![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
35 Cards in this Set
- Front
- Back
|
Waves from high to low: |
radio, micro, infrared, visible, UV, x-ray, cosmic, gamma --> Goes from low to high energy, long to short wavelength |
|
|
UV: 280-380 Visible: 400-750 IR: 750-3200 Microwaves: 1m - 1000m |
length of wavelengths |
|
|
Visible acronym: |
ROYGBIV |
|
|
do electrons beyond the first feel the charge of the nucleus? which element feels the charge of the nucleus? |
NO, they feel the effects of the attraction to the nucleus --> this is less ONLY hydrogen, all others feel a mix of attraction and repulsion from the other electrons - outer electrons are also SHIELDED from attraction to nucleus by inner electrons |
|
|
outer electrons are ________ from the attraction to the nucleus by the innermost electrons an electron is never at the ________ of its orbital |
Shielded node |
|
|
________ means two orbitals are the exact same size. As electrons are added, do they remain the same? Why? The bigger the orbital, the more _______ it is |
degenerate no, the size of orbitals grows as electrons are added. This is because of the forces between them (repulsion, attraction, etc) complex |
|
|
________ is the ability of the other electrons to go into other orbitals, the higher the orbital, the _______ it is to do this for lower orbitals. This is because these electrons are more ________ |
penetration, higher, shielded |
|
|
an s electron is bound ______ tightly than a p electron |
more tightly |
|
|
__________ is the tendency for atoms in the same groups to act the same The three characteristics we are concerned with are: |
periodicity ionization energy, |
|
|
_______ is the ability to lose an electron, or the tendency to want to become a cation _______ is the ability to gain an electron, or the tendency to become an anion. _________ the ability of one atom to steal another atoms electrons |
ionization energy electron affinity electronegativity |
|
|
a low ionization energy means that it is _____ to become a cation, it likely has (3): |
easy, weak effective or nuclear charge, heavily shielded, large atomic radius |
|
|
a low electron affinity has 3: |
high nuclear charge, lightly shielded, small size |
|
|
______ is the smallest atom, as you move from left to right, atomic radius gets ________, this is because of a greater __________ Moving from top to bottom, the atomic radius gets ________, this is because it has more________ |
Hydrogen, SMALLER, greater effective nuclear charge which makes it tighter bigger, orbitals |
|
|
electronegativity has: 3: |
low ionization, negative electron affinity, ...size? It is a combo of factors that are determined by the multiple forces of electrons in an atom. |
|
|
an equation where an electron is lost could be said to represent: an equation where an electron is gained could be said to represent: Note that the delta H will = this energy |
ionization energy electron affinity |
|
|
the more electrons you try to pull out, the _________ energy required, this is because the first electron is ______ so it is easier to pull out |
greater the energy required, less shielded |
|
|
1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p, 8s s = p = d = |
2 6 10 |
|
|
______ electrons do the shielding, they are in the _____ orbitals _______ electrons are not well shielded and are used in chemical reactions |
core electrons, s orbitals valence |
|
|
Why does Aluminum lose electrons more easily than magnesium? |
It has a lower ionization energy, meaning that it is more highly shielded, and a weak nuclear charge |
|
|
*Write orbital diagrams for valence electrons, think about noble metals and whether the transition metals are in their happy, lazy states* If it's possible to make it more stable, do so. This applies to Cr family (column) and Cu family, which happens to be the rest of the noble metals |
see other side |
|
|
MEMORIZE STEPS of Born-Haber cycle and WHAT they mean: Step 1:Sublimation of sodium, Step 2: dissociation of chlorine, Step 3: ionization of sodium, Step 4: formation of chloride ion, Step 5: formation of sodium chloride from ions. |
see other side |
|
|
The _______ rule overrides the _______ rule in drawing Lewis Dot structures. However, the latter should still be used first to get the general idea while the former is used to refine. |
formal charge, octet rule |
|
|
FC (X) = |
# of valence electrons - (1/2 bonding electrons + nonbonding electrons) |
|
|
A stable element has a formal charge of: The element with ______ electronegativity will go in the center |
0 LESS |
|
|
________ are different versions of the same Lewis dot structure _____ means same elements are on same side, _____ means same elements are across from each other |
geometric isomers cis trans - these are the difference between healthy fats and unhealthy ones, between potatoes and wood |
|
|
_______ is the difference between electronegativities |
polarity - this is what makes isomers react differently from each other |
|
|
________ is when you don't change the atom arrangement, but do change the bonding |
resonance |
|
|
you have to ask, can I rearrange the atoms to make isomers? can I rearrange the bonds to make resonance? and always using our rules of electronegativity and formal charge to determine which is the best. |
see other side |
|
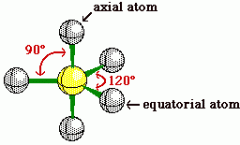
|
see other side |
|
|
Lewis, hybridization, VESPR is only for _____ bonds |
covalent |
|
|
_____________is the temperature/pressure condition where all three states exist simultaneously |
triple point |
|
|
For most substances, freezing point ________ as pressure increases. |
increases |
|
|
Every single point is the boiling point of water - YOU HAVE TO KNOW THE PRESSURE |
see other side |
|
|
_________ is when you can no longer distinguish a gas from a liquid. The pressure at this point is _______ |
critical point critical pressure |
|
|
_______ and _______ are dependent on intermolecular forces A strong force = ________ The lighter it is, the _______ the boiling point. The exception to this is ________. |
boiling point, melting point high boiling point lower, water |

