![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
12 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
Neutralization reaction |
Acid and a base form a salt and water Acid starts with h Base end with OH Metal=grp 1&2 Nonmetal=halogen or poly atomic atom |
|
|
|
Ionization energy |
Ability to remove an electron |
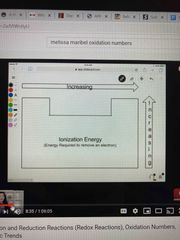
|
|
|
Electronegativity |
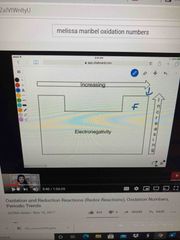
Fluorine is the most electronegative |
|
|
|
Electron affinity |
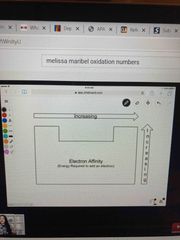
Energy required to add an electron |
|
|
|
Atomic radii |
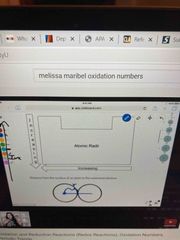
Distance from the nucleus of the atom to the outer most electron |
|
|
|
Oxidation number rules |
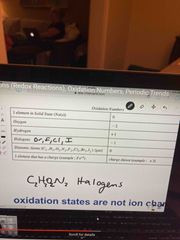
Plus group 1 has a +1 and group 2 has a +2 |
|
|
|
Halogens |
Br, F, Cl, I |
|
|
|
Diatomic atoms |
CHON then halogens Br, F, Cl, and I |
|
|
|
Elements |
Oxidation num = 0 |
|
|
|
Monatomic ions |
Same as it’s charge |
|
|
|
Nonmetals vs metals |
Nonmetal= negative numbers Some are positive in certain compounds ie: oxygen is -2 except in peroxide ion -1 Hydrogen is -1 when bonded to a metal and +1 when bonded to a nonmetal Florine always has -1 Other halogens mostly -1 |
|
|
|
Sum of oxidation numbers |
= 0 in a neutral compound = charge in polyatomoc ion |
|

