![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
53 Cards in this Set
- Front
- Back
|
What is an isomer |
Same molecular formula with different structures (either atomic connectivity or spatial orientation) |
|
|
What are structural isomers |
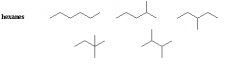
Constitutional isomers Only have same molecular formula but different connectivity |
|
|
What are geometric isomers |
Cis-trans isomers |
|
|
What is cis? |
Substituents on same side of double bond |
|
|
What are trans |
Substituents are on opposite side of double bond |
|
|
How do you determine the highest priority? |
1) Higher atomic weight for the first atom 2) Then go to the next atom --> 2nd atom with the higher atomic weight 3) Double bond takes priority over single bond but higher atomic weight takes priority over double bond |
|
|
Is "cis" Z or E? |
Z |
|
|
Is "trans" Z or E? |
E |
|
|
What does chiral mean? |
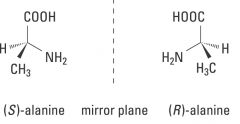
Molecule not superimposible on mirror image |
|
|
What does achiral mean? |

Superimposible mirror image |
|
|
How can carbon atoms be chiral |
If they have four different substituents (asymmetrical) |
|
|
On fischer projections is the horizontal lines wedges or dashes? |
Wedges |
|
|
On fischer projections are the vertical lines wedges or dashes? |
Dashes |
|
|
What does configuration mean |
Spatial arrangment of atoms or funcitonal group of a stereoisomer |
|
|
What is relative configuration |
Configuration in relation to another chiral molecule |
|
|
What is absolute configuration |
Spatial arrangement of atoms or groups within molecules relative to each other (R/S) |
|
|
Is S going clockwise or counterclockwise |
Counterwise |
|
|
Is R going clockwise or counterclockwise |
Clockwise |
|
|
How do you decide if something is R or S? |
Assign priority groups Draw a circle from 1 --> 2 -->3 If 4 is into the page then it is orientated correctly if 4 is out of the page then is orientated opposite |
|
|
What has optical acitivity |
Pairs of enantiomers (opposite R/S designation) |
|
|
What does optical acivity mean? |
Can rotate plane polarized light |
|
|
Will enantiomers rotate the light in the same or opposite directions |
Opposite |
|
|
If you rotate light to the right is that D or L |
D |
|
|
If you rotate light to the right is that positive or negative |
+ |
|
|
If you rotate light to the left is that D or L |
L |
|
|
If you rotate light to the left is that + or -? |
- |
|
|
How do you calcualte specific rotation |
Observed rotation/ Concentration (g/ml) * length (dm) |
|
|
What is a racemic mixture? Does it have optical activity? |
Equal concentration of + and - so no optical activity |
|
|
n chiral centers hve ____ stereoisomers |
2^n |
|
|
What is an epimers? |
Diastereomer that only differs at one carbon center |
|
|
What is a meso compound |
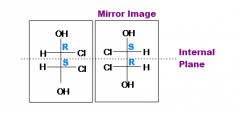
Multiple chiral centers with an internal plane of symmetry. |
|
|
Is a meso compound optically active? |
No |
|
|
What is a conformational isomer |
Differ by rotation about 1 or more single bond can be seen using Newman projections |
|
|
Practice drawing Newman Projections |
x |
|
|
What does a staggered anti newman's projection look like |
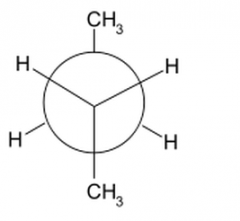
|
|
|
What does a gauche newman's projection look like? |
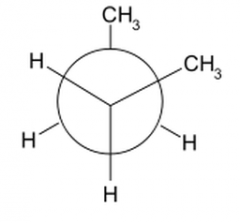
60 degrees |
|
|
What does an eclipsed newman's projection look like? |
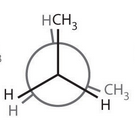
120 degrees |
|
|
What does a totally eclipsed newman projection look like? |
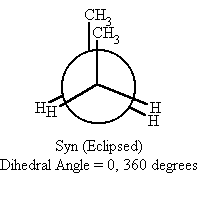
0 degrees |
|
|
What are the potential energy graph for all of the newman projections? |
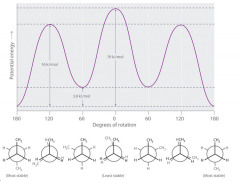
|
|
|
Will very low temperature slow or increase conformational interconversion |
Slow |
|
|
If you don't have enough energy to overcome barrier will the interconversion even rotate |
No |
|
|
What are the types of ring strain? |
*angle strain *Torsional strain *Non bonding strain |
|
|
What is angle strain? |
Bond angles deviate from ideal values |
|
|
What is torsional strain? |
Resistance to bond twisting Such as having the substitutents eclipsed |
|
|
What is non bonded strain? |
Atoms or groups compete for space |
|
|
What sort of orientation does the cyclobutane develop |
Puckered |
|
|
What sort of orientation does the cyclopentane have
|
Envelope |
|
|
What sort of orientation does cyclohexane have? |
*Boat *Chair *Twist boat |
|
|
What is the most stable cyclohexane conformation? |
Chair |
|
|
What is the lest stable cyclohexane conformation |
Boat All atoms are eclipsed |
|
|
What is the middle stable cyclohexane conformation |
Twist boat (Staggered gauche) |
|
|
How can interconservion between two chair conformaiton be stopped |
With bulky group attached |
|
|
Is equatorial or axial favored |
Equatorial Because axial has steric repulsion |

