![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
36 Cards in this Set
- Front
- Back
|
Bronsted Acid |
Proton Donor |
|
|
Bronsted Base |
Proton Acceptor |
|
|
Lewis Acid |
Electron pair acceptor |
|
|
Lewis base |
electron base donor |
|
|
Autoionization |
self ionizing; means that water reacts with itself |
|
|
Kw (Autoionization constant) |
[H3O][OH]=1 x 10^-14 @ 25 degrees than means each one is 1 x 7^-14 |
|
|
pH |
-log[H+] |
|
|
Estimating pH |
if its 1 x 10^-4, pH is 4. You take the negative of the exponent. However, if its a number bigger than one, something like 5 x 10^-6, you have to take the negative of the exponent and just subtract one. |
|
|
Any dilution on the mcat of an acid or a base: |
the solution always moves toward the neutral pH of 7 and will not move more than 1 pH unit. That will be your answer |
|
|
Strong Acid |
Completely dissociates |
|
|
6 Strong acids |
HCl HI Hbr H2SO4:Sulfuric Acid HNO3:Nitric Acid HClO4: Perchloric Acid |
|
|
Strong Bases |
1.) All Group 1 oxides and hydroxides Examples: NaOH or Li2O 2.) some group 2 hydroxides (strontium, calcium, barium)
|
|
|
Weak acid |
does not completely dissociate |
|
|
Ka |
[H+][A-] / HA basic means more acidic |
|
|
Kb |
[OH-][HB] / B bigger means more basic |
|
|
(Ka x Kb)= |
10 ^-14 For acid base pairs (conjugate pairs) |
|
|
pKa + pKb= |
14 for acid base pairs (conjugate pairs) |
|
|
Conjugate pair |
The acid and its conjugate base after removal of a proton |
|
|
Conjugate Rules |
1.) The conjugate of any strong acid or base is pH neutral; the conjugate base of a strong acid has no basic properties; vice versa 2.) The conjugate of any WEAK acid or base is a weak base or acid; the stronger the weak acid or base, the weaker its conjugate |
|
|
For a strong acid or base, what is the H+ or OH- concentration always equal to? |
the solution concentration; pH can easily be found from the concentration |
|
|
Quick way to find pH of solution if they give concentration of a weak acid or base solution |
Ka= x^2 / [Initial concentration] value of x will be H+ concentration which you can use to find pH |
|
|
Neutralization Reaction |
When acid and base react, they form a salt and water every time. (strength of either acid or base does not matter) Reaction is always exothermic and always goes to completion when equal molar amounts of base and acid are used Ex: HCl + KOH --> KCl + H2O |
|
|
Equation for Neutralization reaction |
(a)(Volume of A)[concentration of A]=(b)(volume of B)[Concentration of B] a= number of acid protons that can be donated b= number of protons that can be accepted |
|
|
pH after neutralization reaction |
pH of the resulting solution is determined by the type of salt formed. If the anion of the salt is inactive in water, the solution is pH neutral and thus it is the conjugate base of a strong acid. If the cation is group 1 or 2 cation it will have no effect on pH If the cation is more acidic than H2O, then it is the conjugate acid of a weak base and the pH will be acidic. If the anion is more basic than water, then it is the conjugate base of a weak acid. |
|
|
Buffer Solution |
Resist a change in pH when a small amount of acid or base is added To make a buffer, combine a weak acid or base and its conjugate in roughly equal amounts |
|
|
Henderson Hasselback equation (shortcut to find pH of buffer solution) |
pH= pKa + log [A-]/[HA]
poH= pKb + log [HB+]/[B] |
|
|
How to pick a buffer |
Always pick a buffer with a pKa close to the pH that you are trying to maintain However, if a buffer solution is diluted, the pH is unchanged |
|
|
Indicator |
weak acid or base that changes color when it changes into its conjugate |
|
|
Always pick an indicator with a pKa close to the pH you want to indicate |
YUH |
|
|
Titration |
used to determine an unknown acid or base by determining its pKa or pKb |
|
|
Procedure for titration |
Add a titrant (strong acid or base of known concentration) to the unknown in a dropwise fashion Record the pH changes and plot the results Graph looks like a muthfuckin S nigga |
|
|
Titration Curve |
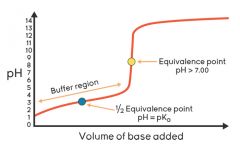
|
|
|
Equivalence point |
Concentration of [H+] is equal to concentration of [OH-] all acid is neutralized |
|
|
Half Equivalence point |
Half of acid is neutralized; [HA] = [A-] pH = pKa of unknown Uknown is found using this point |
|
|
What determines the pH of the equivalence point? |
the salt formed |
|
|
Buffering domain |
Part of titration curve where pH changes very slowly |

