![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
42 Cards in this Set
- Front
- Back
|
Aryl halide
|
Halogen bound to carbon of a benzene ring or another aromatic ring
|
|
|
Vinylic halide
|
halogen bound to carbon of a double bond
|
|
|
Allylic group
|
on carbon adjacent to the double bond
|
|
|
Benzylic group
|
on carbon adjacent to an aromatic ring
|
|
|
Do aryl halides undergo nucleophilic substitution reactions by the Sn1 or Sn2 mechanisms?
|
No
|
|
|
Phenol
|
Hydroxy bound to aromatic ring
|
|
|
Catechol
|
Two adjacent hydroxy groups on aromatic ring
|
|
|
What catalysts dramatically inc reactivity of aryl & vinylic haldies in substitution reactions?
|
transition-metal catalysts
|
|
|
Simple vinylic & aryl halides under sn2 conditions
|
Inert
|
|
|
Why don't vinylic halides undergo Sn2 reactions?
|
hybridization is sp during transition & VDW
|
|
|
Why don't aryl halides undergo sn2 reactions?
|
hybridization is sp during transition, VDW, backside approach would place nuc through plane of ring, & derivative would yield a twisted & highly strained double bond
|
|
|
Base promoted B-elim rxn vinylic halides
|
alkynes
|
|
|
Ph-CH=CH-BR + KOH @ 200 C
|
Ph-CH=-CH-H + K+ Br- + H2O
|
|
|
Ph-CH(Br)-CH(Br)-Ph + 2 KOH in C2H5OH
|
Ph-C=-C-Ph + 2K+ Br- 2H2O
|
|
|
Do vinylic eliminations require harsh conditions?
|
yes heat or strong bases & often enhanced acidity (i.e. benzylic)
|
|
|
Vinylic & aryl halides under Sn1/e1 conditions
|
inert (some forced under extreme conditions but uncommon)
|
|
|
H2C=C(CH3)-Br + C2H5OH --> 55C
|
no rxn
|
|
|
Ph-Br + C2H5OH --> 55C
|
no rxn
|
|
|
Why are vinylic & aryl halides inert under sn1 conditions?
|
unstable vinylic cation (sp hybridized, vacant 2p orbital not conjugated w pi e- system of db system) & e withdrawing polar effect of db discourages formation pos charge & carbon-halogen bonds stronger in vinylic halides (sp2 vs sp3 in alkyl)
|
|
|
Aryl cation
|
carbocation w e deficient carbon part of aromatic ring: carbon prefers linear but impossible too much strain - vacant orbital must remain sp2 = high E (& EW db)
|
|
|
Difference between aryl cation & cation in EAS
|
Carbocation in EAS stabilized by resonance, cation orthogonal (@ right angle) to pi e system
|
|
|
nucleophilic substitution reactions
|
Aryl halides w 1+ nitro group o/p to halogen under mild conditions
|
|
|
NAS obeys rate law
|
second order: K[aryl halide][nucleophile]
|
|
|
speeds up NAS
|
more nitro groups o/p to halogen LG
|
|
|
Reactivities of aryl hlides in NAS
|
Ar-F >> Ar-Cl ~ Ar-Br ~ Ar-I
|
|
|
NAS mechanism
|
nuc reacts @ halide-bearing carbon below/above plane aromatic ring to yield resonance stabilized Meisenheimer complex w neg charge delocalized thru pi e system of ring (RLS) & Nitro group --> loss of halide ion
|
|
|
Why do o/p nitro groups stabilize Meisenheimer?
|
Resonance
|
|
|
Why is strength of carbon-halogen bond (/basicity of halide) not important in determining reaction rate of NAS?
|
Loss of halide is not rate limiting
|
|
|
Frontside substitution of NAS stereochemistry
|
retention
|
|
|
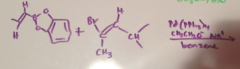
Heck Reaction Mechanism
|

|
|
|
Does Heck reaction occur w/o Palladium catalyst?
|
No
|
|
|
Stereochemistry Heck reaction
|
Retention
|
|
|
Transition metals
|
d block or B groups of periodic table
|
|
|
Why is it convenient to think of e in 4s & 3d orbitals as valence electrons?
|
similar energies
|
|
|
Ligands
|
Coordination compounds or transition-metal complexes: can be neutral or complex (have charge) - All are Lewis bases (donate e- pairs)
|
|
|
L-type ligand
|
dissociates from metal w bonding e pair & becomes neutral mlc - covalent
|
|
|
X-type ligand
|
dissociates from metal w bonding e pair & becomes negative ion - covalent
|
|
|
Dative bond
|
L-type bond emphasized by leaving bonding e pair on ligand & depicting bond as arrow from e to metal
|
|
|
How do alkenes or aromatic rings act as ligands?
|
By donating pi e to a metal
|
|
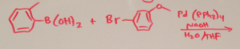
|
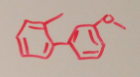
|
|
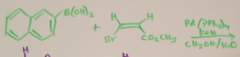
|
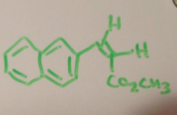
|
|
|

|

