![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
54 Cards in this Set
- Front
- Back
|
Step 1: acylation of benzene mechanism |
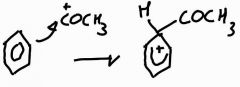
|
|
|
Step 2: acylation of benzene mechanism |
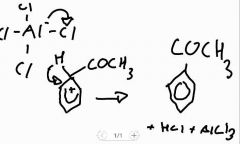
|
|
|
Name the reagents in the acylation of benzene, state the catalyst |
Ethanoyl chloride and the catalyst is aluminium chloride |
|
|
Name the electrophile in the acylation of benzene |
CH3C^+=O |
|
|
How is the electrophile in the acylation of benzene formed? |
The reaction between ethanoyl chloride and aluminium chloride |
|
|
Draw out the structured equation for the reaction of benzene and ethanoyl chloride |
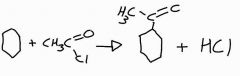
|
|
|
Equation to form the electrophile in the acylation of benzene |
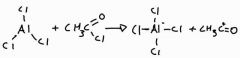
|
|
|
Name a source of the electrophile SO3 |
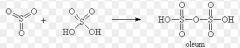
Fuming sulfuric acid H2S2O7 |
|
|
What temp does the sulfonation of benzene occur at? |
Room temp = 298k |
|
|
In the nitrate on of benzene, what is benzene treated with and temp required |
Con nitric acid and con sulphuric acid , not exceeding 50 oc |
|
|
In the nitration of benzene, as temp increases there's more chance of... |
More than one nitro group substituted on the ring |
|
|
Benzene doesn't react with nitric acid , why? |
It's not an ideal electrophile |
|
|
What is the overall equation of the nitration of benzene? |
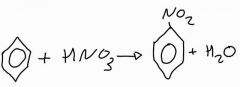
|
|
|
Name the catalyst in the nitration of benzene |
Sulthuric acid |
|
|
Halogenation of benzene in the process of aluminium chloride or.. |
Iron bromide |
|
|
Stage 1: halogenation of benzene with aluminium chloride |
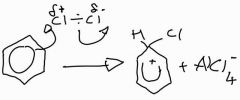
|
|
|
Stage 2: halogenation of benzene with aluminium chloride |
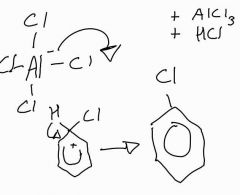
|
|
|
Stage 1: halogenation of benzene with Fe (III) bromide (in the dark) mechanism |
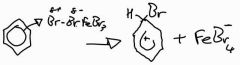
|
|
|
Halogenation of benzene in UV light results in which sort of reaction |
Addition reactions |
|
|
What can be used instead of iron (III) in the halogenation of benzene? Give the equation |

Iron can be used instead because it reacts with some of the bromine present |
|
|
In the halogenation of benzene give the equation of the polarised electrophile |
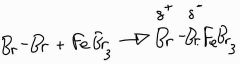
The electrophile first forms a weak pi complex and then the more stable omega bond |
|
|
Stage 1: halogenation of benzene with iron (III) bromide |
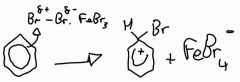
|
|
|
Stage 2: halogenation of benzene using Fe (III) bromide |
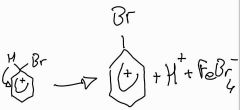
|
|
|
In stage 2 of the halogenation of benzene a proton is lost and then reacts to regenerate the catalyst state the catalyst and give the equation: |
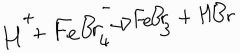
|
|
|
What does alkylation mean |
Substituting an akyl group into a benzene ring |
|
|
During alkylation of benzene what do yoy reflux benzene with |
With a halogenalkane in the presence of a halogen carrier catalyst , such as aluminium chloride |
|
|
Why does benzene have a higher boiling point than ethanol? |
London forces between benzene molecules are stronger than the hydrogen bonds between ethanol molecules |
|
|
Draw out the struture of benzenediazonium ion |
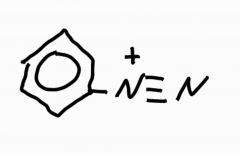
|
|
|
Temperature of benzenediazonium ion must be kept below 10 degrees because it eill turn into a.. |
Phenol |
|
|
Why is a three step synthesis greener than a six step? |
Yield is higher |
|
|
What is a benefit of using a solid catalyst? |
Catalyst is more easily recovered |
|
|
Phenylamine and benzene react with bromine in a similar way. But phenylamine reacts faster, due to the fact that... |
Lone pair of electrons of the nitrogen has on the benzene ring |
|
|
What is the ion formed when dilute nitric acid reacts with phenylamine |
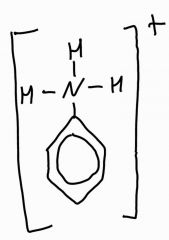
|
|
|
Stage 2: Sulfonation of benzene mechanism |
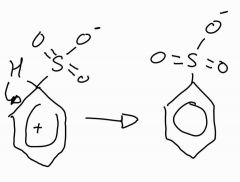
|
|
|
Stage 3 of sulfunation of benzene |
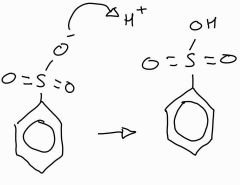
|
|
|
What is the equation of the two stage process of benzenesulfonic with sodium hydroxide forming an intermediate and water? Stage 2 reaction is with HCl |
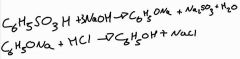
. |
|
|
The standard enthalpy change of benzene is less than the kekule struture. Benzene is more stable. What does this tell you about the bonding in benzene? How does this influence the type of chemical reactions that benzene undergo? |
Benzene is more stable by 152 Double bond electrons are deolciased Suggesting a resonance hybrid Consisting of C-C and C=C Substitution reactions rather than addition The delocalised system remains stable |
|
|
Name the end product of the sulfonation of benzene |
Benzene sulfanoic acid |
|
|
Why is phenol and methoxybezne more reactive than benzene? Oxygen |
The oxygen have lone pairs which effect the benzene system, which increases the electron density of the system Makes it more susceptible to electophile attacks |
|
|
Why is sulfur trioxide a good electrophile? |
It's a polar molecule with three electronegative oxygen attached to the sulfur, giving sulfur a slight postive charge |
|
|
Stage 1: Sulfonation of benzene mechanism |
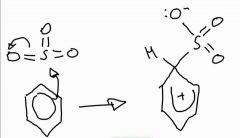
|
|
|
Give the equation for the formation of the electophile in the nitration of benzene |
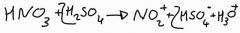
|
|
|
Stage 1: nitration of benzene mechanism |
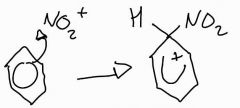
As electophile approaches the benzene, deolciased electrons are strong attracted to the postive charge. Two electrons are part of the system anymore, ring becomes postive |
|
|
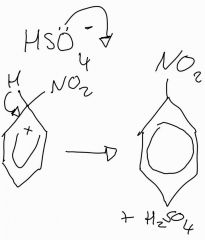
Stage 2: Nitration of benzene mechanism |
|
|
|
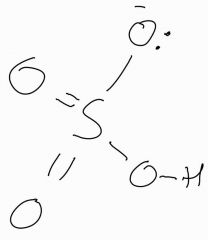
Draw the struture of HSO4^- |
|
|
|
State the two roles the catalyst plays in the nitration of benzene |
Removes the hydrogen from the ring and therefore is regenerated. The electrons are now restablised in the system |
|
|
Apparatus of nitration of benzene |
Round bottom flask held in cold water. The flask has a tap funnel containing benzene. Benzene should be slowly added. Heat is given out, hence why cold water is needed. |
|
|
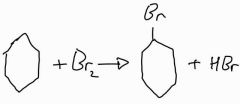
The overall equation of halogenation of benzene |
|
|
|
What is the overall equation of alkylation of benzene? |
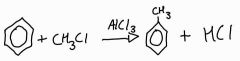
|
|
|
What effect does the aluminium chloride have on the halogenalkane molecule in the alkylation of benzene |
Polarises the halogenalkane molecule, promoting the formation of an electophilic alkyl cation |
|
|
What is the electophile in the alkylation of benzene, write the equation for the electrophile |
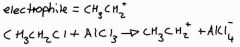
|
|
|
Stage 1: alkylation of benzene mechanism |
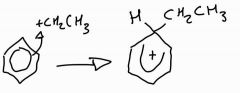
|
|
|
Stage 2: alkylation of benzene mechanism |
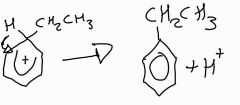
|
|
|
In the alkylation of benzene, what is the equation for the regeneration of the catalyst? |
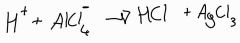
|

