![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
39 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
Anything that has mass and takes up space. |
Matter |
|
|
|
can not be separated on broken down into simpler substances |
An element |
Ex:carbon |
|
|
The smallest unit of an element that maintains the chemical properties of that element. |
Atom |
|
|
|
Truth or false elements can break down into simpler substances? |
False,elements can not get broken down into simpler substances of chemical means |
|
|
|
The elements is Represented by one capital letter or a capital letter with a lowercase letters on__________ |
Periodic table |
Ex:C or Ai |
|
|
A group of Atoms that are held together by chemical forces that is a small unit of matter |
A Molecules |
Ex: Neons, hydrogen |
|
|
a substance made up of Atoms of two or more different elements joined by chemical bonds |
Compound |
|
|
|
Something we are familiar with auction and hydrogen that makes water |
The elements that form a compound |
|
|

Is water a compound true or false |
True, it’s a compound because it has two hydrogen atoms and one oxygen atom |
|
|
|
A compound is used by? |
Chemical formula |
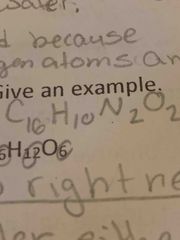
|
|
|
What Are the elements number called right next and below on the atoms |
Subscript |
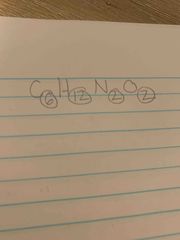
|
|
|
A sample of matter either a single element or a single compound that has definite and physical properties |
Pure substance |
|
|
|
2types of pure substances |
Elements and atoms |
|
|
|
The combination of two or more social says that are not chemically combined |
A mixture |
|
|
|
Chemical combination of substances |
A compound |
|
|
|
Not evenly distributed mixture |
A heterogeneous |
|
|
|
Evenly distributed mixture |
Homogeneous mixture |
|
|
|
True or false can gases mix with liquids |
True, gases can mix with liquids. For example water |
|
|
|
Texture is example of _____of matter |
Physical property |
|
|
|
Shape, color, odor, and texture |
Are 3 examples of physical properties |
|
|
|
The temperature and pressure of a solid that becomes a liquid. |
Melting point |
|
|
|
With solid melts it is now a liquid. When the liquid is in the pot it’s boiling. |
boiling points |
|
|
|
Strength, hardness and magnetis |
Are 3 physical properties that can be measured |
|
|
|
The ratio of the mass of a substance to the volume of this substance |
Density |
|
|
|
Is dissolving a physical or chemical change? |
Dissolving can be physical and chemical changes.Because it can takechemicals from a object and take the shape of a object. |
|
|
|
Chemical changes form new ______ |
Substances |
|
|
|
Bread paint gases of burnt food foaming/fizzing. |
These 4 things are something that you can observe |
|
|
|
True or false. Can a chemical change be reversed by physical means |
FALSE: Chemical change cannot be reversed by physical change. |
|
|
|
How are mixtures separated |
You separate a mixture physically |
|
|
|
How are compounds separated |
You separate compounds chemically |
|
|
|
D= m/v —> density= mass: volume |
Equation used to calculate density |
|
|
|
10.0 g for every 10.0 cm cubes |
Is the density of water |
|
|
|
Solve this equation |
16.52 g divided by 2.26 cm cubes equal _____ density |
7.3 g/centimeters cubes=D |
|
|
Solve this problem 16.2g/2.26 |
7.32g/cm cubes=D |
|
|
|
163g divided by 50.0cm cubes |
D=3.26 g/cm |
|
|
|
Last one to solve: 11.3g/cm= 6.7cm |
75.71 |
Multiply |
|
|
Why do objects float in water? |
Some objects float in water because of the weight |
|
|
|
You can reserve ________ properties in a microscope |
Chemical properties |
|
|
|
A change of matter form one to another without changing the chemicals is? |
Physical change |
|

