![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
44 Cards in this Set
- Front
- Back
|
What did Lavoiser do in the 1790's?
|
Composed the first list of the 23 elements known since prehistoric times (gold, carbon, silver, etc.)
|
|
|
What occurred during the Industrial Revolution (1800s)
|
Many new elements were isolated (compounds were broken down)
|
|
|
In 1860, chemists created a _________ for determining the atomic mass of all elements.
|
Standard
|
|
|
What did John Newlands discover in 1864?
|
Law of Octaves
|
|
|
Law of Octaves
|
When elements are arranged by atomic mass, their properties repeat every eighth element
|
|
|
Why was the Law of Octaves ground breaking and what was the problem with it?
|
-Showed first periodic relationship in elements
-Problem was that not all known elements followed this pattern |
|
|
What did both Meyer and Mendeleev discover in 1869?
|
Both noticed a connection between atomic mass and properties of elements. They arranged elements in order of increasing atomic mass and put elements with similar properties in the same column.
|
|
|
Why did Mendeleev receive more credit than Meyer?
|
Because he published his findings first and left blanks in his table where he knew undiscovered elements would fit such as scandium, gallium, and germanium
|
|
|
What happened to Mendeleev's table when more elements were found?
|
Several elements did not fit anywhere
|
|
|
How did Henry Moseley arrange the elements and what was the result?
|
By atomic number. In result, the misplaced elements fell into place and a clear periodic pattern of elemental properties was found (Periodic Law)
|
|
|
Periodic Law
|
When elements are arranged by increasing atomic number, there is a repitition of chemical and physical properties within a family
|
|
|
Representative elements
|
Groups designated with an A, possess a wide range of chemical and physical properties
|
|
|
Transition elements
|
Groups designated with a B
|
|
|
What are the three main classifications of the elements?
|
1. metals
2. nonmetals 3. metalloids |
|
|
What are the properties of metals?
|
-shiny when smooth and clean
-solid at room temperature -good conductors of heat and electricity -malleable, ductile -loose electrons to form s2p6 (cations) |
|
|
Alkali metals
|
Group 1A elements
|
|
|
Alkaline earth metals
|
Group 2A elements
|
|
|
What are the Group B elements (transition elements) divided into?
|
1. transition metals
2. inner transition metals |
|
|
What two sets of inner transition metals are located at the bottom of the periodic table?
|
Lanthanides and actinides
|
|
|
What are the properties of nonmetals?
|
-usually gases or brittle, dull-looking solids
-poor conductors of heat and electricity -gain electrons to achieve s2p6 |
|
|
Halogens
|
-Highly reactive group 7A nonmetals
-Valence configuration s2p5 -Need 1 electron to achieve s2p6 |
|
|
Noble gases
|
-Extremely unreactive group 8A nonmetals
-Valence configuration of s2p6 |
|
|
Metalloids
|
Elements with physical and chemical properties of both metals and nonmetals
|
|
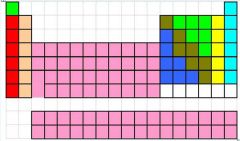
Label the periodic table
|
-Red group: Alkali metals
-Tan group: Alkaline earth metals -Pink elements: Transition metals, "B Elements," D Block -Bottom pink elements: Inner transition metals, "F Block," lanthanides(top) and actinides(bottom) -Yellow group: Halogens -Turquoise group: Noble gases |
|
|
Why do elements in the same group have similar chemical properties?
|
Because their atoms have the same number of valence electrons
|
|
|
All of the elements in group 1A all have a valence configuration of ___.
|
s1
|
|
|
List the metalloids?
|
Boron
Silicon Germanium Arsenic Antimony Tellurium Polonium Astatine |
|
|
Describe the properties of Alkali Metals (Group 1A)
|
-valence configuration s1
-all silver, soft -react explosively with H2O to form alkaline solution |
|
|
Describe the properties of Alkaline Earth Metals
|
-valence configuration s2
-all found naturally in Earth -react with H2O to form alkaline solution |
|
|
What are lanthanides used for?
|
CRT's and TV's
|
|
|
What forms salts?
|
Alkali/Alkaline Earth Metals + Halogens
|
|
|
What makes up the S-Block?
|
Groups 1A and 2A
|
|
|
What makes up the P-Block?
|
Groups 3A through 8A
|
|
|
What makes up the D-Block?
|
Transition Metals
|
|
|
What makes up the F-Block?
|
Inner Transition Metals
|
|
|
What is the atomic radius for metals?
|
Half the distance between adjacent nuclei (half the distance across atom)
|
|
|
What is the atomic radius for nonmetals?
|
Half the distance between nuclei of identical atoms that are chemically bonded together (half the distance across bonded atom)
|
|
|
What pattern is seen in atomic size as you move from left to right across a period? Why?
|
Decrease in atomic radius. Because electron shielding increases, positive nuclear charge pulls outermost electrons closer to nucleus.
|
|
|
What pattern is seen in atomic size as move down a group? Why?
|
Increase in atomic radius. Because the atom becomes bigger due to more energy levels. This creates more electrons and these electrons shield outer electrons from pull of nucleus.
|
|
|
Ion
|
An atom or group of atoms that has a positive or negative charge due to the gaining or losing of electrons
|
|
|
When atoms lose electrons and form positively charged ions, what happens?
|
Atomic radius becomes smaller
|
|
|
Why do atoms become smaller when they lose electrons and form positively charged ions?
|
1. Valence electron leaves, leaving outer orbital empty, causes smaller radius
2. Less electrons so shielding decreases, outer electrons are pulled closer to nucleus, causes smaller radius |
|
|
When atoms gain electrons and form negatively charged ions, what happens?
|
Atomic radius becomes larger
|
|
|
Why do atoms become larger when they gain electrons and form positively charged ions?
|
Because this increases the electrostatic repulsion between the atoms outer electrons, forcing them to move farther apart, causes larger radius
|

