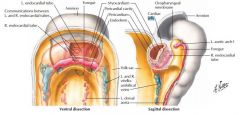
Myocardium = from endocardial mesoderm
Cardiac jelly = acellular matrix around myocardium Epicardium = derived from coelomic wall mesoderm (migrates into place) |
|
|
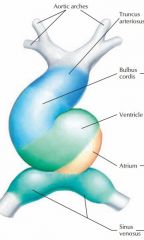
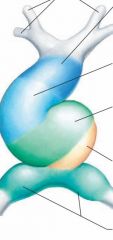
Truncus arteriosus = ascending aorta, pulmonary trunk
Bulbus cordis = Smooth portion of R ventricle, conus arteriosus, aortic sinuses Primitive ventricle = trabeculae carneae Primitive atrium = pectinate muscles (trabeculated muscles) Sinus venosus = coronary sinus, smooth muscle in R atrium Inflow from bottom, outflow from top |
|
|
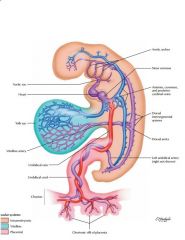
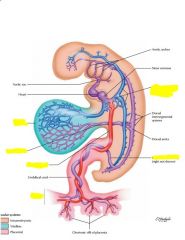
Umbilical veins = from chorion = round ligament of liver
Vitelline veins = from yolk sac = portal system Cardinal veins = caval system |
|
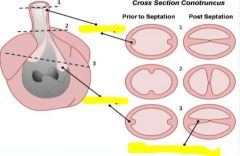
|
Notochord cells move from hindbrain to arches 3, 4, 6 to help with this development
|
|
|
Tetralogy of Fallot
|
1: stenosis of pulmonary trunk
2: hypertrophied right ventricle 3: overriding aorta 4: ventricular septal defect DiGeorge syndrome, environmental, genetic |
|
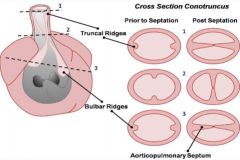
|
Persistent truncus arteriosus (no division between pulmonary trunk and aorta)
|
|
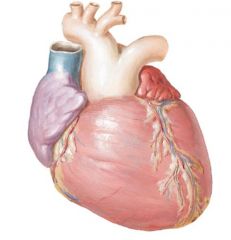
|
D-transposition
RV pumps to aorta, LV pumps to pulmonary No aorticopulmonary septum twist |
|
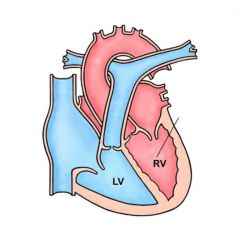
|
L-transposition, functional "corrected transposition"
Viewed as a twist of great vesicles or twist of ventricles |
|
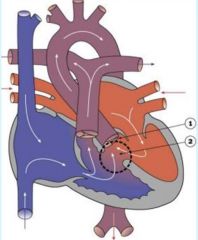
|
Double outlet right ventricle (interventricular septum defect)
|
|
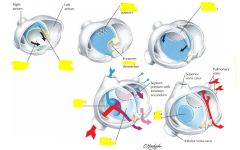
|
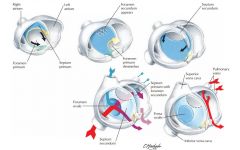
|
|
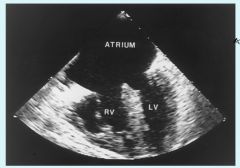
Transesophageal echocardiogram TEE
|
Common atrium (cors triloculare biventricular)
no primary / secondary septum development |
|
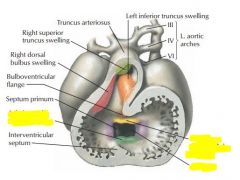
|
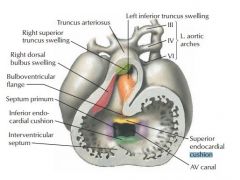
Atrioventricular canal fuses week 5, if no fusion then persistent common AV canal
|
|
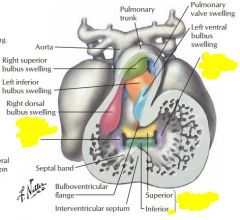
|
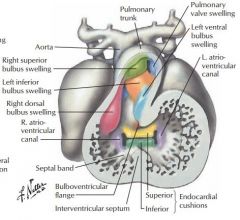
endocardial cushions of atrioventricular valve become AV valves
|
|
|
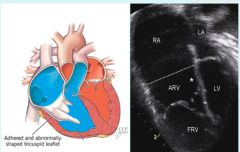
Ebstein's anomaly
Defective tricuspid valve, creates atrialized right ventricle and a small functional right ventricle. |
|
|
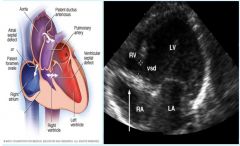
Tricuspid Atresia
right AV canal seals, no valve develops |
|
|
interventricular septum
Derives from crescentic fold (until week 7), composed of muscular and membranous part |
|
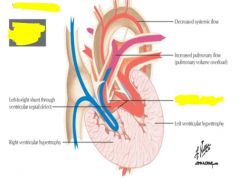
|
venticular septal defect
|
|
|
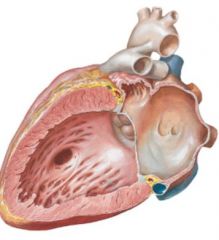
Cor triloculare biatriatum
resorption of myocardial tissue during formation of interventricular septum |
|
|
Node Development
|
1: primitive atrium = temp pacemaker, then sinus venosus takes over
2: SA develops week 5 and part of sinus venosus (part of R atrium) 3: AV node develops from cells in wall of sinus venosus |
|
|
Aortic arch artery 1,3,4,6
|
1: Portion of maxillary sinus
3: Common and internal carotid arteries 4: Right subclavian artery, part of aortic arch 6: ductus arteriosus, proximal pulmonary arteries |
|
|
Intersegmental arteries
|
intercostal arteries, lumbar arteries, common iliac arteries, portions of vertebral, sublcavian, lateral sacral arteries
|
|
|
umbilical arteries
Umbilical Vein |
medial umbilical ligaments
round ligament of the liver |
|
|
Vitelline Arteries
|
Celiac trunk, superior mesenteric artery, inferior mesenteric artery
|
|
|
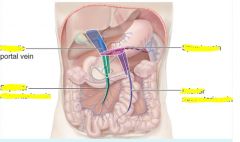
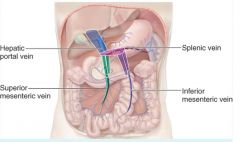
Vitelline veins
|
hepatic portal system, hepatic veins, intrahepatic segment of inferior vena cava
|
|
|
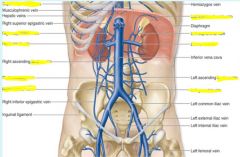
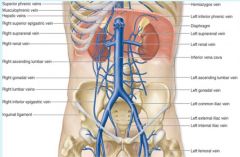
Anterior cardinal veins
|
superior vena cava, brachiocephalic veins, internal jugular veins
|
|
|
Subcardinal veins
|
Lower inferior vena cava, renal / suprarenal veins, gonadal veins
|
|
|
Supracardinal veins
|
azygous system of veins, inferior vena cava between kidney / liver
|
|
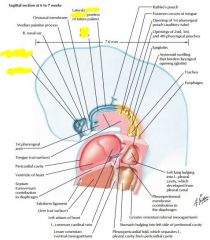
|
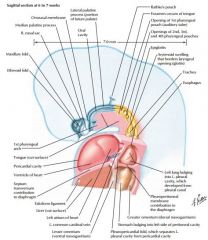
develops as outpocket of gut tube
|
|
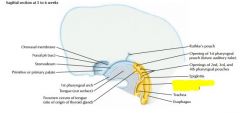
|
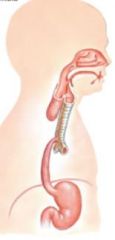
Lower respiratory develops at week 4, starts as median laryngotracheal groove in caudoventral wall at primitive pharynx
deepens into ventricular diverticulum, enlarging into lung bud |
|
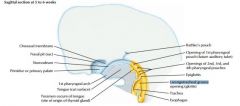
|
|
|
|
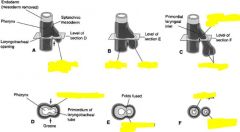
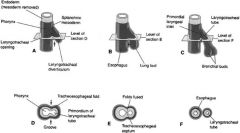
Tracheoesophageal fistula
|
|
|
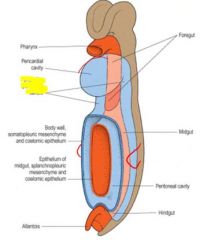
2 bronchial buds grow into pericardioperitoneal cavity, becomes pleural cavities
|
|
|
Lung Bud Development
|
week 5 = each bronchial bud enlarges to primordium of primary bronchus, dividing into secondary and then tertiary bronchi by week 7. Week 24 is 14 divisions, creating respiratory bronchioles. 7 more divisions remaining.
Surrounding mesenchyme creates cartilage, smooth muscle, connective tissue, capillaries |
|
|
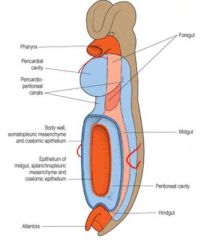
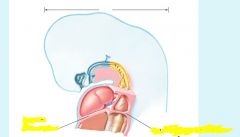
Pleurae
|
Lungs: pleura derived from splanchnic mesenchyme
Thoracic wall: parietal pleura derived from somatic mesoderm |
|
|
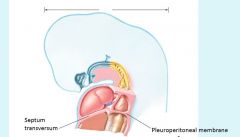
|
|
|
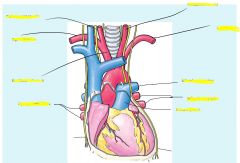
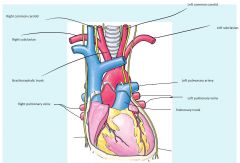
Heart Location
|
Within mediastinum
Oblique from 2nd rib to 5th intercostal space |
|
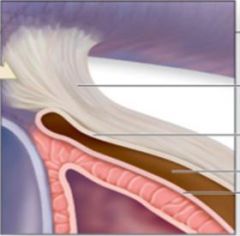
|
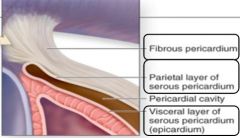
Fibrous pericardium: protection, anchor to diaphragm / great vessels, prevent overfilling
Serous pericardium: parietal layer junction with fibrous pericardium, visceral layer = epicardium, pericardial cavity in between (serous fluid) |
|
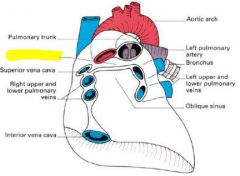
|
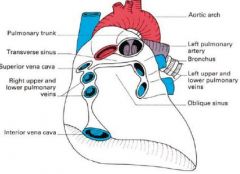
directly under pulmonary trunk / aorta
|
|
|
Cardiac Tamponade
|
tear in myocardium / epicardium, outflow of blood into pericardial cavity leading to heart failure from pressure
|
|
|
Pericardiocentesis
|
Used to treat cardiac tamponade, insert needle into pericardial cavity
|
|
|
Pericarditis
|
may appear as extra liquid on epicardium, or fibrous appearance
|
|
|
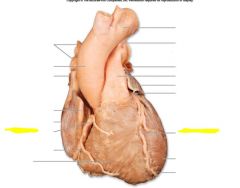
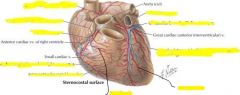
Sternocostal surface of heart
|
R atrium & R ventricle
|
|
|
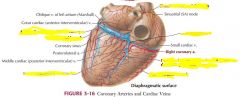
Diaphragm surface of heart
|
posterior & inferior wall of L ventricle
|
|
|
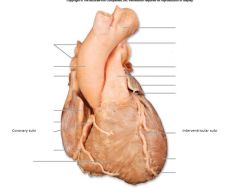
Coronary sulcus: contains coronary sinus
Anterior interventricular sulcus: anterior interventricular branch of L coronary artery and great cardiac vein Posterior interventricular sulcus: posterior interventricular artery and middle cardiac vein |
|
|
Fibrous heart skeleton
|
Collagen (type 1) & elastin
Strengthens pulmonary / aortic trunks Stabilizes, reinforces, provides elasticity |
|
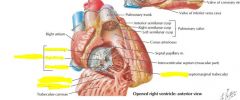
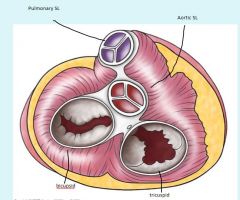
Site of auscultation
|
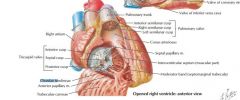
Auscultate at fifth intercostal space, sternal border. Contributes to S1
Cusps = leaflets |
|
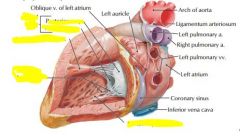
Site of auscultation
|
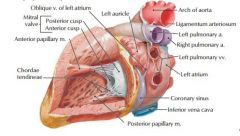
Auscultate at 5th intercostal space, medial to left midclavicular line. Contributes to S1
Bicuspid = mitral. Cusps = leaflets |
|
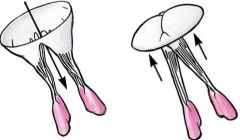
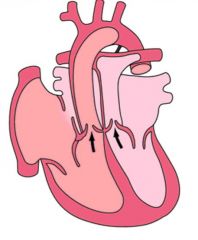
AV function
|
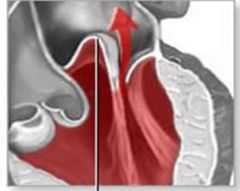
papillary muscles contract and pull chordae tendinae during ventricular contraction in order to keep valve closed and prevent prolapse (as depicted) / regurgitation of blood
function based off of pressure |
|
|
Pulmonary & Aortic Semilunar Valve auscultation sites
|
Pulmonary: L 2nd intercostal space
Aortic: R 2nd intercostal space Both contribute to S2, possible split due to late pulmonary valve closure from decreased thoracic pressure during breath (longer for pulmonary trunk pressure to exceed R ventricle pressure) |
|
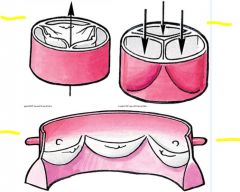
Semilunar valve function
|
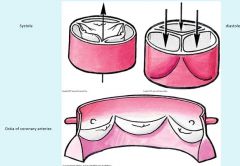
Valves function based off of gravity acting on blood rather than pressure
|
|
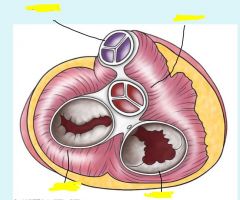
|
|
|
|
|
|
|
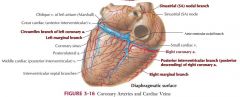
|
|
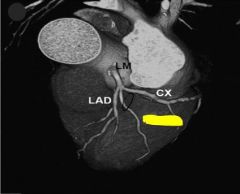
|
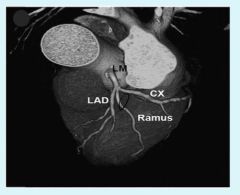
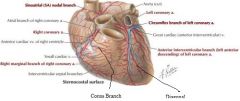
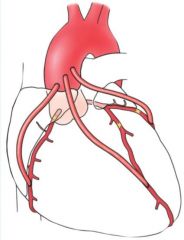
Left coronary artery can trifurcate instead of bifurcate, into anterior interventricular branch, circumflex branch, as well as the variation of a "Ramus Branch" in between
|
|
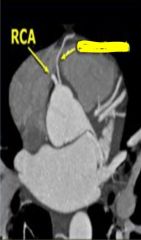
|
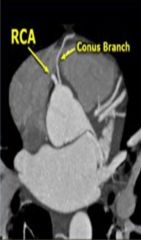
Conus branch normally extends off of the right coronary artery, however a variant can extend directly off of aorta and is referred to as the third coronary artery
|
|
|
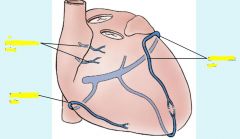
Coronary artery bypass surgery
Great Saphenous vein or internal thoracic vein can be used as candidate vessels. |
|
|
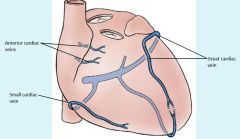
anterior cardiac veins unique because drain directly into right atrium rather than cardiac sinus,
|
|
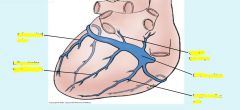
|
Coronary Sinus empties through the Thebsian valve into the right atrium.
|
|
|
Venae Cordis Minimae
|
smallest cardiac veins which empty myocardium, most abundant in right atrium, least abundant in left ventricle
|
|
|
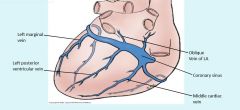
L sinus venosus horn vs R sinus venosus horn
|
L sinus venosus becomes coronary sinus
R sinus venosus becomes sinus venarum (smooth portion of R atrium) |
|
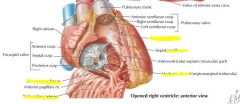
|
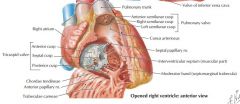
|
|
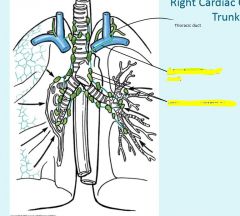
|
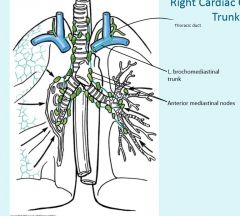
Thoracic duct empties into venus angle
|
|
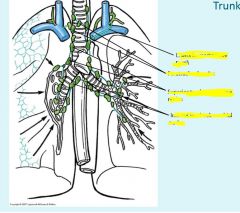
|
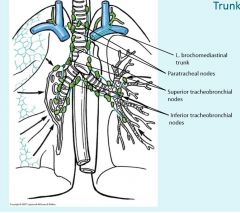
|
|
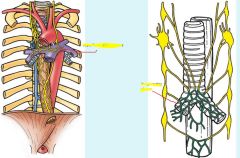
|
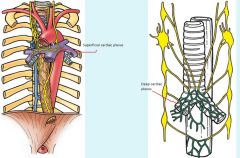
|
|
|
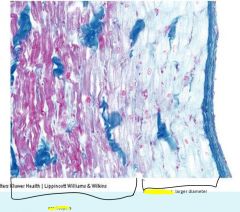
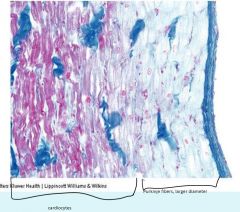
Cardiocytes
|
Identified by intercalated discs made with adhesions
|
|
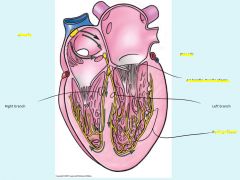
|
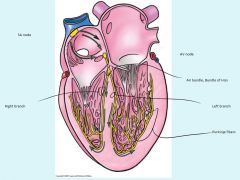
|
|
|
|
|
|
|
|
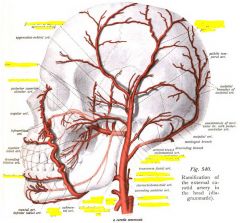
|
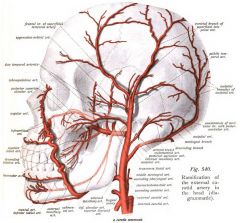
external maxillary artery = facial artery
Maxillary artery 1st part: Middle meningeal, inferior alveolar 2nd: buccinator 3rd sphenopalantine |
|
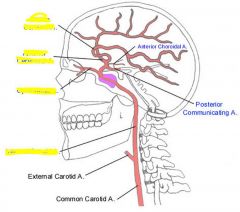
|
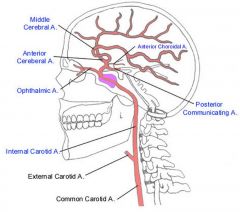
Forms portion of circle of willis
|
|
|
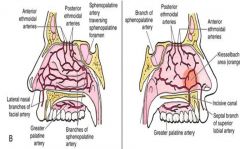
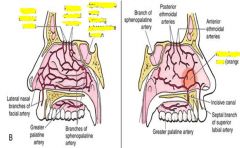
Anterior kiesselbach area = majority of epistaxis
posterior from ethmoidal arteries or sphenopalatine arteries may result in a more serious epistaxis |
|
|
|
|
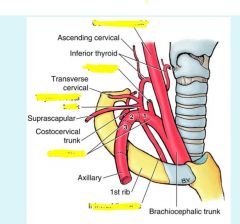
|
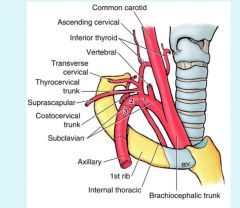
vertebral artery runs up through transverse froamen of cervical spine into skull
|
|
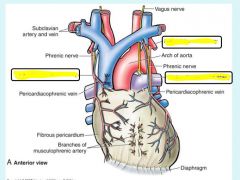
|
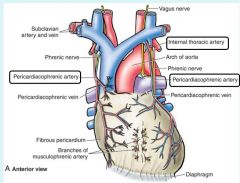
Pericardiacophrenic artery stems from internal thoracic artery
|
|
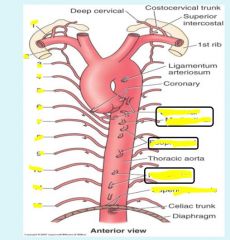
Visceral branches of thoracic aorta
|
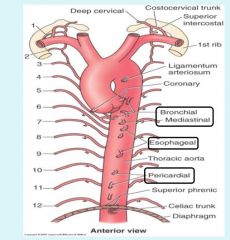
|
|
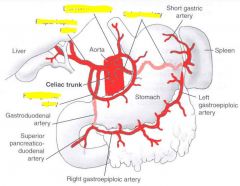
|
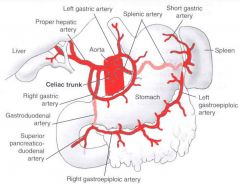
|
|
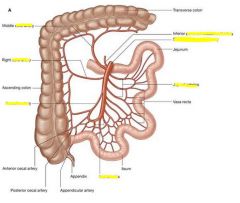
|
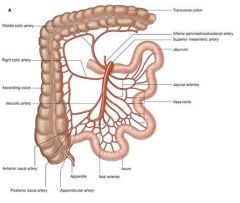
superior mesenteric artery can actually obstruct duodenum because it lies directly over it
can also compress left renal vein |
|
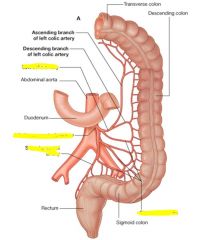
|
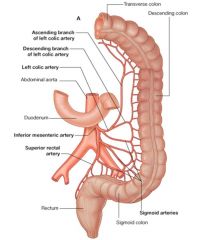
|
|
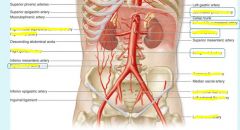
|
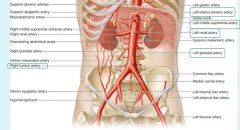
internal iliac supplies, bladder, walls of pelvis, genitalia
|
|
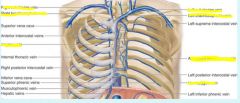
|
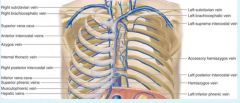
|
|
|
|
|
|
|
|
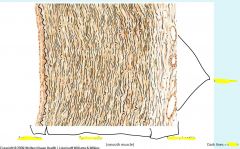
|

|
|
|
Long QT syndrome genetic concepts
|
Inheritable
Locus heterogeneity (single phenotype could be caused by many different loci mutations). Panethnic Incomplete penetrance Variable expressivity genetic susceptibility to meds |
|
|
Long QT syndrome phenotype
|
Long QT interval: >470 male, >480 female, normal ≤ 400
T wave abnormalities: tachyarrhythmias, torsades de pointe (ventricular tachycardia) Syncope, sudden death |
|
|
KCNQ1 protein
|
Interacts with KCNE proteins to form potassium channels
Found in inner ear, kidney, lung, GI, heart Mutations cause Romano-Ward syndrome Some mutations linked with SIDS |
|
|
Romano-Ward Syndrome (elongated QT syndrome)
|
most common form, autosomal dominant (incomplete penetrance, variable expressivity)
point mutations, insertions, deletions in KCNQ1 (>140 known) potassium channels work at reduced efficiency, prolong the amount of time for repolarization. Also lower threshold for depolarization. |
|
|
KCNH2 protein
|
interacts with KCNE2 to form potassium channel
mutations also cause Romano-Ward syndrome |
|
|
Jervell & Lange-Nielsen syndrome (long QT syndrome)
|
Less common, autosomal recessive
Result of a mutation creating short nonfunctional KCNQ1 protein, potassium channels do not function More severe than Romano-Ward syndrome |
|
|
Long QT syndrome prognosis
|
syncope from arrhythmias = common symptom, 30-50% never show syncope however.
Cardiac episodes frequent in preteens-20s, risk decreases with age. 30% are within normal range, stress EKG needed to identify elongated QT very dangerous to prescribe QT elongating drugs |
|
|
Congenital Heart defect genetic concepts
|
Multifactorial inheritance (many contributing genes and environment)
Single gene or chromosome defect (usually associated with other defects) Somatic mosaicism (problem may occur during embryogenesis, not all cells will be affected) Empiric risks (population risk given rather than personalized risk) Carter effect (if one sex is less susceptible and you have a sibling of that sex with the disease, then you are at a higher risk) |
|
|
Syndromic congenital heart disease
|
Uncommon and due to single gene defects, therefore associated with other defects in development
Noonan, Alagille, Holt-Oram, Simpson-Goabi-Behmel (don't memorize probably) recurrence risk is the same from one population tothe next |
|
|
Isolated Congenital Heart Defects
|
More common, many forms with different empiric risks
Flow lesions, defects in cell migration or death, abnormal extracellular matrix, defects in targeted growth Multifactorial inheritance, low penetrance, likely environmental factors, polygenic recurrence risk is different from one population to the next two hit hypothesis may also apply somatic mosaicism has been observed in some forms 22q11 = tetralogy of fallot |
|
|
Multifactorial inheritance concepts
|
higher risk if more family members affected
higher risk if severe expression in family member higher risk if family member is of less commonly affected sex (Carter effect) risk decreases for more distant relatives additive risk |
|
|
Cardiac defects in
Trisomy 13 (patau) Trisomy 18 (edward) Trisomy 21 (down) |
13 = right sided heart rather than left, atrial septal defect, patent ductus arteriosus, ventricular septal defect
18 = atrial septal defect, patent ductus arteriosus, ventricular septal defect 21 = atrioventricular septal defect, atrial septal defect, patent ductus arteriosus, ventricular septal defect |
|
|
|
|
|
Chest Radiograph
|
common, cheap, fast
Good for pulmonary / bony structures Bad for heart / soft tissue |
|
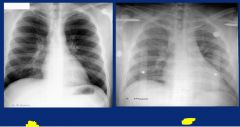
Chest radiograph direction
|
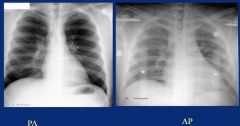
PA = posterior to anterior (normal)
AP = anterior to posterior (sick, bedridden patients) Anything further from film is magnified, so you would prefer to have the heart closer to the film (PA) |
|
|
|
|
|
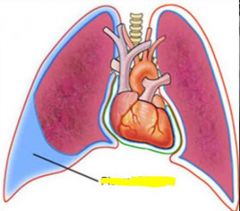
Pleural calcification (asbestos)
|
|
|
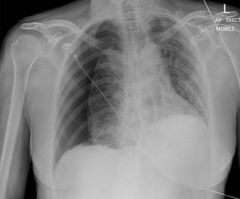
Pneumothorax (collection of air between visceral and parietal pleura)
Displaces the heart, compressing blood supply and causing cardiac arrest May result from trauma or central line placement (subclavian vein near pleura) Air hides shadows, appears blacker than usual |
|
|
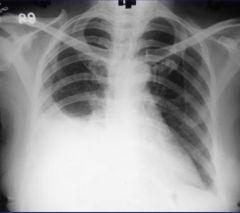
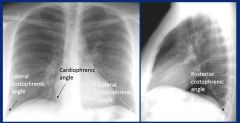
Pleural effusion
Liquid collects first in the costophrenic angles due to gravity, may traverse upwards due to surface tension. Appears white, loses sharp angles at bottom of lung. Ultrasound good for localizing spot for chest tube due to the presence of liquid rather than air (always insert above rib, never below) |
|
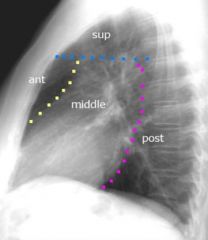
Sections of the mediastinum
|
Superior: thymus, superior VC & brachiocephalic veins, aortic arch / branches, trachea, esophagus, vagus & phrenic nerves, thoracic duct
Anterior: thymic remnants, areolar tissue, lymph nodes. Middle: heart, ascending aorta, lower half of superior VC with azygos vein, bifurcation of trachea & two bronchi, pulmonary artery, R&L pulmonary veins, phrenic nerve, lymph nodes, pericardiacophrenic vessels. Posterior: Descending aorta, esophagus, thoracic duct, azygos veins, vagus nerve |
|
|
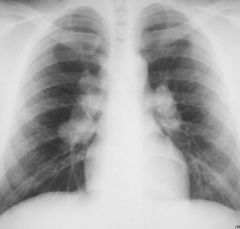
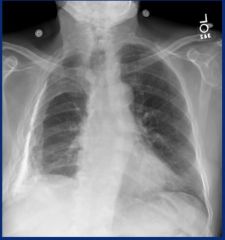
hilum, made up of pulmonary artery, bronchus, pulmonary vein, lymph nodes
may become enlarged due to enlarged lymph nodes (picture above: hilar adenopathy) |
|
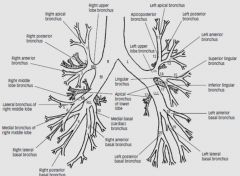
|
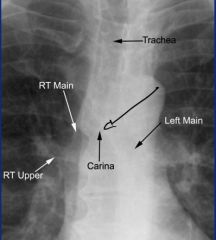
Right bronchus is wider and shorter than left
Endotracheal tube should be 5cm above carina, can find carina by drawing 45 degree line from aorta. If it falls into a bronchus (more often the right due to size) it will collapse the opposite lung. |
|
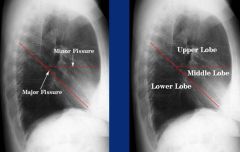
|
Right lung (three lobes)
Left long has only one pulmonary fissure (major) and is two lobed |
|
|
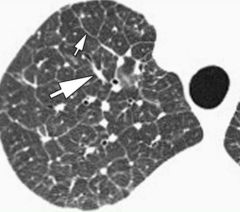
Secondary pulmonary lobule, smallest lung unit surrounded by connective tissue septum.
1-2cm, 5-15 pulmonary acini containing alveoli. Center is supplied by terminal bronchiole with centrilobular artery Interlobular septum supplied by pulmonary veins and lymphatics |
|
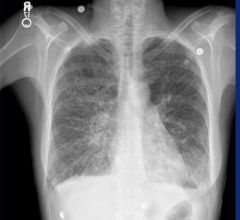
|
Interstitial process (such as pneumonia, tuberculosis, silicosis)
Lines referred to as Kerley Lines |
|
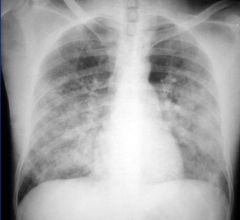
|
Pulmonary edema
Presents as bilateral alveolar infiltrate, appears as batwing |
|
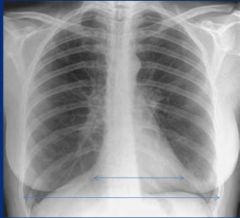
|
Cardiothoracic ratio
ratio should be less than 50% (remember should be done on a PA chest) |
|
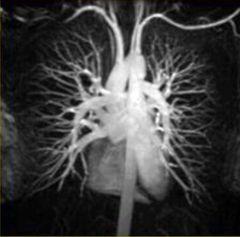
|
MRA: magnetic resonance angiography, no need for catheter with contrast
|
|
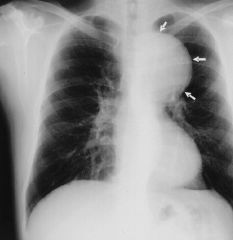
|
aortic aneurysm of the ascending aorta
|
|
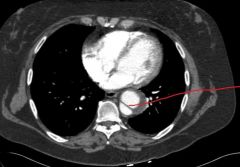
|
aortic dissection
|
|
|
Central line placement
|
tip of catheter ends at superior vena cava, must make sure the catheter doesn't track up one of the jugular veins
|
|
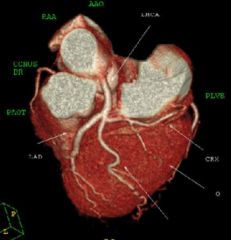
|
Coronary CTA (computer tomography angiography)
Used to search for occlusions or other vascular problems |
|
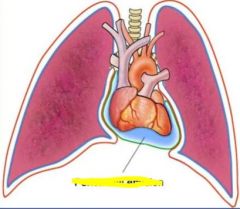
|
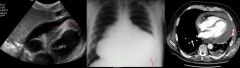
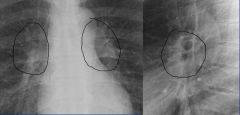
Pericardial effusion
cannot tell on chest radiography (middle) if due to chamber enlargement or pericardial effusion. Easier to see on ultrasound (left) or CT (right) |
|
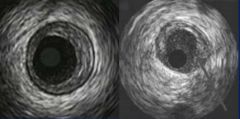
|
intravascular ultrasound. Left = normal, right = occluded from plaque buildup
|
|
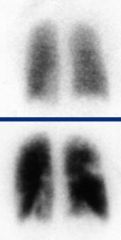
|
Ventilation perfusion scan (V/Q)
ventilation imaged with xenon perfusion with technetium labelled albumin. White spots = defects in perfusion |
|

|
myocardial perfusion imaging, darker areas = ischemic due to not taking up radioactive tracer
good to do under stress to identify ischemic areas |
|
|
Body fluid compartments
|
80% circulating fluid in interstitium
20% in plasma |
|
|
Flow equation
|
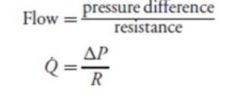
|
|
|
Resistance equation
|
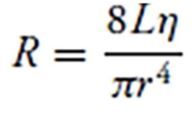
L = tube length
n = fluid viscosity r = internal radius of tube |
|
|
Flow equation with full resistance equation
|
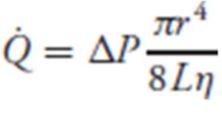
|
|
|
peripheral vascular resistance
Rt = RA+ Ra + Rc + Rv + RV |
|
|
Total peripheral resistance
|
Organs are arranged in parallel (except GI and liver in series)
1/TPR = 1 / Rorgan + 1 /Rorgan.....+ 1/Rorgan Removing one source of parallel resistance will increase resistance |
|
|
Cardiac output
|
CO = Stroke volume X heart rate
Average = 5L /min Blood volume = 5.5 - 6L |
|
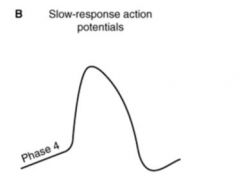
|
Pacemaker cells, slow response
Funny channels open, sodium moves in, potassium out T calcium channels open, calcium moves in L calcium channels open, rapid depolarization Potassium channels open, potassium moves out, repolarization |
|
|
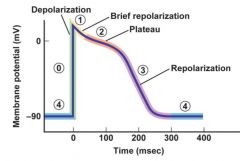
Contractile cell action potential (ventricular action potential, fast response)
0 = sodium channels open, sodium moves in rapid depolarization 1 = sodium channels close, sodium movement slows 2 = potassium inward channels open, calcium L channels open. Calcium moves in causing plateau against potassium moving out 3 = potassium delayed channels open, calcium L channels close. Potassium moves out, repolarizing 4 = potassium channels open, sodium and calcium closed. |
|
|
Autonomic influences on heart
|
Sympathetic = B1 norepinephrine = increased heart rate, supplied to SA node, AV node, ventricular myocardium (contractility). Increases open state of calcium channels, faster spontaneous depolarization
Parasympathetic = M2 acetylcholine = decreased heart rate, supplied to SA and AV node from vagus nerve. More open K+ and more closed Ca+, decreased rate of spontaneous depolarization / hyperpolarizes. Blood vessels: alpha 1 norepinephrine = contraction, beta 2 dilates some. Both sympathetic |
|
|
Cardiovascular transport equation
|
![Convective transport
X = transport rate, Q = flow rate, [X] = concentration](https://images.cram.com/images/upload-flashcards/88/20/63/4882063_m.jpg)
Convective transport
X = transport rate, Q = flow rate, [X] = concentration |
|
|
Fick principle (utilization rate)
|
![Xtc = transcapillary efflux rate
Q = blood flow
[X]a, v = arterial / venouc concentration](https://images.cram.com/images/upload-flashcards/88/20/66/4882066_m.jpg)
Xtc = transcapillary efflux rate
Q = blood flow [X]a, v = arterial / venouc concentration |
|
|
Filtration vs reabsorption
|
filtration = net fluid movement out of capillary
reabsorption = fluid movement into capillary |
|
|
Starling forces / equation
|
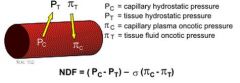
NDF > 0 = filtration
NDF < 0 = reabsorption edema occurs if constant filtration (either increased capillary hydrostatic pressure, or decreased capillary osmotic pressure) |
|
|
Cardiac cycle
|
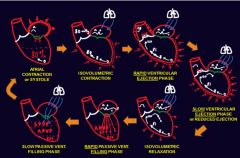
Atrial contraction, isovolumetric contraction, rapid ventricular ejection, slow ventricular ejection, isovolumetric relaxation, rapid passive filling, slow passive ventricular filling
|
|
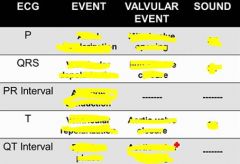
|
|
|
|
Stroke volume
Ejection fraction |
SV = End diastolic volume - end systolic volume
Ejection fraction = Stroke volume / end diastolic volume |
|
|
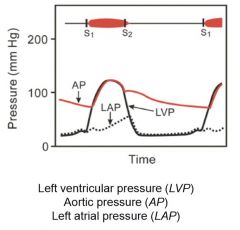
S1
S2 S3 S4 |
S1= closure of mitral / tricuspid valves (isovolumetric contraction)
S2 = closure of semilunar valves (isovolumetric relaxation) S3 = occurs during rapid filling of ventricle if ventricle is very compliant S4 = occurs with atrial contraction against stiff ventricle (concentric hypertrophy, or myocardial infarction) |
|
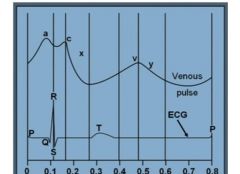
Venous pulse
|
Varies with right side of heart and respiratory cycle. Read at end of expiration when intrapleural pressure is close to 0
a = right atria contraction c = tricuspid valve bulging due to isovolumetric contraction x = atrial relaxation v = filling of atrium during ventricular contraction y = rapid filling of right ventricle upon opening of tricuspid valve |
|
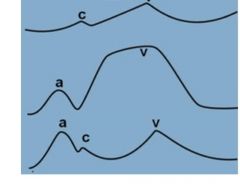
|
First = atrial fibrillation, no full contraction so pressure never builds up except during ventricular systole
Second = tricuspid valve insufficiency causing backflow of blood during systole increasing venous pressure Third = stenotic tricuspid valve, atrial pressure is increased against the stiff valve |
|
|
|
|
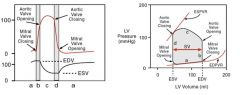
|
a = ventricular filling
b = isovolumetric contraction c = ventricular ejection d = isovolumetric relaxation |
|
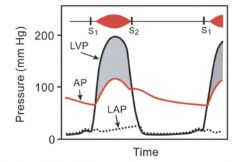
|
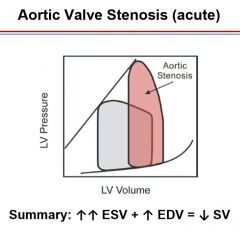
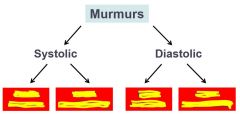
Aortic valve stenosis (left ventricle must create more pressure against a stiff valve). Pressure gradient above aortic pressure
Crescendo-decrescendo murmur heard loudest over 2nd intercostal space at sternal border |
|
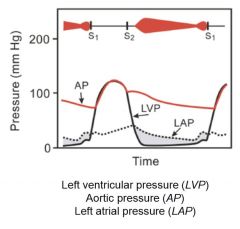
|
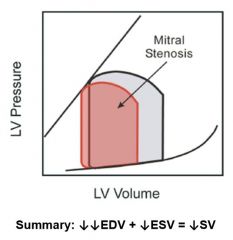
Mitral valve stenosis, left atrial pressure higher than 15, blood builds up against stiff mitral valve and not as much is pushed through. Pressure gradient is higher than left ventricular pressure during diastole.
S1 may be louder due to closure of stiff valve, S2 is followed by snap opening of mitral valve followed by rumbling deep murmur. Best heard over apex |
|
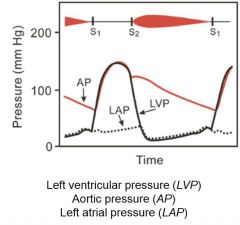
|
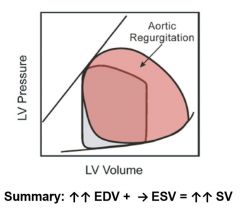
Aortic Valve regurgitation. No pressure gradient, however pressure is still high. Blood leaks back and increases EDV, larger fall off of aortic pressure during diastole.
Murmur begins with diastole and is described as a decrescendo. |
|
|
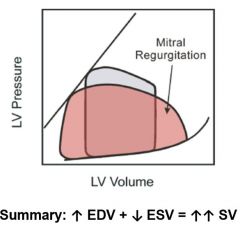
Mitral regurgitation. No pressure gradient but heightened left atrial pressure. Mitral valve leaks allowing blood back into left atrium, extra blood increases EDV which increases SV. More complete emptying of the heart decreases ESV also increasing SV.
Murmur is heard during systole, is holosystolic (high frequency plateau, no crescendo-decrescendo) |
|
|
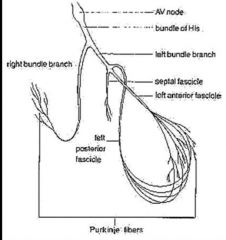
Because of reduced conductance through myocardium, contraction follows action potential by roughly 250msec
|
|
|
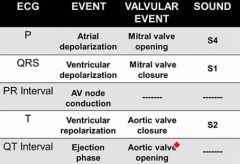
EKG box values
|
Horizontal: 1 large = .2sec
1 small = .04 sec Vertical: 1 large = .5mV one small = .1mV |
|
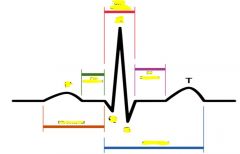
|
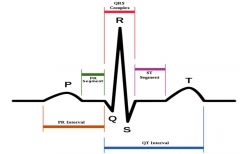
segment = connect two waves
interval = contain at least 1 wave |
|
|
P wave
|
Left half of wave = right atria (first to depolarize)
Right half of wave = left atria |
|
|
PR Segment
|
Caused by AV node, creating a pause
|
|
|
QRS
|
Ventricular depolarization, septal fascicle depolarizes septum first, then anterior and posterior fascicles depolarize the rest of the ventricle
Q = first downward deflection before any up R = first upward deflection S = first downward deflection after any up |
|
|
T wave
|
ventricular repolarization (atrial repolarization is hidden by QRS)
|
|
|
EKG lead
|
Positive deflection = depolarization towards positive electrode or repolarization away from positive electrode
Negative deflection = depolarization away from positive electrode, or repolarization toward positive electrode. Result = QRS and T wave usually same deflection because repolarization starts where depolarization stopped Biphasic wave = depolarization perpendicular to electrode |
|
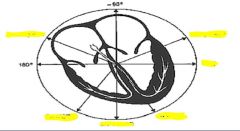
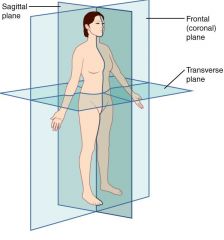
Limb leads
|
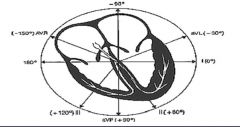
3 standard and 3 augmented leads
All measure in frontal (coronal) plane I = Right to left arm II = R arm to left leg III = L arm to right leg AVL = All to left arm AVR = All to right arm AVF = All to feet |
|
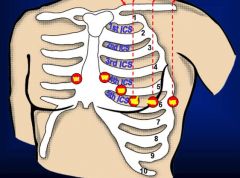
Precordial leads
|
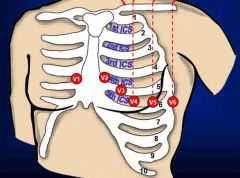
6 leads placed, measure in the transverse plane (horizontal)
|
|

|

|
|

|
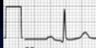
Standard for 1mV set at beginning of each EKG, usually 10mm. Sometimes 1/2 standard for very high deflections.
Speed of paper can also vary |
|

|
HR = 300 / # large boxes
Or 300, 150, 100, 75, 60, 50 |
|
|
Rhythm
|
Rate between 60-100 (bradycardia or tachycardia if beyond)
Every P wave followed by QRS (if no P wave then QRS didn't originate from SA node) P wave upright in leads I & II. Inverted in aVR |
|
|
Axis
L axis deviation? I, II, aVF R axis deviation? I, II, aVF |
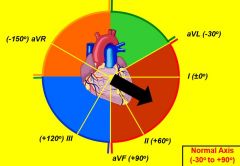
Determining the ventricular axis (vector of the electric conductance)
QRS should be positive in I & II L axis deviation will be positive in I but negative in II, negative aVF R axis deviation will be positive in II but negative in I, positive aVF |
|
|
Intervals (PR, QRS, QT)
|
PR interval = .12 - .20 sec, 3-5 little boxes
QRS = less than .10 sec, or less than 2.5 little boxes QT interval less than half of R-R interval |
|
|
Morphology
|
Amplitude and duration of waves
|
|
|
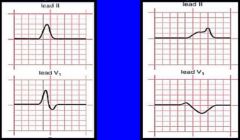
P wave morphology
|
should be upright in II, biphasic in V1
|
|
P wave
|
Right atrial enlargement, left atrial enlargement
(Taller, wider) In R atria enlargement, L atria still needs to contract and is normal so duration is the same but R atria has more myocytes so increased voltage. In L atria enlargement right atria has already contracted and L atria takes longer to contract, so the P wave is elongated |
|
|
QRS morphology
|
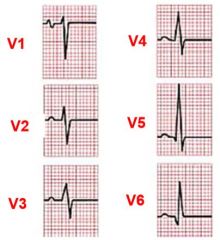
less than .1 seconds, variable amplitude
conduction disturbance = wider than normal ventricular hypertrophy = taller than normal Q wave = normally from septal fascicle V1/2 = negative, V3/4 = biphasic, V5/6 = positive If 1/2 are positive, could be R ventricular hypertrophy |
|
|
ST morphology
|
flat and at isoelectric point
Used for diagnosing: ischemia (depression), infarction (regional elevation), pericarditis (diffuse elevation) |
|
|
T wave morphology
|
Ventricular repolarization, should be same direction as QRS complex
Diagnostic: ischemia (peaked & inverted), hyperkalemia (peaked) |
|
|
Mean arterial pressure short term regulation
|
detected by baroreceptors, integrated in brainstem, controlled by autonomic NS, effecting heart & blood vessels
|
|
|
Baroreceptors
|
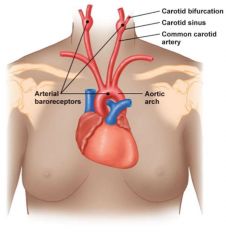
Have a baseline frequency of action potentials, increased pressure increases frequency, decreased pressure decreases frequency
|
|
|
Medulla oblongata role in pressure
|
Medulla oblongata = cardiovascular control center, uses arterial baroreceptors, low pressure baroreceptors, chemoreceptors, proprioceptors, and higher brain centers to control output via ANS.
Low pressure receptors located in large veins and in right atrium Parasympathetic: SA and AV node Sympathetic: SA & AV node, ventricular myocardium, arterioles, veins (increased tone) |
|
|
Afferent nerves for baroreceptor information
|
Carotid sinus baroreceptors: cranial nerve 9 (glossopharyngeal)
Aortic Arch baroreceptors: cranial nerve 10 (vagus) |
|
|
Long term blood pressure regulation
|
Happens via blood volume using renin angiotensin aldosterone system (RAAS)
Juxtaglomerular cells around afferent arterioles in the kidney sense decreased blood pressure, decreasing NaCl levels, or b1 receptor activation (sympathetic). They secrete renin in response to these stimuli, converted to angiotensin 1 by angiotensinogen (from liver). Converted to angiotensinogen 2 by ACE in pulmonary capillaries. Angiotensin 2 stimulates aldosterone release. |
|
|
Angiotensinogen effects
|
vasoconstriction of arterioles
aldosterone secretion, increased sodium reabsorption in late distal tubules & collecting ducts Anti-diuretic hormone secretion, increasing water reabsorption in late distal tubules / collecting duct Thirst stimulation in hypothalamus |
|
|
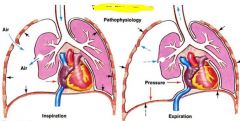
Thoracic / abdominal changes in pressure during respiration
|
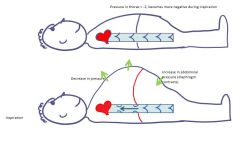
|
|
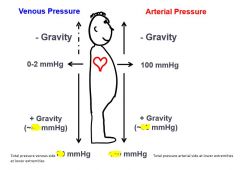
|
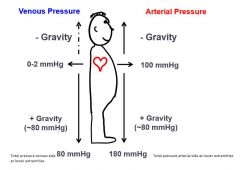
This is why highest arterial pressure = femoral arteries while standing up, due to gravity.
|
|
|
Reaction to exercise
|
skeletal muscle blood flow x10, heart x3, brain unchanged, kidney 1/4th, skin x4, splanchnic organs 1/2
vasodilation of skeletal arterioles due to adenosine, potassium, lactate PO2 and PCO2 are unchanged in arterial system due to increased ventilation, venous system PO2 is decreased and PCO2 is increased pH may decrease due to lactic acid MAP increased Blood flow large increase, TPR large decrease (dilation of skeletal muscle arterioles) Pulmonary circulation = flow increase, vascular resistance large decreases, increased blood volume, increased perfused capillaries |
|
|
Effect of physical conditioning
|
Increased arteriole number, decreasing resistance during exercise
Increased capillary density Increased oxidative enzymes in mitochondria |

