![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
14 Cards in this Set
- Front
- Back
|
Elememt |
A substance that only contains 1 sort of atom |
|
|
Compound |
Atoms of 2 or more elements, that are chemically bonded in fixed proportions |
|
|
Reactants |
The substances that react (left side of equation) |
|
|
Atom |
The smallest part of an element that can exist |
|
|
Products |
The new substances that are formed (right side of equation) |
|
|
Mixtures |
2 or more elements or compounds that aren't chemically combined |
|
|
Filtration |
Separates soluble solids or liquids from insoluble solids |
|
|
Crystalisation |
Used to obtain a soluble solid from a solution. 1) the mixture is gently warmed 2) the water evaporates leaving crystals of pure salt |
|
|
Simple distillation |
Used to obtain a solvent from a solution |
|
|
Fractional distillation |
Used to obtain separate mixtured where the components have different boiling points |
|
|
Chromatography |
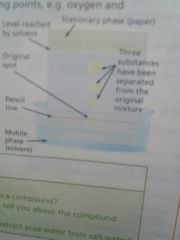
Used to separate the different soluble,coloured component of a mixture |
|
|
Subatomic particlez |
Electron- mass (negligible) charge (-1) orbits around the nucleus in energy shells; protons- mass (1) charge (+1) in nucleus of atom (atomic number); neutrons- mass (1) charge (0) in nucleus of atom (mass number- atomic number) |
|
|
Isotope |
A form of an element with the same amount of protons but different number of neutrons |
|
|
Ion |
Atoms that have lost or gained electrons and gained a charge due to the imbalance of protons and electrons that are usually equal |

