![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
5 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
What is an Exothermic reaction? |
A reaction where energy exits the reaction (it gets hot) |
EXOthermic, what happens to the energy in the reaction? |
|
|
What is an endothermic reaction? |
A reaction where energy enters the reaction (it gets cold) |
ENDOthermic, what happens to the energy in the reaction? |
|
|
What are two examples of an Exo and Endothermic reaction (one each) |
Exothermic: HCL + Magnesium ribbon Endothermic: NaHCO3 + citric acid |
Think elements |
|
|
Describe energy profile diagrams for both endo and exothermic reactions, labeling the energy change and the progress of reaction. (Draw in head if possible) |
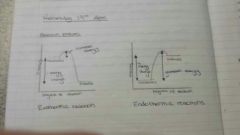
Exothermic: y-axis= energy change X-axis= progress of reaction reactants straight line at the top, goes up to a dome and then drops to a straight line labeled products Endothermic: (same x and y-axis) Reactans straight line at bottom, rises up to a dome and then drops a little to a straight line of products |
Think x and y-axis and what the graph would look like in energy change (ENDO? EXO? What happens to the energy?) |
|
|
Describe what is meant by activation energy |
Activation energy is the minimum amount of energy for particles to react |
|

