![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
22 Cards in this Set
- Front
- Back
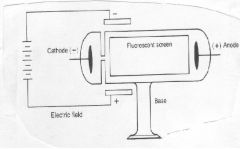
Which experiment involved this equipment, leading to the discovery of the electron by J. J. Thomson?
|
Cathode Ray Tube Experiment
|
|
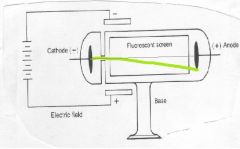
Thomson noticed that the beam of what looked like "light" bent toward the positive plate. This caused him to deduce that the beam is made of ______ charged _______.
|
negatively
particles |
|
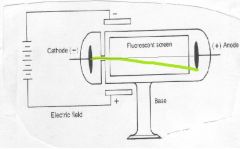
This experiment led to the discovery of _____. This led Thomson to create his ____ ____ model of the atom
|
electrons
Plum Pudding |
|
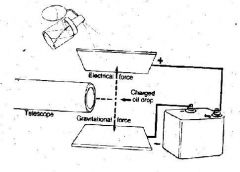
Millikan used this experiment to determine how much charge was on an electron. This experiment is called....
|
the Oil Drop Experiment
|
|
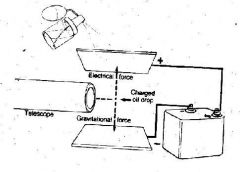
What was discovered with this experiment that involved oil being sprayed onto the upper plate?
|
the amount of negative charge on an electron
|
|
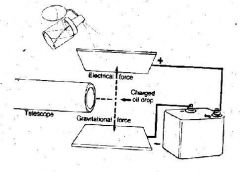
Gravity pulls objects downward. By altering the amount of electricity on the plates, Millikan was able to ___ an oil drop. The bottom plate ____ the drop while the top plate ____ the oil drop.
|
levitate
repelled attracted |
|
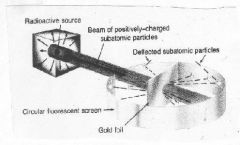
What experiment is this?
|
Gold Foil Experiment
|
|
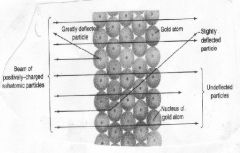
Rutherford wondered why there were some alpha (positive) particles bounced back or were deflected sideways. He concluded that....
|
the atom has hard centers that he would call nuclei
|
|
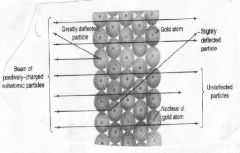
Not only did Rutherford propose that the atoms had hard centers, he deduced that the atom is mostly ____ ____, in which the electrons spin.
|
empty space
|
|
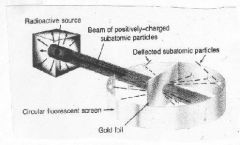
As a result of this experiment, Rutherfords model of the atom takes the electrons out of the nucleus and has them orbiting the nucleus much like.....
|
the planets circling the sun
|
|
|
Bohr did experiments whereby he energized elements with electricity or fire and made colors. His experiments were called....
|
spectroscopy
|
|
|
Bohr reasoned that the electrons absorb energy and move to new orbits and when they fall, they emit light that indicates how far they fell. This led him to deduce that electrons are in ____ ____
|
energy levels
|
|
|
Bohr's model of the atom is different from Rutherford's model in that he proposed that there are distinct ___ ___ in which the electrons reside, not random orbiting.
|
energy levels
|
|

Atom model of electrically neutral isotope: Protons = + & . neutrons not shown. Electrons not shown. Mass is given.
Number of protons = _____ Number of electrons = _____ Number of neutrons = _____ Atomic number = _____ Mass number= 135 This Isotope is ______ |
55p
55e 80n 55 (A#) Cs-135 |
|
Nucleus model of an electrically charged isotope: Protons = + & neutrons = O sign.
Number of protons = _____ Number of electrons = 12 Number of neutrons = _____ Atomic number = _____ Mass number = _____ This isotope is ______ What charge does this isotope have? ________ |
14p
16n 14 (A#) Si-30 +2 charge |
|
Nucleus model of electrically charged isotope:Protons = + & neutrons + O sign.
Number of protons = _____ Number of electron = 18 Number of neutrons = _____ Atomic number = _____ Mass number = _____ The isotope is _____ What charge does this atom have? ______ |
19p
22n 19 (A#) K-41 +1 charge |
|

Here is a square from the Periodic Table.
Number of protons = _____ Average atomic mass number = ____ Number of electrons = _____ Atomic number = _____ Number of neutrons = _____ |
protons = 29
mass number = 64 electrons = 29 Atomic number = 29 neutrons = 35 |
|

Number of protons = _____
Average atomic mass number = ____ Number of electrons = _____ Atomic number = _____ Number of neutrons = _____ |
protons = 42
mass # = 96 electrons = 42 atomic # = 42 neutrons = 54 |
|

Number of protons = ___
Average atomic mass number = ___ Number of electrons = ____ Atomic number = ____ Number of neutrons = ____ |
protons = 25
mass number = 55 electrons =25 Atomic number = 25 neutrons = 30 |
|

# of protons & neutrons together = ___
# particles, zero charge = __ # nucleons = ___ # particles, + charge = ____ # particles, (-) charge ____ |
# p & n = 64
# particles zero charge = 35 # nucleons = 64 # postives = 29 # negatives = 29 |
|

#protons & neutrons together in nucleus = _____
# particles,zero charge = __ # nucleons = ____ # particles, pos. chrge = ___ # particles, neg. chrge = ___ |
# p & n nucleus = 96
# particles, zero chrg = 54 # nucleons = 96 # positive charge = 42 # negative charge =42 |
|

# of protons & neutrons in nucleus = _____
# of particles, zero charge = ___ # of nucleons = ____ # of particles, positive charge = ___ # of particles, negative charge = ___ |
# p & n in nucleus = 55
# particles, zero chrg = 30 # nucleons = 55 # positive charges = 25 # negative charges = 25 |

