![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
15 Cards in this Set
- Front
- Back
|
What is enthalpy? |
The measure of heat in a system |
|
|
System -> surroundings |
Exothermic |
|
|
Surroundings -> system |
Endothermic |
|
|
Negative enthalpy change |
•exothermic •surroundings get hotter •arrow must point down •reactants have more energy than products •system loses energy |
|
|
Positive enthalpy change |
•endothermic •surroundings get colder •arrow must point up •system gains energy •products have more energy than the reactants |
|
|
Exothermic |
Energy released when making bonds is greater than the energy required when breaking bonds |
|
|
Endothermic |
Energy required when breaking bonds is greater than energy released when making bonds |
|
|
Standard conditions |
100kPa 298K (25°C) |
|
|
Enthalpy of combustion |
The enthalpy change when one mole of a compound is burned completely in excess oxygen under standard conditions |
|
|
Enthalpy of formation |
The enthalpy change when one mole of a compound is formed from its elements under standard conditions |
|
|
Enthalpy of neutralisation |
The enthalpy change for the reaction of acid with base to produce 1 mole of water under standard conditions |
|
|
Direct method of measuring enthalpy change |
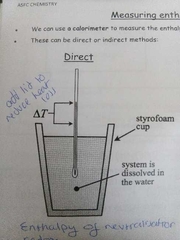
|
|
|
Indirect method of measuring enthalpy change |

|
|
|
Q(J) = |
m c ∆T m c ∆Tm = mass of water(indirect)/aq solution in g(direct) c = 4.18 JK-¹g-¹ |
|
|
∆H (JK-¹g-¹) = |
-Q / n (of one reactant) |

