![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
35 Cards in this Set
- Front
- Back
|
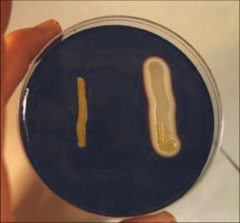
STARCH BREAKDOWN Left: Negative (Gram's iodine able to complex with starch)
Right: Positive (starch breakdown in zone of clearance, no complexation) |
|
|
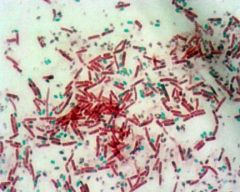
SPORE STAIN - pink/red are vegetative cells due to safranin counterstain - green are spores due to malachite green |
|

|
UREASE TEST - middle is negative - others are positive; turned pink due to alkali production from urea |
|

|
HEMOLYSIN PRODUCTION/HEMOLYSIS TEST - first is a-hemolysis; no zone of clearance but green area is due to Hb oxidation to methemeglobin; technically, no lysis occurred - seen in viridans group (verte=green) - ex. S. aureus
- middle is b-hemolysis; zone of clearance indicates lysis; seen in pyogenic group -ex. B. cereus
- last is y-hemolysis; no lysis - ex. E.coli |
|

|
COAGULASE TEST - left is positive due to clotting - ex. S. aureus
- right is negative - ex. S. epiderdimis |
|
|
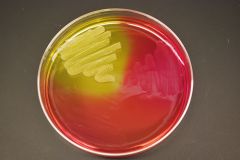
MANNITOL SALT AGAR PLATE - yellow is pathogenic Staphylococcus aureus (grows in salt, uses mannitol and makes acid) - clockwise is S. epiderdimis; it can tolerate salt but doesn't use mannitol - below is E.coli; doesn't grow in salt and doesn't mannitol |
|
|
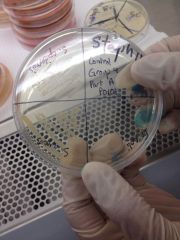
STAPHYLOCOCCUS MEDIUM 110 - S. aureus (bottom left) has orange growth due to mannitol breakdown and can live in salt - S. epidermidis has white growth since it doesn't use mannitol but can still live in salt - E. coli has no growth |
|

|
INDOLE TEST - left is positive: Kovak's reagent forms red ring immediately because it combines with indole (formed from Trp) - right is negative |
|
|
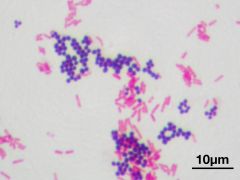
GRAM'S STAIN purple: positive pink: negative |
|
|
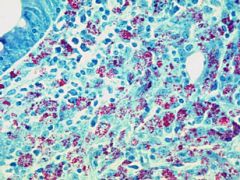
ACID FAST Purple: Acid-fast positive due to mycolic acid (ex. Mycobacterium, M. plei); stained by carbol fuschin
Blue: negative; was decolourized by acid-alcohol and counter-stained by methylene-blue (ex. S. epiderdimis) |
|

|
HANGING DROP E. coli: rods gently moving in waves P. vulgaris: gentle movement S. epiderdimis: |
|

|
MOTILITY AGAR: Left is positive; bacteria grew outside central tube by moving (ex. E.coli)
|
|
|
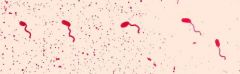
FLAGELLA STAIN - bacteria placed on one end of slide - slide was tilted and air-dried - fixed with mordant - add Ziehl's carbol fuschin and wash with water
|
|
|
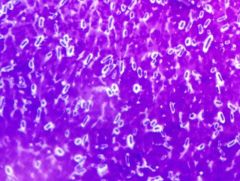
CAPSULE STAIN - capsules are blue; purple due to crystal violet - Klebsiella pneumoniae is positive
- capsules are extracellular layer formed to fight phagocytosis |
|
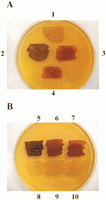
|
GLYCOGEN MICROCHEM TEST - Gram's iodine was added to suspension of Saccharomyces cerecisiae - glycogen turns reddish brown |
|

|
FAT RESERVE TEST - Sudan Black dissolves in fat and stains black - Bacillus megaterium |
|
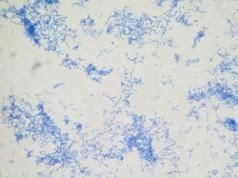
|
VOLUTIN TEST - B. cereus, stained with acidified methylene blue |
|

|
CARB TEST - if BTB turns, acid production due to glucose metabolism - inverted vial may also have bubbles (CO2) |
|
|
INVERTASE TEST |
- uses Fehling test - sucrose is hydrolyzed by invertase to glucose and fructose - solution should go from blue to red |
|
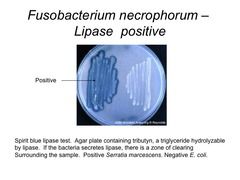
|
LIPASE TEST - hydrolysis of lipid to glycerol and fatty acids; is oxidized to yield dark blue ppt. - B. cereus and E. coli are positive - S. aureus is negative
|
|
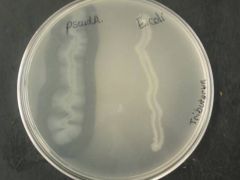
|
CASEIN HYDROLYSIS TEST - casein gives the cloudy white colour of milk - E. coli does not have casein (right) - B. cereus is casein positive (clear zones) |
|

|
GELATIN HYDROLYSIS - left is positive; remained liquid despite temperature being below melting point of gelatin - B. cereus is positive |
|

|
DECARBOXYLATION TEST - enzymes cleave COOH group to form amine and CO2 - amine leads to pH increase; CO2 detected by inverted vial - at yellow, acidic ex. Proteus vulgaris - at purple, basic (positive)... ex. Salmonella typhimunum |
|

|
CATALASE TEST - 2H202 -> 2H2O + O2 - oxygen observes as white froth - ex. B. cereus, E.coli, S. aureus are positive |
|

|
OXIDASE TEST - positive if it immediately turns purple (ex. B. cereus) upon addition of oxidizing reagent, which is reduced by cytochrome C in ETC |
|

|
O/F Test - start with blue solutions (bromothymol blue indicator), one with layer of oil - after incubation, if both still blue/green, no oxidation/fermentation occurred and thus, glucose metabolism not performed - if oil is blue but open is yellow, oxidation occurred only - if both are yellow, could be fermentation or both |
|

|
CITRATE TEST - start with green Kosher's citrate medium - if it turns blue, citrate was used as the carbon source |
|
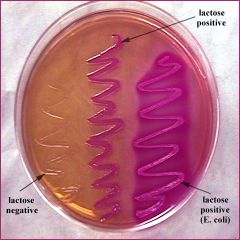
|
MACCONKEY AGAR PLATE - both selective and differential - non-enteric bacteria will not grow - enteric and lactose fermenting (coliform) will have pink colonies (ex. E. coli) - enteric but non-lactose fermenting will yield tan colonies |
|

|
METHYL RED TEST - tests for stable acid production between coliforms - those that ferment glucose will cause pH decrease and make indicator red (Ex. E. coli) - in others, pyruvic acid is decarboxylated and pH is raised, becomes yellow (ex. K. aerogenes) |
|

|
VP Test - tests for acetoin presence - turns peach/pink upon addition of reagent makes it positive (ex. B. cereus) |
|

|
NITRATE REDUCTION TEST 1) Add naphthalene and sulphanilic acid, if it turns red (2nd), it has nitrase reductase and nitrite is detected 2) If no colour change (1st), add zinc. 3) If addition of Zn still makes it colourless, it has nitrate and nitrite reductase (positive) 4) If addition of Zn makes it red (4th), negative nitrate reductase |
|
|
What happens when you plate bacteria on MacConkey agar plate? |
Lactose-fermenting bacteria make red colonies
Non-lactose fermenting bacteria make white colonies |
|
|
Provide the names of the following AB: 1. C 2. P 3. AM 4. N 5. S 6. TE 7. E 8. SSS |
1. Vanconmycin 2. Penicillin 3. Bacitracin 4. Neomycin 5. Streptomycin 6. Tetracylin 7. Erythromycin 8. Triple sulpha |
|
|
Components of PCR Master Mix |
- pH buffer (Tris-HCl) - dNTPS - reverse and forward primers - MgCl2 - KCl - Taq polymerase
|
|
|
Steps of PCR |
1. 95C for 10 minutes 2. 30 cycles of 95C for 60 sec to denature and separate the two DNA strands; 55C for 60 sec so primers can bind to ssDNA; 72C for 60sec to extend DNA 3. 72C for 10 minutes |

