![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
23 Cards in this Set
- Front
- Back
|
Molecular formula of water |
H2O |
|
|
Structural formula of water |
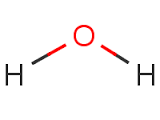
|
|
|
Why are water molecules are polar |
H 2.2 O 3.4 there is more than .5 difference in electromagnetic charge |
|
|
Hydrogen bond between water molecules |
the attraction between oppositely charged regions of polar molecules |
|
|
Cohesion |
hold together |
|
|
Adhesion |
Attraction to charged surfaces |
|
|
Hydrophilic |
Attraction to water - Charged substances dissolve in water |
|
|
Hydrophobic |
Repelled by water - noncharged won't dissolve in water |
|
|
Liquid water is necessary for life on earth |
because 75% of living things are water and water is necessary for cell processes as well as regulate temperature and environment |
|
|
Water's high heat capacity is important to living things as a temperature buffer |
It helps regulate environmental temperature and since all living things are made of mostly water they are better able to resist change of their temperature |
|
|
Water's high heat vaporization is important to living things |
change from liquid to gas requires significant energy so evaporation from surface takes away heat and helps stabilize temperature |
|
|
Why does ice float |
Water is one of the few substances that becomes less dense in solid form. If ice sank then eventually ponds lakes and ocean would freeze solid |
|
|
Water is the basis of scientific measurement |
calorie (cal) + amount of heat it takes to raise the temperature of 1 gram of water by 1 degree. 1g= 1mL at sea level. It is based on water |
|
|
Aqueous solution |
solute dissolved in water |
|
|
Molarity (M) |
Moles of solute per liter solution 1M solution= 1 mole (6x10 ^23) of solute in 1L of solition |
|
|
Formula for concentration using Molarity |
(Molecular weight)(L)(M)=________g Divided by 1000 for mL |
|
|
Percentage % |
Grams of solid solute per mL of a solution 100% solution = 1g of solute in 1mL of solution or = 1mL of solute in 1mL of solution |
|
|
Formula concentration using Percentage |
(percent in desimal)(mL)=_________g/mL multiply by 1000 if L |
|
|
pH |
is the hydrogen concentration in an aqueous solution. |
|
|
pH Scale |
The higher the hydrogen concentration the lower the number on the pH scale. |
|
|
Acidic |
0-6.99 |
|
|
Basic |
7.01-14 |
|
|
Buffer |
A chemical substance that maintains the pH of a solution. By either donating a hydrogen or accepting a hydrogen from the solution |

