![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
38 Cards in this Set
- Front
- Back
|
The simplest arene |
Benzene |
|
|
How many degrees of unsaturation does benzene have |
4 degrees of unsaturation (1 ring and 3 double bonds) |
|
|
Why is benzene highly reactive |
It's high degree of unsaturation |
|
|
Benzene only undergoes substitution reaction. It does NOT undergo addition reaction like the other alkenes. Why? |
Because of it's high degree of unsaturation. |
|
|
Addition reaction of benzene with bromine |

|
|
|
Substitution reaction of benzene with halogens |

Benzene does not undergo addition reaction with halogens but will only react with them in the presence of a Lewis acid(electron pair donor) e.g FeBr3, AlCl3 or FeCL3 in a substitution reaction. |
|
|
Resonance structure of benzene |

|
|
|
How many pie bonds and electrons does benzene have |
3 π bonds and 6 π electrons |
|
|
Why is benzene called a conjugated compound |
Because of the alternation between single and double bonds |
|
|
Ethyl benzene |

|
|
|
Tert-butyl benzene |
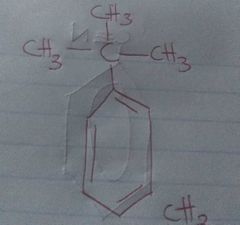
2-methyl propyl benzene |
|
|
Chloro benzene |

|
|
|
Toluene |

Methyl benzene |
|
|
Phenol |

Hydroxylbenzene |
|
|
Aniline |

Amino benzene |
|
|
Anisol |

Methoxybenzene |
|
|
Benzonitrile |

|
|
|
Benzoic acid |

|
|
|
Benzaldehyde |

|
|
|
Acetophone |

|
|
|
Mesitylene |

1,3,5-methyl benzene |
|
|
TNT |

2,4,6-trinitrotoluene. An explosive. |
|
|
What is used to show the relative positions of two substituents on a benzene ring |
Ortho, meta and para nomenclature |
|
|
Orthodibromobenzene |

1,2-dibromobenzene; o-dibromobenzene |
|
|
Phenyl group (Ph-) |

C6H5; a benzene that has lost one hydrogen atom |
|
|
Benzyl group |

C6H5CH2; a benzene with an extra CH2- attached. |
|
|
Types of compounds based on aromaticity |
Aromatic, antiaromatic and non aromatic compounds |
|
|
Criteria for aromaticity |
1) the molecule must be cyclic (i.e a closed ring) 2) the molecule must be planar 3) the molecule must be completely conjugated ( i.e alternation of single and double bond) 4) the molecule must satisfy Huckel's rule |
|
|
When is a compound most likely to lose planarity |
When it has more than 7sides |
|
|
When is a compound said to be planar |
All adjacent p orbitals must be aligned (overlap) so that the π electron density can be delocalized |
|
|
Huckel's rule for aromatic compounds |
It must contain 4n + 2 π electrons. |
|
|
What is an antiaromatic compound based on Huckel's rule |
An antiaromatic compound is a cyclic, planar, and completely conjugated compound that contains 4n π electrons |
|
|
Difference in stability of aromatic and anti aromatic compounds |
Aromatic compounds are more stable than anti aromatic compounds |
|
|
What is Huckel's rule concerned with |
It deals with the number of π electrons not the number of atoms in a particular ring. |
|
|
Illustration of Huckel's rule |

|
|
|
How is benzene an aromatic compound based on Huckel's rule |

It has 6 π electrons. From the table, this falls under the aromatic table |
|
|
What kind of compound is cyclobutadiene |

It is an anti aromatic compound because it has 4 π electrons that fall into the antiaromatic table |
|
|
Priority functional group when naming aromatic compounds |
1) carboxylicacid (-COOH) 2) ester (-COO-) 3) acid chloride (CoCl) |

