![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
133 Cards in this Set
- Front
- Back
|
What is the mechanism of Heparin? |
Cofactor for the activation of antithrombin, ↓ thrombin, and ↓ factor Xa
Short t1/2 |
|
|
What are the uses of Heparin?
|
- Immediate anticoagulation for Pulmonary Embolism, Acute Coronary Syndrome, MI, and Deep Venous Thrombosis
- Used during pregnancy (doesn't cross placenta) |
|
|
What do you need to monitor in patients taking Heparin?
|
Follow PTT
|
|
|
What are the toxic side effects of Heparin?
|
- Bleeding
- Thrombocytopenia (HIT) - Osteoporosis - Drug-drug interactions |
|
|
What is HIT?
|
Heparin Induced Thrombocytopenia (HIT)
- Development of IgG antibodies against heparin bound to platelet factor 4 (PF4) - Antibody-heparin-PF4 complex activates platelets → thrombosis and thrombocytopenia |
|
|
How do you rapidly reverse an overdose of Heparin? Mechanism?
|
Protamine Sulfate (positively charged molecule that binds negatively charged heparin)
|
|
|
What are the types of low molecular weight heparins (LMWH)? Mechanism? How are they different?
|
Enoxaparin and Dalteparin
- Act more on Factor Xa - Better bioavailability and 2-4 times longer half-life - Can be administered subcutaneously and without lab monitoring - Not as easily reversible |
|
|
What drugs can you use in a patient who requires anticoagulation but was taking heparin and got heparin-induced thrombocytopenia (HIT)? Source?
|
Argatroban or Bivalirudin
- Derivatives of hirudin, the anticoagulant used by leeches |
|
|
What is the mechanism and use of Argatroban?
|
- Inhibits thrombin directly
- Used instead of heparin for anticoagulating patients with Heparin-induced thrombocytopenia (HIT) |
|
|
What is the mechanism and use of Bivalirudin?
|
- Inhibits thrombin directly
- Used instead of heparin for anticoagulating patients with Heparin-induced thrombocytopenia (HIT) |
|
|
What is the mechanism of Warfarin?
|
Interferes with normal synthesis and γ-carboxylation of vitamin K-dependent clotting factors II, VII, IX, and X and proteins C and S
|
|
|
What clotting factors and proteins are affected by Warfarin?
|
- Factor II
- Factor VII - Factor IX - Factor X - Protein C - Protein S |
|
|
What is the effect of warfarin on blood tests?
|
Increases PT (extrinsic pathway) / INR
|
|
|
What are the clinical uses of Warfarin?
|
- Chronic anti-coagulation (after STEMI, venous thromboembolism prophylaxis, and prevention of stroke in atrial fibrillation)
*NOT used in pregnant women (can cross placenta = teratogen) |
|
|
What are the toxic side effects of Warfarin?
|

- Bleeding
- Teratogen - Skin / tissue necrosis - Drug-drug interactions |
|
|
How can you reverse a warfarin overdose?
|
- Give Vitamin K
- For severe overdose, give fresh frozen plasma |
|
|
What drugs are direct factor Xa inhibitors?
|
- Apixaban
- Rivaroxaban |
|
|
What is the mechanism of Apixaban and Rivaroxaban?
|
Binds and directly inhibits activity of factor Xa
|
|
|
What are the clinical uses of Apixaban and Rivaroxaban?
|
- Treatment and prophylaxis of DVT and PE (rivaroxaban)
- Stroke prophylaxis in patients with atrial fibrillation - Oral agents do not require coagulation monitoring! |
|
|
What are the side effects of Apixaban and Rivaroxaban?
|
Bleeding (no specific reversal agent available)
|
|
|
What is the structure of heparin and warfarin?
|
- Heparin: large, anionic, acidic polymer
- Warfarin: small lipid-soluble molecule |
|
|
What is the route of administration of heparin and warfarin?
|
- Heparin: parenteral (IV, SC)
- Warfarin: oral |
|
|
What is the site of action of heparin and warfarin?
|
- Heparin: blood
- Warfarin: liver |
|
|
What is the relative onset of action of heparin and warfarin?
|
- Heparin: rapid (seconds)
- Warfarin: slow, limited by half-lives of normal clotting factors |
|
|
What is the mechanism of action of heparin and warfarin?
|
- Heparin: activates antithrombin, which ↓ the action of IIa (thrombin) and factor Xa
- Warfarin: impairs the synthesis of vitamin K-dependent clotting factors II, VII, IX, and X (vitamin K antagonist) |
|
|
What is the duration of action of heparin and warfarin?
|
- Heparin: acute (hours)
- Warfarin: chronic (days) |
|
|
Do heparin and warfarin inhibit coagulation in vitro?
|
- Heparin: yes
- Warfarin: no |
|
|
How do you treat an acute overdose of heparin and warfarin?
|
- Heparin: protamine sulfate
- Warfarin: IV vitamin K and fresh frozen plasma |
|
|
What should you monitor in patients taking heparin and warfarin?
|
- Heparin: PTT (intrinsic pathway)
- Warfarin: PT / INR (extrinsic pathway) |
|
|
Do heparin and warfarin cross the placenta?
|
- Heparin: no
- Warfarin: yes (teratogenic) |
|
|
What are the types of thrombolytics?
|
- Alteplase (tPA)
- Reteplase (rPA) - Tenecteplase (TNK-tPA) |
|
|
What is the mechanism of Alteplase (tPA), Reteplase (rPA), and Tenecteplase (TNK-tPA)?
|
Thrombolytics
- Directly or indirectly aids conversion of plasminogen to plasmin - Plasmin cleaves thrombin and fibrin clots - ↑ PT, ↑ PTT, and no change in platelet count |
|
|
What are the clinical uses of Alteplase (tPA), Reteplase (rPA), and Tenecteplase (TNK-tPA)?
|
- Early MI
- Early ischemic stroke - Direct thrombolysis of severe PE |
|
|
What are the side effects and contraindications of Alteplase (tPA), Reteplase (rPA), and Tenecteplase (TNK-tPA)?
|
- Bleeding
- Contraindicated in patients with active bleeding, history of intracranial bleeding, recent surgery, known bleeding diatheses, or severe hypertension |
|
|
How do you treat toxicity of Alteplase (tPA), Reteplase (rPA), and Tenecteplase (TNK-tPA)? Mechanism?
|
- Aminocaproic Acid - inhibits fibrinolysis
- Fresh frozen plasma and cryoprecipitate can also be used to correct factor deficiencies |
|
|
What is the mechanism of aspirin (ASA)?
|
- Irreversibly inhibits cyclooxygenase (both COX-1 and COX-2) enzyme by covalent acetylation
- Platelets cannot synthesize new enzyme so effect lasts until new platelets are produced |
|
|
What lab values change in patients taking aspirin (ASA)?
|
- ↑ Bleeding time, ↓ TXA2 and prostaglandins
- No effect on PT or PTT |
|
|
What are the clinical uses of aspirin (ASA)?
|
- Antipyretic
- Analgesic - Anti-inflammatory - Antiplatelet (↓ aggregation) |
|
|
What are the toxic side effects of aspirin (ASA)?
|
- Gastric ulceration
- Tinnitus (CN VIII) - Chronic use can lead to acute renal failure, interstitial nephritis, and upper GI bleeding - Reye syndrome in children with viral infection |
|
|
What can an overdose of aspirin (ASA) cause?
|
Respiratory alkalosis initially, which is then superimposed by metabolic acidosis
|
|
|
What is the cause of Reye syndrome?
|
Children with a viral infection who take aspirin
|
|
|
What are the types of ADP receptor inhibitors?
|
- Clopidogrel
- Ticlopidine - Prasugrel - Ticagrelor |
|
|
What is the mechanism of Clopidogrel, Ticlopidine, Prasugrel, and Ticagrelor?
|
- Inhibit platelet aggregation by irreversibly blocking ADP receptors
- Inhibits fibrinogen binding by preventing glycoprotein IIb/IIIa from binding to fibrinogen |
|
|
What are the clinical uses of Clopidogrel, Ticlopidine, Prasugrel, and Ticagrelor?
|
- Acute coronary syndrome
- Coronary stenting - ↓ Incidence or recurrence of thrombotic stroke |
|
|
What are the side effects of Clopidogrel, Ticlopidine, Prasugrel, and Ticagrelor?
|
- Neutropenia (ticlopidine)
- TTP / HUS may be seen |
|
|
What drugs inhibit platelet aggregation via inhibition of phosphodiesterase III?
|
Cilostazol and Dipyridamole
|
|
|
What is the mechanism of Cilostazol and Dipyridamole?
|
- Phosphodiesterase III inhibitor
- ↑ cAMP in platelets, thus inhibiting platelet aggregation - Vasodilates |
|
|
What are the uses of Cilostazol and Dipyridamole?
|
- Intermittent claudication
- Coronary vasodilation - Prevention of stroke or TIAs (combined with aspirin) - Angina pectoris |
|
|
What are the side effects of Cilostazol and Dipyridamole?
|
- Nausea
- Headache - Facial flushing - Hypotension - Abdominal pain |
|
|
What are the types of GpIIb/IIIa inhibitors?
|
- Abciximab
- Eptifibatide - Tirofiban |
|
|
What is the mechanism of Abciximab, Eptifibatide, and Tirofiban?
|
- Binds to the glycoprotein receptor IIb/IIIa on activated platelets, preventing aggregation
- Abciximab is made from monoclonal antibody Fab fragments |
|
|
What are the uses of Abciximab, Eptifibatide, and Tirofiban (GpIIb/IIIa inhibitors)?
|
- Unstable angina
- Percutaneous transluminal coronary angioplasty |
|
|
What are the side effects of Abciximab, Eptifibatide, and Tirofiban (GpIIb/IIIa inhibitors)?
|
- Bleeding
- Thrombocytopenia |
|
|
What are the drugs that interact with the cell cycle? Which part?
|
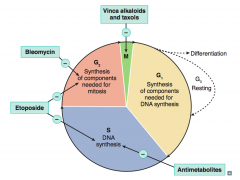
- Antimetabolites (affect S)
- Etoposide (affects S and G2) - Bleomycin (affects G2) - Vinca alkaloids and taxols (affects M) |
|
|
What anti-neoplastic drugs affect nucleotide synthesis? How?
|
- Methotrexate and 5-Fluorouracil: ↓ thymidine synthesis
- 6-Mercaptopurine: ↓ purine synthesis |
|
|
What anti-neoplastic drugs inhibit DNA synthesis? How?
|
- Alkylating agents, Cisplatin: cross-link DNA
- Dactinomycin, Doxorubicin: intercalates into DNA - Etoposide: inhibits topoisomerase II |
|
|
What anti-neoplastic drugs inhibit cellular division? How?
|
- Vinca alkaloids: inhibit microtubule formation
- Paclitaxel: inhibits microtubule disassembly |
|
|
What are the anti-metabolite drugs?
|
- Methotrexate (MTX)
- 5-Fluorouracil (5-FU) - Cytarabine (Arabinofuranosyl Cytidine) - Azathioprine, 6-Mercaptopurine (6-MP), 6-Thioguanine (6-TG) |
|
|
What is the mechanism of Methotrexate (MTX)?
|
Folic acid analog that inhibits Dihydrofolate Reductase → ↓ dTMP → ↓ DNA and ↓ protein synthesis
|
|
|
What are the clinical uses of Methotrexate (MTX)?
|
- Cancers: leukemias, lymphomas, choriocarcinoma, sarcomas
- Non-neoplastic: abortion, ectopic pregnancy, rheumatoid arthritis, psoriasis, IBD |
|
|
What are the toxic side effects of Methotrexate (MTX)?
|
- Myelosuppression
- Macrovesicular fatty change in liver - Mucositis - Teratogenic |
|
|
How can you reverse the myelosuppression induced by Methotrexate (MTX)?
|
Reversible with leucovorin (folinic acid) "rescue"
|
|
|
What is the mechanism of 5-Flurouracil (5-FU)?
|
- Pyrimidine analog bioactivated to 5F-dUMP, which covalently complexes folic acid
- This complex inhibits thymidylate synthase → ↓ dTMP → ↓ DNA and ↓ protein synthesis |
|
|
What is 5-fluorouracil (5-FU) used for?
|
- Colon cancer
- Pancreatic cancer - Basal cell carcinoma (topical) |
|
|
What are the toxic side effects of 5-fluorouracil (5-FU)?
|
- Myelosuppression
- Photosensitivity |
|
|
How can you reverse the myelosuppression induced by 5-Fluorouracil (5-FU)?
|
Reversible with uridine "rescue"
|
|
|
What is the mechanism of Cytarabine (Arabinofuranosyl Cytidine)?
|
Pyrimidine analog → inhibition of DNA polymerase
|
|
|
What is Cytarabine (Arabinofuranosyl Cytidine) used for?
|
- Leukemias
- Lymphomas |
|
|
What are the toxic side effects of Cytarabine (Arabinofuranosyl Cytidine)?
|
- Leukopenia
- Thrombocytopenia - Megaloblastic anemia - CYTarabine causes panCYTopenia |
|
|
What is the mechanism of Azathioprine, 6-Mercaptopurine (6-MP), and 6-Thioguanine (6-TG)?
|
- Purine (thiol) analogs → ↓ de novo purine synthesis
- Activated by HGPRT |
|
|
What are the clinical uses of Azathioprine, 6-Mercaptopurine (6-MP), and 6-Thioguanine (6-TG)?
|
- Preventing organ rejection
- Rheumatoid Arthritis - Azathioprine: Systemic Lupus Erythematosus (SLE) - 6-MP and 6-TG: Leukemia and Inflammatory Bowel Disease |
|
|
What are the toxic side effects of Azathioprine, 6-Mercaptopurine (6-MP), and 6-Thioguanine (6-TG)?
|
- Bone marrow, GI, and liver toxicity
- Azathioprine and 6-MP are metabolized by xanthine oxidase; thus both have ↑ toxicity with allopurinol, which inhibits their metabolism |
|
|
What are the anti-tumor antibiotics?
|
- Dactinomycin (Actinomycin D)
- Doxorubicin (Adriamycin), Danorubicin - Bleomycin |
|
|
What is the mechanism of Dactinomycin (Actinomycin D)?
|
Intercalates in DNA
|
|
|
What are the clinical uses of Dactinomycin (Actinomycin D)?
|
- Wilms tumor
- Ewing sarcoma - Rhabdomyosarcoma - Used for childhood tumors ("children act out") |
|
|
What are the toxic side effects of Dactinomycin (Actinomycin D)?
|
Myelosuppression
|
|
|
What is the mechanism of Doxorubicin (Adriamycin) and Daunorubicin?
|
- Generates free radicals
- Intercalates in DNA → breaks in DNA → ↓ replication |
|
|
What are the clinical uses of Doxorubicin (Adriamycin) and Daunorubicin?
|
- Solid tumors
- Leukemias - Lymphomas |
|
|
What are the toxic side effects of Doxorubicin (Adriamycin) and Daunorubicin?
|
- Cardiotoxicity (dilated cardiomyopathy)
- Myelosuppression - Alopecia - Toxic to tissues following extravasation - Dexrazoxane (iron chelating agent), used to prevent cardiotoxicity |
|
|
What is the mechanism of Bleomycin?
|
Induces free radical formation, which causes breaks in DNA strands
|
|
|
What are the clinical uses of Bleomycin?
|
- Testicular cancer
- Hodgkin lymphoma |
|
|
What are the toxic side effects of Bleomycin?
|
- Pulmonary fibrosis
- Skin changes - Mucositis - Minimal myelosuppression |
|
|
What are the alkylating agent drugs?
|
- Cyclophosphamide, Ifosfamide
- Nitrosoureas (Carmustine, Lomustine, Semustine, Streptozocin) - Busulfan |
|
|
What is the mechanism of Cyclophosphamide and Ifosfamide?
|
- Covalently X-link (interstrand)
- DNA at guanine N-7 - Requires bioactivation by liver |
|
|
What are the clinical uses of Cyclophosphamide and Ifosfamide?
|
- Solid tumors
- Leukemia - Lymphomas - Some brain cancers |
|
|
What are the toxic side effects of Cyclophosphamide and Ifosfamide?
|
- Myelosuppression
- Hemorrhagic cystitis |
|
|
How can you limit the hemorrhagic cystitis caused by Cyclophosphamide and Ifosfamide? Mechanism?
|
Mesna - thiol group of mesna binds toxic metabolites
|
|
|
What is the mechanism of Nitrosoureas (Carmustine, Lomustine, Semustine, and Streptozocin)?
|
- Requires bioactivation
- Crosses blood-brain-barrier → CNS - Cross-links DNA |
|
|
What are the clinical uses of Nitrosoureas (Carmustine, Lomustine, Semustine, and Streptozocin)?
|
Brain tumors (including glioblastoma multiforme)
|
|
|
What are the toxic side effects of Nitrosoureas (Carmustine, Lomustine, Semustine, and Streptozocin)?
|
CNS toxicity (convulsions, dizziness, ataxia)
|
|
|
What is the mechanism of Busulfan?
|
Cross-links DNA
|
|
|
What are the clinical uses of Busulfan?
|
- CML
- Also used to ablate patient's bone marrow before bone marrow transplantation |
|
|
What are the toxic side effects of Nitrosoureas (Carmustine, Lomustine, Semustine, and Streptozocin)?
|
- Severe myelosuppression (in almost all cases)
- Pulmonary fibrosis - Hyperpigmentation |
|
|
What are the types of microtubule inhibitors?
|
- Vincristine and Vinblastine
- Paclitaxel and other Taxols |
|
|
What is the mechanism of Vincristine and Vinblastine?
|
- Vinca alkaloids that bind β-tubulin
- Inhibits its polymerization into microtubules - Prevents mitotic spindle formation (M-phase arrest) |
|
|
What are the clinical uses of Vincristine and Vinblastine?
|
- Solid tumors
- Leukemias - Lymphomas |
|
|
What are the toxic side effects of Vincristine and Vinblastine?
|
- Vincristine: neurotoxicity (areflexia, peripheral neuritis), paralytic ileus
- Vinblastine: blasts bone marrow (myelosuppression) |
|
|
What is the mechanism of Paclitaxel and other Taxols?
|
- Hyperstabilize polymerized microtubules in M phase so that mitotic spindle cannot break down (anaphase cannot occur)
- It is taxing to stay polymerized |
|
|
What are the clinical uses of Paclitaxel and other Taxols?
|
Ovarian and breast carcinomas
|
|
|
What are the toxic side effects of Paclitaxel and other Taxols?
|
- Myelosuppression
- Alopecia - Hypersensitivity |
|
|
What is the mechanism and clinical uses of Cisplatin and Carboplatin?
|
- Cross-links DNA
- Used for testicular, bladder, ovary, and lung carcinomas |
|
|
What are the toxic side effects of Cisplatin and Carboplatin?
|
- Nephrotoxicity
- Acoustic nerve damage - Prevents nephrotoxicity with amifostine (free radical scavenger) and chloride diuresis |
|
|
What is the mechanism and clinical uses of Etoposide and Teniposide?
|
- Inhibits topoisomerase II → ↑ DNA degradation
- Used for solid tumors (particularly testicular and small cell lung cancer), leukemias, and lymphomas |
|
|
What are the toxic side effects of Etoposide and Teniposide?
|
- Myelosuppression
- GI irritation - Alopecia |
|
|
What is the mechanism and clinical uses of Irinotecan and Topotecan?
|
- Inhibits topoisomerase I and prevents DNA unwinding and replication
- Used for colon cancer (irinotecan) - Used for ovarian and small cell lung cancers (topotecan) |
|
|
What are the toxic side effects of Irinotecan and Topotecan?
|
- Severe myelosuppression
- Diarrhea |
|
|
What is the mechanism and clinical uses of Hydroxyurea?
|
- Inhibits ribonucleotide reductase → ↓ DNA synthesis (S-phase specific)
- Used for melanoma, CML, sickle cell disease (↑ HbF) |
|
|
What are the toxic side effects of Hydroxyurea?
|
- Bone marrow suppression
- GI upset |
|
|
What is the mechanism of Prednisone and Prednisolone?
|
- May trigger apoptosis
- May even work on non-dividing cells |
|
|
What are the clinical uses of Prednisone and Prednisolone?
|
- Most commonly used glucocorticoids in cancer chemotherapy
- Used in CLL, non-Hodgkin lymphomas (part of combination chemotherapy regimen) - Also used as immunosuppressants (eg, autoimmune disease) |
|
|
What are the toxic side effects of Prednisone and Prednisolone?
|
Cushing like symptoms:
- Weight gain - Central obesity - Muscle breakdown - Cataracts - Acne - Osteoporosis - Hypertension - Peptic ulcers - Hyperglycemia - Psychosis |
|
|
What is the mechanism of Tamoxifen and Raloxifene?
|
- Selective estrogen receptor modulator (SERMs)
- Receptor antagonists in breast and agonists in bone - Blocks the binding of estrogen to ER (+) cells |
|
|
What are the clinical uses of Tamoxifen and Raloxifene?
|
- Breast cancer treatment (tamoxifen only) and prevention
- Raloxifene also useful to prevents osteoporosis |
|
|
What are the toxic side effects of Tamoxifen and Raloxifene?
|
- Tamoxifen: partial agonist in endometrium, which ↑ the risk of endometrial cancer, "hot flashes"
- Raloxifene: no ↑ in endometrial carcinoma because it is an endometrial antagonist |
|
|
What is the mechanism of Trastuzumab (Herceptin)?
|
- Monoclonal antibody against HER-2 (c-erbB2), a tyrosine kinase receptor
- Helps kill breast cancer cells that overexpress HER-2, through inhibition of HER2-initiated cellular signaling and antibody-dependent cytotoxicity |
|
|
What are the clinical uses of Trastuzumab (Herceptin)?
|
HER-2 positive breast cancer and gastric cancer (Tras2zumab)
|
|
|
What are the toxic side effects of Trastuzumab (Herceptin)?
|
- Cardiotoxicity ("HEART"ceptin damages the heart)
|
|
|
What is the mechanism of Imatinib (Gleevec)?
|
Tyrosine kinase inhibitor of bcr-abl (Philadelphia chromosome fusion gene in CML) and c-Kit (common in GI stromal tumors)
|
|
|
What are the clinical uses of Imatinib (Gleevec)?
|
- CML
- GI stromal tumors |
|
|
What are the toxic side effects of Imatinib (Gleevec)?
|
Fluid retention
|
|
|
What is the mechanism of Rituximab?
|
Monoclonal Ab against CD20, which is found on most B-cell neoplasms
|
|
|
What are the clinical uses of Rituximab?
|
- Non-Hodgkin Lymphoma (CD20 positive cells)
- Rheumatoid arthritis (with MTX) - Idiopathic Thrombocytopenic Purpura (ITP) |
|
|
What are the toxic side effects of Rituximab?
|
Increased risk of progressive multifocal leukoencephalopathy
|
|
|
What is the mechanism and use of Vemurafenib?
|
- Small molecule inhibitor of forms of the B-Raf kinase with the V600E mutation
- Used for metastatic melanoma |
|
|
What is the mechanism and clinical uses of Bevacizumab?
|
- Monoclonal antibody against VEGF, inhibits angiogenesis
- Used for solid tumors (colorectal cancer, renal cell carcinoma) |
|
|
What are the toxic side effects of Bevacizumab?
|
Hemorrhage and impaired wound healing
|
|
|
Which chemo drug(s) cause(s) acoustic nerve damage?
|
Cisplatin / Carboplatin
(and nephrotoxicity) |
|
|
Which chemo drug(s) cause(s) peripheral neuropathy?
|
Vincristine
|
|
|
Which chemo drug(s) cause(s) pulmonary fibrosis?
|
- Bleomycin
- Busulfan |
|
|
Which chemo drug(s) cause(s) cardiotoxicity?
|
- Doxorubicin
- Trastuzumab |
|
|
Which chemo drug(s) cause(s) nephrotoxicity?
|
Cisplatin and Carbolatin (and acoustic nerve damage)
|
|
|
Which chemo drug(s) cause(s) hemorrhagic cystitis?
|
Cyclophosphamide
|
|
|
Which chemo drug(s) cause(s) myelosuppression?
|
- 5-FU
- 6-MP - Methotrexate |

