![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
36 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
Who were the scientists who contributed to the atomic model? |
John Dalton (1808), J.J. Thomson (1897), Hantaro Nagaoka (1904), Ernest Rutherford (1911), Niels Bohr (1913), Edwin Schrodinger (1925), James Chadwick (1932) |
7 people |
|
|
John Dalton (1808)'s discoveries? |
- all elements are composed of atoms - atoms of the same element are identical - atoms cannot be broken down any further - two or more elements combined make up molecules and compounds |

|
|
|
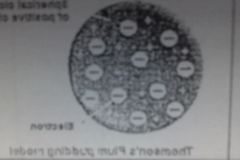
J.J Thomson (1897) |
- atoms were a solid, pudding-like body with a positive charge - negatively charged electrons particles were scattered throughout the pudding-like positive body |
|
|
|
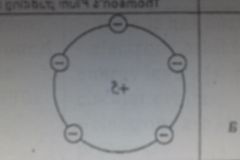
Hantaro Nagaoka (1904) |
- Atoms have a center sphere that is positively charged - negatively charged electrons orbit the positively charged center - Atom model looked a bit like a solar system or a planet with a ring |
|
|
|
Ernest Rutherford (1911) |
- able to prove atoms a positively charged nucleus - proposes atoms are mostly filled with empty space and have electrons orbiting the nucleus in random patterns |

|
|
|
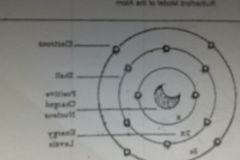
Niels Bohr (1913) |
- determined that electrons move in definite orbital levels (energy levels or shells) around the nucleus - electrons can move from one shell to another by giving off or absorbing energy |
|
|
|
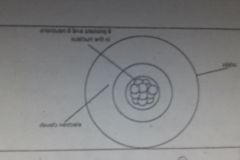
Edwin Schrodinger (1925) |
- proposes electrons orbit the nucleus in a wave shape rather than perfectly circular orbits (thus creating a cloud of electron activity around the nucleus) |
|
|
|
James Chadwick (1932) |
- discovered the neutron (neutral particle in the nucleus of an atom) |
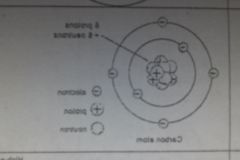
|
|
|
How did Mendeleev arrange the first Periodic Table of Elements? |
Eventually arranged by order of atomic mass and bonding power |
|
|
|
Family 1 |
Alkali metals: Most Reactive all have 1 valence electron |
|
|
|
Family 2 |
Alkaline earth metals: not as reactive
|
|
|
|
Family 3 |
Boron: 3 valence electrons |
|
|
|
Family 4 |
Carbon: gain, lose, or share 4 electrons when reacting 4 valence electrons |
|
|
|
Family 5 |
Nitrogen: 5 valence electrons Tend to share electrons when they bond |
|
|
|
Family 6 |
Oxygen: share electrons when they bond combines with almost all elements |
|
|
|
Family 7 |
Halogen family: most active non metals never found free in nature react with alkali metals to form salts |
|
|
|
Family 8 |
Noble gases: all are gas at room temp, extremely unreactive
|
|
|
|
What do the groups show? |
each group has elements that share similar properties |
|
|
|
Names of groups in order |
Alkali metals, Alkaline Earth Metals, Transition metals, rare-earth metals, Metaliods or semi-metals, non metals, halogens, nobel or inert gases, |
|
|
|
What is the general name for the columns in the periodic table? |
Group |
|
|
|
What is the general name for the rows in the periodic table? |
Periods |
|
|
|
How can u tell the number of valence electrons in an element? |
Its the same number as its family |
|
|
|
How can you tell the number of energy levels in an element? |
Its the same number as its period |
|
|
|
What elements are gas at room temp? |
all of the inert gases (He, Ne, Ar, Kr, Xe, Rn) and H, N, O, F, Cl |
henoffcle |
|
|
What elements are liquid at room temp? |
Br (Bromine) and Hg (Mercury) |
|
|
|
What are the natural elements? |
elements 1-92 |
|
|
|
What are the man-made elements? |
elements 93+ (and up) |
|
|
|
What are the radioactive elements? |
elements 83+ (and up) |
|
|
|
Alpha decay |
not a very harsh damage done to living organisms release of 1 alpha particle decreases atomic number by 2 and mass number by 4 can be blocked by paper |
alpha particle: consists of 2 protons and 2 neutrons |
|
|
Beta decay |
medium damage done release of 1 beta particle mass number stays the same but atom number increases by 1 can be blocked by aluminum sheet |
beta particle: a fast moving electron given off by a nucleus |
|
|
Gamma decay |
severe and harsh damage gamma radiation is the release of high energy waves atomic number and mass number stay the same can barely be blocked by concrete wall |
|
|
|
What is mass? |
a measure of how much matter is in an object |
|
|
|
What is volume? |
the amount of space that matter occupies |
|
|
|
What is density? |
The ratio of the mass of a substance to its volume |
|
|
|
What is the equation for density? |
Denisty= Mass/volume |
|
|
|
If you know that the density of water is 1g/cm3, how can you determine if an object will float if you are not allowed to place the object in water? |
If it has a lower density, meaning it is less dense, it will float if it has a higher density, meaning it is more dense, it will sink |
|

