![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
60 Cards in this Set
- Front
- Back
|
5 Basics of Cell Chemistry |
1. Carbon 2. Water 3. Selectively Permeable Membranes 4. Polymerization (synthesis) of small molecules. 5. Self-Assembly |
|
|
Carbon |
Chemistry of the cell is the chemistry of carbon. Chemistry (Organic) of Carbon containing compounds. Backbone of biologically important molecules. |
|
|
Fundamental properties of Carbon |
Valence of four. Outermost electron orbital missing for electrons. Atoms may share electrons by covalent bonds. |
|
|
Different types of bonds |
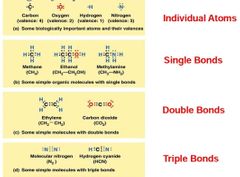
|
|
|
Carbon containing molecules are stable |
Carbon valence and low atomic weight - provide versatility and stability for carbon containing molecules.
Bond energy provide stability.
Bond energy - amount of energy required to break 1 mole of such bonds.
Stability - large energy required to break C-C bonds (83 kilocalories per mol). |
|
|
Diversity of Carbon Molecules |
The tetravalent nature of the carbon atom provides great diversity, especially for biological molecules.
Linear or circular molecules.
With hydrogen - forms hydrocarbon.
Hydrocarbons are insoluble in water. |
|
|
Simple Hydrocarbon Molecules |
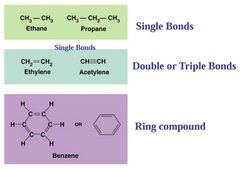
|
|
|
Oxygen, Nitrogen, Phosphorus, and Sulfur. |
Part of functional groups. Functional groups confer water solubility and chemical reactivity on the molecules. Phospholipid molecules with hydrocarbon tails provide non aqueous nature of the membrane. |
|
|
Common Functional Groups |
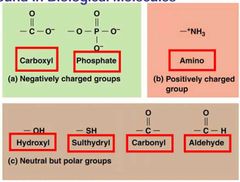
Provide physical and chemical properties - water solubility and chemical reactivity. |
|
|
Carbon can form Stereoisomers |
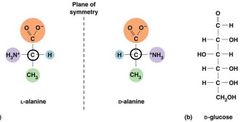
Tetrahedral structure - geometric symmetry.
Asymmetric carbon atom.
Two configurations. |
|
|
Water - Unique Properties |
Polarity
Cohesiveness
Temperature-stabilizing Capacity
Solvent |
|
|
Water - Polarity |
One part partial negative and partial positive charge because O's more electronegative atom draws electrons towards that region.
Uneven distribution old charge within a molecule. |
|
|
Water - Cohesiveness |
H²O molecule affinity for each other by Electronegative (Oxygen) and Electropositive (Hydrogen) charges. |
|
|
Water molecules are polar |
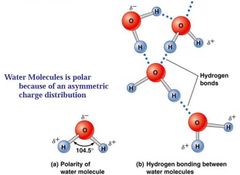
Oxygen atom in the water molecule is negative. Draw electrons towards it. Leaving the other end (+) charge top draw Hydrogen atoms. |
|
|
Water molecules are cohesive |
Electronegative oxygen atom if one molecule associated with the electropositive hydrogen atom of the adjacent molecules.
Property of Hydrogen bonds - being broken and reformed.
ICE - Hydrogen bonds - extensive and rigid.
Cohesivenes accounts for surface tension, high boiling point, high specific heat, high heat of vaporization, etc. |
|
|
High Temperature Stabilizing Capacity |
Due to Hydrogen bonding between adjacent water molecules - provide this property.
Specific Heat - amount of heat absorbed by a substance/gram to raise 1° Centigrade.
High heat of Vaporization - energy required to convert one gram of liquid into vapor. |
|
|
Specific Heat |
Amount of heat absorbed by a substance/gram to raise 1° Centigrade. |
|
|
High heat of Vaporization |
Energy required to convert one gram of liquid into vapor. |
|
|
Water - An Excellent Solvent |
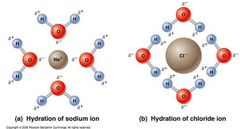
Solvent - Fluid. Solute - substance dissolved in the solvent. Solute's Affinity Hydrophobic - water fearing. Hydrophilic - water loving. |
|
|
Selectively Permeable Membranes (Part 1) |
Membrane - hydrophobic permeability barrier consisting of Phospholipids Glycolipids Membrane proteins Sterols - Cholesterol (animal cell) - Phytosterol (plant cell) - Ergosterol (fungus) |
|
|
Selectively Permeable Membrane (Part 2) |
Most membrane cells are not simply hydrophobic or hydrophilic. Amphipathic Phospholipid molecules of two long nonpolar tails. |
|
|
Basic structure of lipid bilayer |
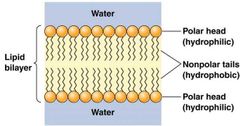
|
|
|
Membranes and Membrane Structures Visual |
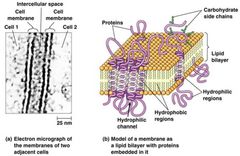
|
|
|
Membrane - Selectively (Part 1) |
Hydrophobic interior - permeable to nonpolar molecules. - Impermeable to most polar molecules and ions.
Cellular constituents - polar or charged - no affinity to interior.
Charge on an ion and hydration sphere - determines the permeability of a membrane. |
|
|
Membrane - Selectivity (Part 2) |
Hydrophilic Channel - specialized transmembrane protein.
Carrier Protein - binds a solute/molecule and transport across.
Transport - ions, proteins, solutes, gases regulated by cellular needs - selectively permeable. |
|
|
Essentials of Synthesis by Polymerization |
Cellular Structure - Ribosomes, Chromosomes, Membranes, Cell Walls etc. are all made up of linear polymers known as Macromolecules.
Macromolecules - Nucleic Acids (DNA and RNA), Polysaccharides (Glycogen, Starch, and Cellulose), Proteins. |
|
|
Macromolecule Hierarchy |
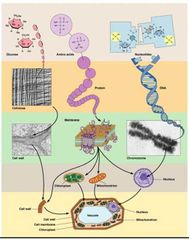
Level 1 - Small Organic Molecules Level 2 - Macromolecules Level 3 - Supramolecular Structures Level 4 - Organelles and other Structures. Level 5 - The Cell |
|
|
Macromolecule Hierarchy - Level 1 |

Water soluble Organic molecules obtained from the other cells or synthesized from simple nonbiological molecules. - Carbon dioxide, Ammonia, or Phosphate ions. |
|
|
Macromolecule Hierarchy - Level 2 |
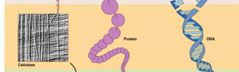
Biological Macromolecules - polymerization of organic molecules. - Nucleic Acids (DNA, RNA), Proteins, Polysaccharides. |
|
|
Macromolecule Hierarchy - Level 3 |
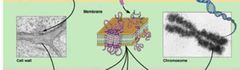
Assembly of supramoleculer structures – Cell wall, membranes, chromosomes. |
|
|
Macromolecule Hierarychy - Level 4 |
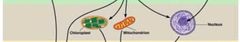
Intercellular structures. - Organelles |
|
|
Macromolecule Hierarchy - Level 5 |
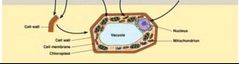
The Cell |
|
|
Synthesis of Biomolecules - Polymerization |
Macromolecules - building blocks of cellular components; mitochondria, membranes, chromosomes, and ribosomes. - Linear and Branched Linear - DNA, RNA, Proteins. Starch, Cellulose, and Glycogen. |
|
|
General Principal of Macromolecule |
Macromolecules that are responsible for most of the form and order characteristic of living systems are generated by the polymerization of small organic molecules. |
|
|
Three Important Macromolecules |
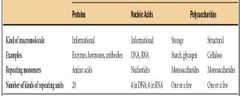
Proteins Nucleic Acids Polysaccharides |
|
|
Three Important Macromolecules - Protein |
Kind of Macromolecule - Informational.
Examples - Enzymes, Hormones, and Antibodies.
Repeating Monomores - Amino Acids.
# of kinds of Repeating Units - 20 |
|
|
Three Important Macromolecules - Nucleic Acids |
Kind of Macromolecule - Informational. Examples - DNA and RNA. Repeating Monomores - Nucleotides. # of kinds of Repeating Units - 4 in DNA and 4 in RNA. |
|
|
Three Important Macromolecules - Polysaccharides |
Kind of Macromolecule - Storage: Structural
Examples - Starch, Glycogen: Cellulose
Repeating Monomores - Monosaccharides: Monosaccharides
# of kinds of Repeating Units - One or a few:One or a Few |
|
|
Macromolecule - Basic Synthesis |
1. Stepwise Polymerization 2. Condensation Reaction 3. Activated Monomer 4. Carrier Molecule 5. Energy Retirement 6. Directionality |
|
|
1. Stepwise Polymerization |
Macromolecules are always synthesized by the stepwise polymerization of similar or identical small molecules called monomers |
|
|
2. Condensation Reaction |
Addition of each monomeric units occurs with the removal of a water molecules. |
|
|
3. Activated Monomer |
Monomeric units must be in activated form before condensation to occur. |
|
|
4. Carrier Molecule |
Activation involves coupling of the activated monomer to a carrier molecule. |
|
|
5. Energy Requirement |
Energy to couple monomers to the carrier molecule is provided by ATP, Adenosine Triphosphate. |
|
|
6. Directionality |
Macromolecules have a inherent directionality. It means that the two ends of the polymers are chemically different from each other. |
|
|
Activation |
Amino acids are activated by linking to carrier molecule transfer RNA (tRNA)
Polysaccharides are synthesized from sugar molecules which are activated by derivatives of nucleotides
ADP for starch and UDP for Glycogen
Nucleic Acid Synthesis – No carrier molecule because ATP and GTP are high-energy molecules |
|
|
Protein Folding Requirements |
Information needed to specify the folding of macromolecules and their interactions to form more complicated structures with specific biological function is inherent in the polymers themselves. |
|
|
Many proteins self assemble |
Immediate product of amino acid polymerization is polypeptide.
To become proteins - bonding and folding is required to acquire a three-dimensional structure for biological activities.
Molecule Chaperones - required for protein folding. |
|
|
Denature and Renaturing |
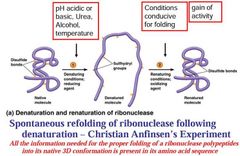
|
|
|
Self Assembly: Strict vs. Assisted |
Strict Self Assembly - no factors are required.
Assisted Self Assembly - Chaperones are required. |
|
|
Non-covalent Folding Interactions |
Hydrogen Bonds - electronegative to electropositive. Ionic Bonds - electrostatic Van der Walls Interactions - weak interactions between atoms. Hydrophobic - nonpolar groups in a macromolecule. |
|
|
Virus Self Assembly |
Complex of proteins and nucleic acids (DNA or RNA). Not functional until it meets its host. Invade and infect living cells. Utilized host's cellular machinery. Reproduce its progeny in the host cell to produce virion. |
|
|
Tobacco Mosaic Virus |
Plant Virus with rod like particle - 18 nm diameter, 300 nm length. Genome - RNA of 6395 nucleotides (6.3 kilobase). 2130 copies of single kind polypeptide. Polypeptide 158 Amino Acids - Coat Protein. |
|
|
Heinz Fraenkel-Conrat |
In vitro assembled RNA and protein components with functional virus.
Regenerated the virus particles.
First demonstration that the components on complex biological systems can reassemble into functional entities without external information.
One strain of virus RNA can be mixed with other strain of viral protein - infective. |
|
|
Tobacco Mosaic Virus (TMV) Structural Model |
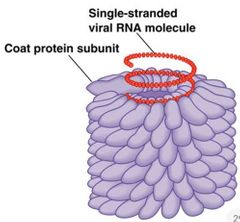
|
|
|
TMV Self Assembly |
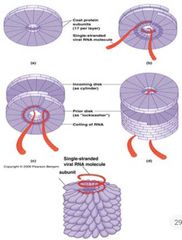
Basic unit consists of two-layered disc of coat protein Each layer has 17 identical subunit Each disc initially a cylindrical structure Conformational changes occur Tighten the helical shape to interact with 102 nts. of RNA |
|
|
Self Assembly Limitations |
Information required to specify configuration lies in the macromolecule/polymer. This allow the self-assembly systems achieve stable 3D configurations without additional information. Some systems –depends on Additional information which is supplied by a pre-existing structure The ultimate structure arises NOT by assembling the components (But by Ordering) into the matrix of an existing structure. Examples –Membranes, cell walls, chromosomes. |
|
|
Cellular Advantages to Self Assembly (Part 1) |
Chemical Specificity Efficiency of Assembly
All structure found in organisms synthesized from 30 small precursor molecules.
Polymers/macromolecule derived from different kind of condensation reactions. |
|
|
Cellular Advantages to Self Assembly (Part 2) |
Quality control issues – QC exerted at every step of the synthesis of polymers. If a defective part – discarded at early stage of the assembly. |
|
|
Self Assembly Hierarchy |
Monomers < polymers < simple molecules < complex molecules < supramolecular assembly < organelles cells < tissue < organ < body of condensation reactions |

