![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
64 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
show the initiation, propagation, and termination steps for the monochlorination of cyclohexane.
|
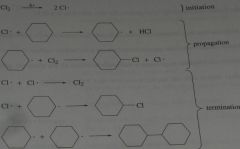
|
11.1
|
|
|
Write the mechanism for the formation of tetrachloromethane, CCl_4. From the reaction of methane with Cl_2 + hv.
|
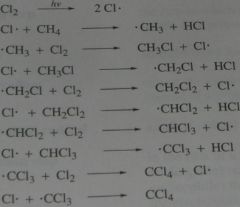
.
|
11.2
|
|
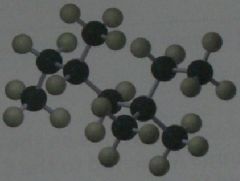
Which of the hydrogens in the following structure is the easiest for a chlorine radical to remove? How many secondary hydrogens does the structure have?
|
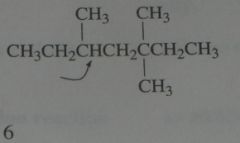
.
|
11.3
|
|
|
.
|
.
|
11.4
|
|
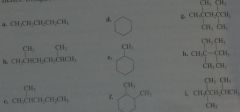
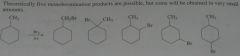
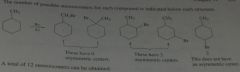
How many alkyl chlorides can be obtained from monochlorination of the following alkanes? Disregard stereoisomers.
|

|
11.5
|
|
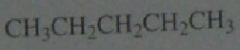
Calculate the percent yield of each product obtained if chlorination is carried out in the presence of light at room temperature.
|
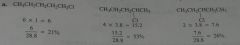
.
|
11.6.a
|
|
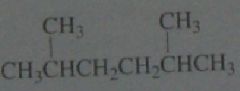
Calculate the percent yield of each product obtained if chlorination is carried out in the presence of light at room temperature.
|
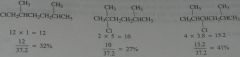
|
11.6.b
|
|
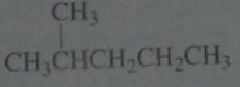
Calculate the percent yield of each product obtained if chlorination is carried out in the presence of light at room temperature.
|
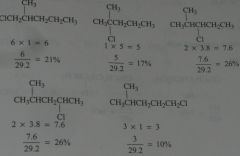
|
11.6.c
|
|

|

|
11.7
|
|
|
Carry out the calculations that predict 2-bromobutane will be obtained in 98% yield.
|
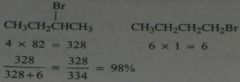
.
|
11.8.a
|
|
|
Carry out the calculations that predict 2-bromo-2,5,5,trimethylhexane will be obtained in 82% yield.
|

.
|
11.8.b
|
|
|
would chlorination or bromination produce a greater yield of 1-halo-2,3-dimethylbutane?
|
Chlorination, because the halogen is substituting for a primary hydrogen
|
11.9.a
|
|
|
would chlorination or bromination produce a greater yield of 2-halo-2,3-dimehtylbutane?
|
Bromination, because the halogen is substitution for a tertiary hydrogen
|
11.9.b
|
|
|
would chlorination or bromination produce a greater yield of 1-halo-2,2-dimethylpropane?
|
Because the molecule has only one kind of hydrogen, only one monohalogenated product will be obtained by both bromination and chlorination.
|
11.9.c
|
|
|
How could betanone be prepared from butane?
|
.
|
11.10
|
|
|
If 2-methylpropane is brominated at 125 C in the presence of light, what percent of the product will be 2-bromo-2-methylpropane? Compare your answer with 36% for chlorination of 2-methylpropane.
|

.
|
11.11
|
|
|
Calculate what percentages of monobromination products would be.
|
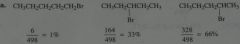
.
|
11.12.a
|
|
|
Calculate what percentages of monobromination products would be.
|
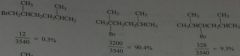
.
|
11.12.b
|
|
|
Calculate what percentages of monobromination products would be.
|

.
|
11.12.c
|
|
|
Write out the propagation steps for the addition of HBr to 1-methylcyclohexene in the presence of a peroxide.
|
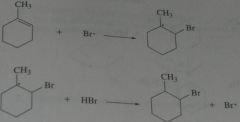
.
|
11.13
|
|
|
product of 2-methyl-2-butene with HBr
|

.
|
11.14.a
|
|
|
product of 2-methyl-2-butene with HCl
|
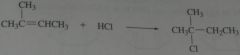
.
|
11.14.b
|
|
|
product of 2-methyl-2-butene with HBr and peroxide
|

.
|
11.14.c
|
|
|
product of 2-methyl-2-butene with HCl and peroxide
|

.
|
11.14.d
|
|
|
What hydrocarbon with molecular formula C_4H_10 forms only two monochlorinated products? Both products are achiral
|

.
|
11.15.a
|
|
|
What hydrocarbon with molecular formula C_4H_10 forms only three monochlorinated products? one is achiral.
|
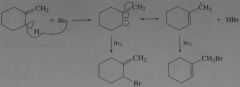
.
|
11.16
|
|
|
Two products are formed when methylenecyclohexane reacts with NBS. Explain how each is formed.
|
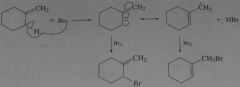
.
|
11.16
|
|
|
How many allylic substituted bromoalkenes are formed from the reaction of 2-pentene with NBS? Disregard stereoisomers
|

.
|
11.17
|
|
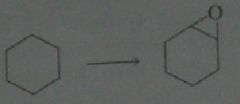
synthesis
|

.
|
11.19.a
|
|
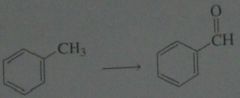
synthesis
|

.
|
11.19.b
|
|
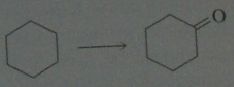
synthesis
|

.
|
11.19.c
|
|
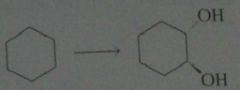
synthesis
|

.
|
11.19.d
|
|
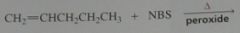
Give the product, disregard stereoisomers
|

.
|
10.e.20.a
|
|
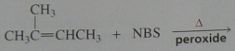
Give the product, disregard stereoisomers
|

.
|
10.e.20.b
|
|
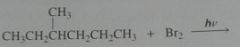
Give the product, disregard stereoisomers
|
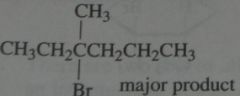
.
|
10.e.20.c
|
|
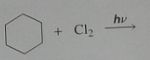
Give the product, disregard stereoisomers
|
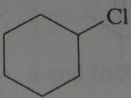
.
|
10.e.20.d
|
|
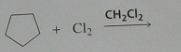
Give the product, disregard stereoisomers
|

.
|
10.e.20.e
|
|
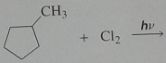
Give the product, disregard stereoisomers
|
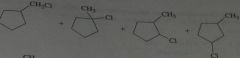
.
|
10.e.20.f
|
|
|
Identify an alkane with molecular formula C_5H_12 that forms only one monochlorinated product when eared with Cl_2
|
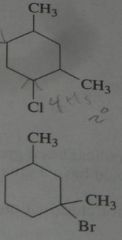
.
|
10.e.21.a
|
|
|
Identify an alkane with molecular formula C_7H_12 that forms only seven monochlorinated product when eared with Cl_2
|
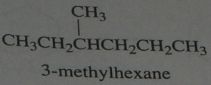
.
|
10.e.21.b
|
|
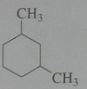
Give the major product when treated with excess Cl_2 in light. disregard stereoisomers. Br_2 @ 125 degrees?
|

.
|
10.e.22-23.a
|
|
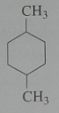
Give the major product when treated with excess Cl_2 in light. disregard stereoisomers. Br_2 @ 125 degrees?
|
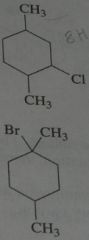
.
|
10.e.22-23.b
|
|
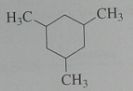
Give the major product when treated with excess Cl_2 in light. disregard stereoisomers. Br_2 @ 125 degrees?
|
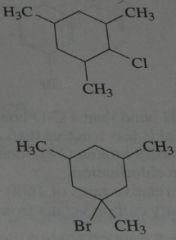
.
|
10.e.22-23.c
|
|
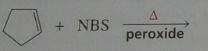
Major product. disregard stereoisomers.
|
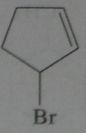
.
|
10.e.24.a
|
|
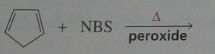
Major product. disregard stereoisomers.
|
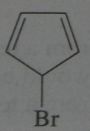
.
|
10.e.24.b
|
|
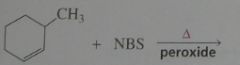
Major product. disregard stereoisomers.
|
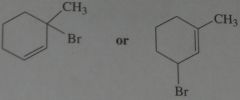
.
|
10.e.24.c
|
|
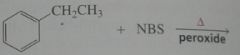
Major product. disregard stereoisomers.
|
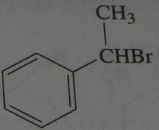
.
|
10.e.24.d
|
|
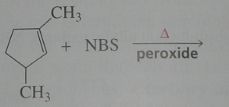
Major product. disregard stereoisomers.
|
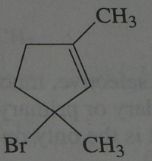
.
|
10.e.24.e
|
|
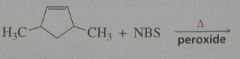
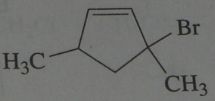
Major product. disregard stereoisomers.
|
.
|
10.e.24.f
|
|
|
Iodine does not react with ethane, even though I_2 is more easily cleaved homolytically than the other halogens. explain
|
Abstraction of a hydrogen atom from ethane by an iodine radical is a highly endothermic reaction (DEL-H = 101-71=30kcal/mol; see Table 3.2), so the iodine radicals will reform I_2 rather than abstract a hydrogen atom.
|
10.e.25
|
|
|
Propose a mechanism to account for the products formed in the following reaction:
|
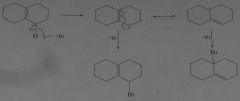
.
|
10.e.26
|
|
|
The deuterium kinetic isotope effect for chlorination of an alkane is defined in the following equation. Predict whether chlorination or bromination would have a greater deuterium kinetic isotope effect.
|
.
|
10.e.27
|
|
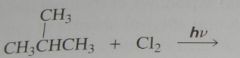
major product. Disregard stereoisomers.
|
.
|
10.e.29.a
|
|
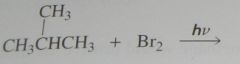
major product. Disregard stereoisomers.
|

.
|
10.e.29.b
|
|
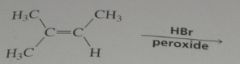
major product. Disregard stereoisomers.
|
.
|
10.e.29.c
|
|
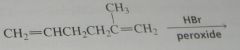
major product. Disregard stereoisomers.
|
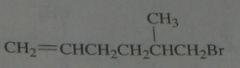
.
|
10.e.29.d
|
|
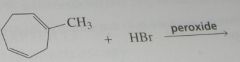
major product. Disregard stereoisomers.
|
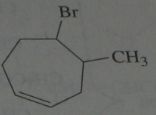
.
|
10.e.29.e
|
|
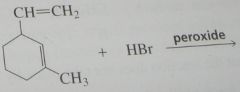
major product. Disregard stereoisomers.
|
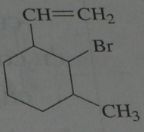
.
|
10.e.29.f
|
|
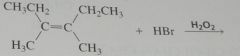
What stereoisomers would be obtained from the following reaction?
|
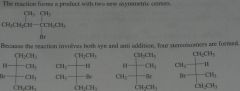
.
|
10.e.31
|
|

synthesis
|

.
|
10.e.32.a
|
|
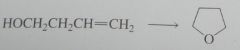
synthesis
|

.
|
10.e.32.b
|
|

synthesis
|
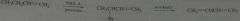
.
|
10.e.32.c
|
|
|
synthesis
|

.
|
10.e.32.d
|
|
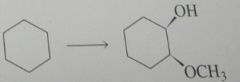
synthesis
|
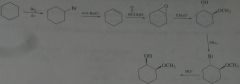
.
|
10.e.32.e
|

