![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
9 Cards in this Set
- Front
- Back
|
Primary Structure Components Shape Bonds |
The sequence in which the amino acids of a protein are formed. This will determine how the protein folds. Bonds: Peptide bonds |
|
|
Secondary structure: Alpha Helix Amino Acids, interactions |
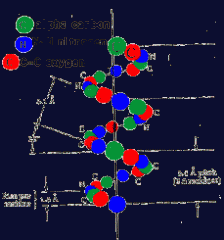
The oxygen, hydrogen and Nitrogen atoms of the basic repeating structure of amino acids interact. Hydrogen Bonds may form within the chain pulling it into a coil shape. The Alpha Helix |
|
|
Secondary structure: Beta-pleated sheet |
Polypeptide chains can lie parallel to each other and be joined by hydrogen bonds forming a sheet-like structure, the pattern appears pleated. |
|
|
Secondary Structure: R groups |
The coiling/folding of proteins into their secondary structure brings R-groups of different amino acids closer together. They are the close enough to interact |
|
|
Interactions between Amino Acids |
Hydrophilic/hydrophobic Polar/non polar r-group interactions Hydrogen Bonds Ionic Bonds (between oppositely charged R-groups) Disulfide Bonds - the strongest bonds only form between R-groups which contain sulfur atoms. |
|
|
Tertiary Structure |
The 'folding' of the polypeptide chain into its final shape. |
|
|
Quaternary Structure Interactions |
The association of two or more individual proteins called 'subunits'. The Interactions are the same as tertiary only between subunits and not within a molecule. |
|
|
Insulin |
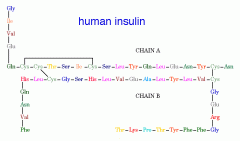
Two different subunits |
|
|
Haemoglobin |
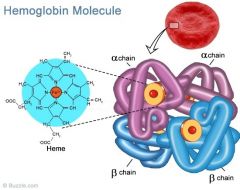
Four subunits, two sets of two identical subunits. Haem groups containing iron |

