![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
11 Cards in this Set
- Front
- Back
|
How element is arrange in periodic table |
*Increasing order of proton *Same group, Same chemical properties *7 horizontal row- period (no of shell occupied) * 18 vertical column- group ( no of valence electron) |
|
|
G18 - NOBLE GAS (properties) |
* mono atomic ,, chemically unreactive * insoluble in water * poor conductor of heat and electricity * GOING DOWN - melting n boiling point in crease * inert - duplet/ octet |
|
|
Why melting n boiling point of noble gas increase when going down grup18? |
Since atomic size increase down the group, force of attraction become stronger. Thus, more heat require to overcome the force |
|
|
List of group 18 |
1) helium 2) neon 3) argon 4) krypton 5) xenon 6) radon |
|
|
Group 1 physical properties |
* soft metal, low density, low melting point * silvery shiny metal * good conductor of heat n electricity * going down - atomic size increase - hardness, melting n boiling point decrease |
|
|
Chemical properties alkali metal |
"Burn in oxygen "Dissolve in water "Burn in chlorine gas "Burn in bromine gas |
|
|
Alkali metal rx |

|
|
|
Chemical properties |

Going down: - darker - increase atomic size - increase melting n boiling point -Physical state change.- Gas to solid |
|
|
Chemical properties halogen |
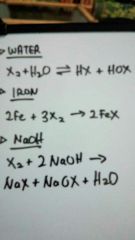
|
|
|
Period 3 properties (semi metal) |

|
|
|
Transition element properties (metal) |
-High melting n boiling point -Good conductor of heat and electricity 3 Special characteristics: 1) differ oxidation no in their compound 2) form coloured ion / compound 3) good catalyst |

