![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
52 Cards in this Set
- Front
- Back
|
nociceptive pain
|
Stimulation of peripheral nerve fibers;
Alcohol in a cut, cold, burn, paper cut, hammer to thumb vice: neuropathic pain which is burning, tingling, pins and needles... Herpetic neuralgia and phantom limb are neuropathic |
|
|
how do opioids work?
|
Produces analgesia through actions at G protein-coupled receptors in the CNS and spinal cord regions involved in the transmission and modulation of pain
|
|
|
opioid MOA
|
Close voltage-gated Ca2+ channels on presynapticerve terminals →reducing transmitter release
--Glutamate (principle EAA released from nociceptive nerve terminals), acetylcholine, norepinephrine, serotonin, substance P Hyperpolarize (and inhibit) post-synaptic neurons by opening K+ channels |
|
|
Potential receptor mechanisms of analgesic drugs
|
Primary afferent neuron originates in the periphery and carries pain signals to the dorsal horn of the spinal cord, where it synapses via glutamate and neuropeptide transmitters with the secondary neuron. Pain stimuli can be attenuated in the periphery (under inflammatory conditions) by opioids acting at μ-opioid receptors (MOR) or blocked in the afferent axon by local anesthetics. Action potentials reaching the dorsal horn can be attenuated at the presynaptic ending by opioids and by calcium blockers (ziconotide), α2 agonists, and possibly, by drugs that increase synaptic concentrations of norepinephrine by blocking reuptake (tapentadol). Opioids also inhibit the postsynaptic neuron, as do certain neuropeptide antagonists acting at tachykinin (NK1) and other neuropeptide receptors.
|
|
|
opioid chemistry
|
Semi-synthetic alkaloids derived from opium and synthetic surrogates
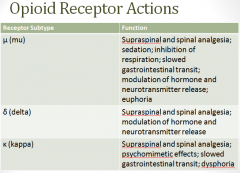
Opioids can be: Full agonists, partial agonists, or antagonists Opioids have actions at: µ (mu) -, κ (kappa)-, or δ (delta)-receptors Opioids can be agonists at one receptor AND antagonist at another EX: Nalbuphine (κ-agonist and µ-antagonist) Opioids can be modified in the liver to more ACTIVE compounds |
|
|
Endogenous Opioid Peptides
|
Derived from precursor proteins that yield several other proteins:
-Prepro-opiomelanocortin (POMC) -Preproenkephalin (proenkephalin A) -Preprodynorphin (proenkephalin B) |
|
|
the three families of endogenous opioid peptides
|
Endorphins
Enkephalins (Met(hionine)-enkephalin and Leu(cine)-enkephalin) Dynorphins |
|
|
Transmission
|
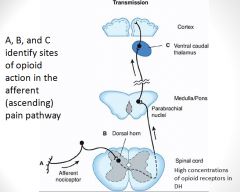
Putative sites of action of opioid analgesics. Sites of action on the afferent pain transmission pathway from the periphery to the higher centers are shown. A: Direct action of opioids on inflamed or damaged peripheral tissues. B: Inhibition also occurs in the spinal cord. C: Possible sites of action in the thalamus.
|
|
|
Opioid receptors
|
Opioid receptors are located on:
Spinal cord pain transmission neurons Primary afferents that relay message to spinal cord Receptor subtypes can dimerize in presynaptic terminals Heterodimerization of µ and δ contribute to µ-agonist efficacy |
|
|
What is the action of opioid agonists in the periphery?
|
Opioid agonists in the periphery:
-Inhibit the release of excitatory transmitters from primary afferents |
|
|
What is the action of opioid agonists in the lower CNS?
|
Opioid agonists in the lower CNS:
-Directly inhibit dorsal horn pain transmission neurons (part of the ascending pain pathway) -Exert power analgesic effect directly on spinal cord --Direct application of agonists to spinal cord provides regional analgesia without supraspinal adverse drug reactions (respiratory depression, N/V, sedation) from systemic drug administration |
|
|
Systemic distribution of opioids
|
Opioids in the (higher) CNS:
-Descending (modulatory) pain pathways --Opioids directly inhibit neurons (just like before) in similar regions --But these actions activate inhibitory neurons that send signals to the spinal cord and inhibit pain transmission neurons ---Overall effect is an INCREASE of analgesic effect |
|
|
Systemic distribution of exogenous opioids
|
Involves the release of endogenous peptides
-EX: Morphine may act directly (and primarily) through µ-receptors, but this action can release endogenous peptides that primarily target δ and κ receptors --Pain modulation/analgesia is extremely complex!!! |
|
|
Descending Pain Pathway
|
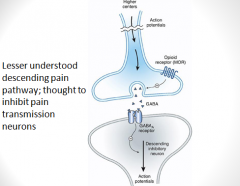
Brainstem local circuitry underlying the modulating effect of μ-opioid receptor (MOR)–mediated analgesia on descending pathways. The pain-inhibitory neuron is indirectly activated by opioids (exogenous or endogenous), which inhibit an inhibitory (GABAergic) interneuron. This results in enhanced inhibition of nociceptive processing in the dorsal horn of the spinal cord
|
|
|
Descending Inhibitory Pathway
|
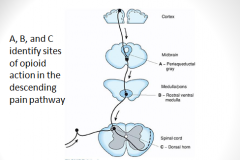
Opioid analgesic action on the descending inhibitory pathway. Sites of action of opioids on pain-modulating neurons in the midbrain and medulla including the midbrain periaqueductal gray area (A), rostral ventral medulla (B), and the locus caeruleus indirectly control pain transmission pathways by enhancing descending inhibition to the dorsal horn (C).
|
|
|
Frequent, repeated doses of a drug produce a gradual loss of effectiveness. This defines________
|
tolerance
|
|
|
When a drug is stopped or an antagonist is administered and the person experiences a characteristic withdrawal syndrome. This defines_______.
|
dependence; have to have to withdrawal
|
|
|
Tolerance and Dependence
|
Exact mechanisms are poorly understood.
-However, persistent activation of µ-receptors plays a primary role in induction and maintenance |
|
|
possible hypotheses of tolerance and dependence (4)
|
1. upregulation – associated with tolerance, but not the only explanation
2. NMDA: NMDA receptor antagonists can block tolerance -Persistent administration of opioids INCREASES the sensation of pain 3. Receptor recycling: Normally: ligand binds to GPCR, induces signaling and then receptor is endocytosed, dephosphorylated (re-sensitized) and returned to plasma membrane -NOT with µ-opioid receptors morphine fails to induce endocytosis 4. Receptor uncoupling: Due to a dysfunctional interaction between G-proteins, second messenger systems and target ion channels – probably linked to recycling |
|
|
Degrees of Tolerance
|
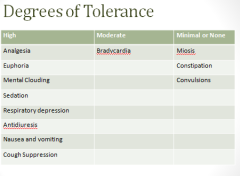
A lot of drugs dliate pupils, opioids shrink the pupils
|
|
|
PK- absorption
|
Subcutaneous, IM, Oral
Undergo intense first –pass effect (patient dependent) -Makes predicting an effective oral dose difficult -Exceptions: codeine and oxycodone Nasal insufflation results in rapid therapeutic blood levels Lozenges provide administration via oral mucosa Transdermal patches -Iontophoretic transdermal system (ITS) --Needle-free patient-controlled anesthesia |
|
|
PK: distribution and elimination
|
Localize rapidly to highly perfused tissue sites
-Brain, kidneys, lungs, liver, and spleen Skeletal muscle is main reservoir of drug -Despite low overall concentrations Highly lipophilic drugs (EX: fentanyl) can accumulate in fatty tissue Drugs are excreted mainly through the urine |
|
|
PK: metabolism
|
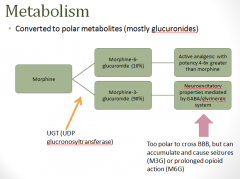
|
|
|
PK: more metabolism
|
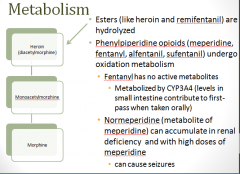
|
|
|
PK: more more metabolism
|
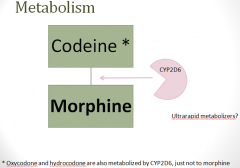
|
|
|
CNS effects of opioids
|
Analgesia – both sensory and affective
Euphoria – pleasant, floating sensation with lessened anxiety and distress -Dysphoria – unpleasant, characterized by restlessness and malaise Sedation –common; little to no amnesia; sleep induced in elderly; very deep sleep (when combined with sedatives); disrupts REM and non-REM eye movements at normal doses Respiratory depression – dose-related; decreased respiratory function (rate, volume, and exchange) Cough suppression – suppresses cough reflex |
|
|
more CNS effects of opioids
|
Miosis – pupillary constriction
Truncal rigidity – increased tone in large muscle groups after IV exposure to highly lipid-soluble opioids (interferes with ventilation) -Treat with opioid antagonist or neuromuscular blocking agent? Nausea and vomiting – activate the brainstem chemoreceptor trigger zone; ambulation increases N/V (vestibular component) Temperature – homeostatic regulation is mediated (in part) by endogenous opioids -µ-agonists administered directly to the hypothalamus produce hyperthermia -κ – agonists produce hypothermia |
|
|
Peripheral effects of opioids
|

|
|
|
more Peripheral effects of opioids
|

|
|
|
Clinical Uses of opioids
|

|
|
|
Clinical uses - Anesthesia
|

|
|
|
Routes of Administration
|

|
|
|
Specific Agents
|
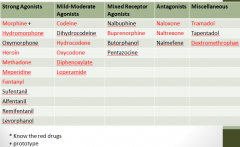
|
|
|
methadone
|

|
|
|
Fentanyl and Meperidine
|

|
|
|
Cholinergic/Anti-cholinergic
|

|
|
|
Codeine
|

|
|
|
Hydrocodone and Oxycodone
|

|
|
|
Diphenoxylate and Loperamide
|
Used in the treatment of diarrhea
Used in combination with atropine to further discourage abuse potential Diphenoxylate is very unlikely to be abused (Schedule V) Loperamide’s acts on peripheral µ-receptors -Available without a prescription |
|
|
Naloxone and Naltrexone
|

|
|
|
Naloxone
|

|
|
|
Buprenorphine
|

|
|
|
Tramadol
|

|
|
|
Dextromethorphan
|

|
|
|
Adverse Drug Reactions
|

|
|
|
Tolerance, Dependence, Addiction
|

|
|
|
Tolerance
|

|
|
|
Dependence
|

|
|
|
Withdrawal (Abstinence) Syndrome
|

|
|
|
Addiction
|

|
|
|
Contraindications/Cautions
|

|
|
|
Key Drug Interactions
|
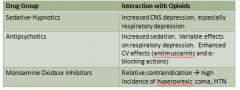
|

