![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
56 Cards in this Set
- Front
- Back
|
Enzymes can be __ or __
|
Proteins or RNA
|
|
|
In one sentence, how to enzymes catalyze reactions?
|
Enzymes catalyze reactions by stabilizing transition states.
|
|
|
What determines the specificity of an enzyme?
|
The interactions of substrate with enzyme, as a result of 3D structure of enzyme protein.
|
|
|
What are tightly bound cofactors called?
|
Prosthetic Groups
|
|
|
What are small organic molecules that act as cofactors called?
|
Coenzymes
|
|
|
What are coenzymes?
|
Small organic molecules that act as cofactors (activating apoenzyme). Usually loosely associated with enzyme; can be used by a variety of enzymes.
|
|
|
How can we tell if a reaction will occur spontaneously?
|
If ∆G is negative = exergonic
|
|
|
What does exergonic mean?
|
∆G is negative, and the reaction can occur spontaneously
|
|
|
What is ∆G if a system is at equilibrium?
|
Zero -- no net change can take place
|
|
|
What does it mean if ∆G = 0?
|
The system is at equilibrium and no net change can take place
|
|
|
What is ∆G if a reaction cannot occur spontaneously?
|
Positive = endergonic
|
|
|
What does it mean if ∆G is positive?
|
The reaction can't occur spontaneously, it is said to be endergonic
|
|
|
What does the ∆G of a reaction depend on?
|
The free energy of the products (final state) minus the free energy of the reactants (initial state). Does not depend on the mechanism of reaction
|
|
|
Does the mechanism of the reaction have an effect on the ∆G (whether the reaction can occur spontaneously)?
|
No, only depends on free energy of products and reactants
|
|
|
Does ∆G tell us about the rate of reaction?
|
No. Tells us whether reaction can occur spontaneously, but not whether it is at a realistic rate.
|
|
|
What does the free energy of activation tell us?
|
The rate of reaction. Largely unrelated to the ∆G of the reaction.
|
|
|
What can enzymes alter?
|
Can alter reaction rate.
NOT reaction equilibrium. |
|
|
How do enzymes accelerate reactions?
|
They facilitate the formation of transition states. They decrease the activation energy.
|
|
|
What is "activation energy"?
|
The difference in free energy between transition state and substrate. Also known as Gibbs free energy of activation.
|
|
|
Do enzymes shift equilibrium positions?
|
No
|
|
|
What accounts for the equilibrium of a reaction?
|
The free energy of reactants and products
|
|
|
What are the attractions between enzyme and substrate?
|
Weak, noncovalent attractions such as hydrogen bonds or van der Waals.
|
|
|
What is the study of rates of enzyme-catalyzed reactions called?
|
Enzyme Kinetics
|
|
|
What is competitive inhibition?
|
The inhibitor binds to the same active site as the substrate
|
|
|
How does a competitive inhibitor work?
|
It diministhes the rate of catalysis by reducing the proportion of enzyme molecules bound to a substrate
|
|
|
What types of inhibition can be overcome by adding more substrate?
|
Competitive inhibition (more substrate - more likely it will bind to enzyme rather than inhibitor binding to enzyme). Uncompetitive inhibition cannot be overcome by adding more substrate.
|
|
|
What is non-competitive inhibition?
|
The inhibitor and substrate have different active sites. Cannot be overcome by adding more substrate.
|
|
|
What does the formation of ES complex depend on?
|
Both the [S] and [E]:
more like a 2nd order reaction: E+S ⇋ ES ⇋ E+P |
|
|
What is noncompetitive inhibition?
|
Inhibitor binds to non-active site. Substrate can still bind, but no product can form from ESI.
|
|
|
What type of inhibition prevents substrate from binding to enzyme?
|
Competitive inhibition
|
|
|
What type of inhibition occurs when inhibitor binds to site other than the active site: substrate can still bind but no product is formed?
|
Noncompetitive inhibition
|
|
|
What are the different types of inhibition?
|
Reversible:
* Competitive * Non-competitive |
|
|
What does it mean if:
ΔG < 0 |
Exergonic: reaction will occur spontaneously
|
|
|
What does it mean if:
ΔG > 0 |
Endergonic: needs an input of energy for reaction to occur
|
|
|
Does ΔG correlate with rate of reaction?
|
NO
|
|
|
Competitive Inhibition
|
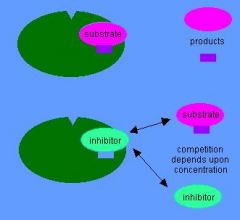
What type of inhibition is this?
|
|
|
What does non-competitive inhibition look like?
|
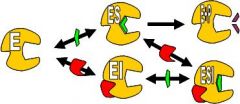
Non-Competitive Inhibition
|
|
|
What regulates enzymes
|
Allosteric Regulation
|
|
|
What is a Ligand?
|
An ion or molecule attached to a metal atom by coordinate bonding.
|
|
|
What does allostery refer to?
|
A change in shape as result of the binding of a ligand
|
|
|
Give two examples of inorganic cofactors
|
Mg and Zn
|
|
|
Give two examples of organic cofactors
|
Vitamins and Coenzyme A
|
|
|
What is a distinguishing feature of an allosteric enzyme?
|
The presence of allosteric sites "regulatory sites"
|
|
|
Are allosteric sites separate from the active sites?
|
Yes
|
|
|
What are the two forms of allosteric enzymes
|
low affinity and high affinity forms
|
|
|
What binds the alloseric site?
|
Allosteric effector
|
|
|
What does the allosteric activator do?
|
Stabilizes active state of allosteric enzyme
|
|
|
What does the allosteric inhibitor do?
|
Stabilizes inactive state of allosteric enzyme
|
|
|
What is an example of allosteric enzyme cooperativity and how does it work?
|
Oxygen binding in Hb. Substrate binds to activation site and changes enzyme shape which increases catalytic activity
|
|
|
What is another term for "end product inhibition"?
|
Feedback inhibition
|
|
|
Describe Feedback inhibition
|
An end product binds to the allosteric site which changes the enzyme shape so that the initial substrate cannot bind with the active site.
|
|
|
In terms of thermodynamics, what can photosynthesis be thought of as?
|
An anabolic process
|
|
|
What bonds the phosphate groups in ATP?
|
phosphoanhydride bonds
|
|
|
What two macromolecules are bonded by ester linkages?
|
ATP and lipids
|
|
|
What type of reaction occurs with the hydrolysis of ATP
|
Exergonic
|
|
|
What four ways do enzymes lower the Energy of Activation?
|
Active site act as template
Induced fit Suitable micro environment in active site covalent bond reorganization |

