![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
76 Cards in this Set
- Front
- Back
|
What are some functions for lipids?
|
Nutrition (10-40% calories intake), metabolic energy (60-80% energy production), membrane and lipoprotein structure and function, cell recognition and interaction with envt, biologically active, polysaccharide carriers, covalent mod
|
|
|
What is the basic building blocks of most lipids?
|
Fatty acids
R group attached to carboxyl acid (deprotonated at physiological pH) R-CO2- |
|
|
Saturated fatty acids
|
Fully reduced fatty acids
Can be short (2-6 C), long, (C14+) or medium chain (C8-C12) |
|
|
Acetic Acid
|
CH2COOH
Short Chain |
|
|
Propionic / Propanoic acid
|
CH3CH2COOH
Short Chain |
|
|
Butyric/Butanoic Acid
|
CH3-CH2-CH2-COOH
Short chain Unpleasant odor, it's a sign of bacterial activity |
|
|
Lauric (Dodecanoic) Acid
|
12 carbon medium chain
|
|
|
Palmitic (Hexadecanoic) Acid
|
16 carbon long chain
Most common long chain |
|
|
Stearic (octadecanoic) Acid
|
18 carbon long chain
2nd most common long chain |
|
|
Diets high in saturated fat associated with what?
|
Elevated LDL, Lower HDL, increased risk of atherosclerosis and coronary artery disease
C12-C16 chains have most effect. |
|
|
Oleic Acid
|

18:1delta9
18:1omega9 |
|
|
Linoleic Acid
|

18:2d9,12
18:2o6 |
|
|
Linolenic acid
|
18:3d9,12,15
18:3o3 |
|
|
Eicosatrienoic acid
|
20:3d8,11,14
20:3o6 PGE1 (prostaglandin) precursos |
|
|
Arachidonic Acid
|
Eicosatetraenoic Acid
20:4d5,8,11,14 20:4o6 PGE2 precursor |
|
|
Eicosapentaenoic Acid
|
EPA!
20:5d5,8,11,14,17 20:5o3 PGE3 precursor |
|
|
Docosahexaenoic Acid
|
DHA
22:6d4,7,10,13,16,19 22:6o3 |
|
|
Short hand nomenclature of fatty acids
|
delta - double double position from carboxyl end
omega - double bond positioned from the methyl end |
|
|
Essential fatty acids
|
Linolenic and Linoleic Acids
Precursors for all o6 and o3 fatty acids |
|
|
Resolvins
|
compounds that are made by the human body from the omega-3 fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA)
Probably anti-inflammatory |
|
|
Essential fatty acid deficiency
|
Symptoms reflect abnormal membrane properties and lack of precursors for synthesis of prostaglandins
Infants particularly susceptible |
|
|
What does substitution of saturated by cis-unsaturated fatty acids do? o6? o3?
|
Lowers serum cholesterol
o6 lowers both LDL and HDL 03 lowers only LDL and raises HDL |
|
|
Trans fatty acids
|
Unsaturated fatty acids whose double bonds are trans. Common in fats and oils that have been industrially processed.
More similar biologically to sat'd fatty acids. Exception are conjugated Linoleic acid (in stomachs of ruminants) |
|
|
What are high intakes of trans fats associated with?
|
Decreased HDL and increased LDL
Increased risk of atherosclerosis/heart disease |
|
|
How does chain length and saturation affect fatty acid melting point?
|
Lowers melting point: shorter chains, more double bonds/unsaturation (esp cis, trans has less effect)
Raises melting point: Longer chains, more saturation |
|
|
How does chain length and saturation affect water solubility?
|
Longer chains mean less solubility
Ionized fatty acids form aggregate structures like micells and multilamellar forms |
|
|
Glycerol Structure
|

|
|
|
Triacylglycerol / Triglyceride Basic structure
|

Commonly called "fats" if solid at room temp, "oils" if liquid.
|
|
|
Physiological significance of di/triacylglycerols
|
1) Triacylglycerols: Major source of mammalian (and plant) energy storage
2) Diacylglycerols: impt intracellular second messenger activating protein kinase C |
|
|
Benefits of consuming/producing hydrogenated oils
|
1) Cultural factors (some ppl prefer butter to oil)
2) Desirable physical props 3) Chemical stability because they're less susceptible to oxidation |
|
|
Cons of consuming/producing hydrogenated oils
|
1) Increased saturated fat
2) Alters biological activity 3) Trans form preferentially occurs because it's more stable (Trans is bad!) |
|
|
Phosphoglycerides
|

Have phosphate at one position
Named as derivates of phosphatidic acid (e.g., phosphatidyl-X where X = choline, inositol, etc.) |
|
|
Reactions fatty acids go through at carboxyl group
|
1) Esterification
2) Amidation 3) Hydrolyis |
|
|
Reactions fatty acids go through at double bond
|
1) Hydrogenation (reduction)
2) Auto-oxidation |
|
|
What is saponification?
|
Triacylglycerols + H20 --> glycerol + salts (-O-CR=O)
In the presence of a base (or lipase) Eg. lye soap example |
|
|
Catalytic hydrogenation
|
Uses a heavy metal and H2 in industrial processing of dietary fats and oils. Hydrogenation may be partial or complete. In partial hydrogenation, large portion of remaining double bonds may be isomerized to trans
|
|
|
Auto-oxidation
|
In the presence of oxygen, unsaturated fatty acids can form free radicals, implicated in etiology and pathology of many diseases (aging, atherosclerosis, diabetes, and cancer).
In the cell it's controlled by natural antioxidants and by enzyme catalyzed reactions. The more double bonds there are, the more auto-oxidation that occurs. Unsaturated fats are less stable than saturated. It becomes important in the context of atherosclerosis because when this occurs, LDLs are taken up by macrophages indiscriminately b/c they're recognized as exogenous compounds (ie., like bacteria). The macrophages fill up with them and it kills them. The dead macrophages deposit themselves into arterial walls, eventually causing plaques. |
|
|
Nomenclature for modified carboxyl group in lipids
|
1) Protonated acid: StearIC
2) Deprotonated/ionized acid: StearATE 3) Derivatized fatty acid: X-stearate (e.g., methyl palmitate) 4) Another compound derivatized with fatty acid: StearOYL-X |
|
|
Phosphatidylethanolamine
|
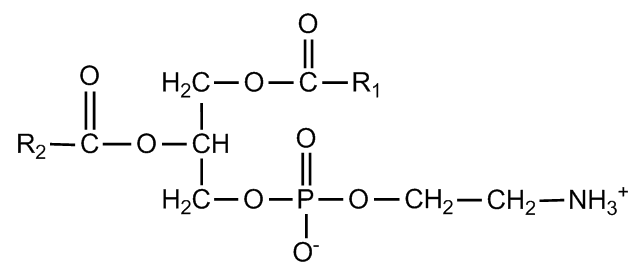
|
|
|
Phosphatidylcholine
|

|
|
|
Phosphatidylinositol
|

|
|
|
Significance of phosphoglyceride
|
main component of biological membranes.
|
|
|
Plasmalogen
|

an ether lipid where the first position of glycerol binds a vinyl residue (from a vinyl alcohol) with the double bond next to the ether bond.
Glyceryl ethers, named as phosphatidal-X As much as 60% of some plasma membranes. Example: |
|
|
phospholipases: defn and where do they cleave?
|
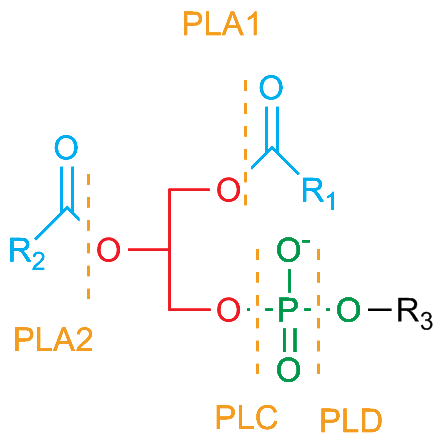
Enzymes that hydrolyze phophoglycerides at specific locations. Involved in digestion of phospholipids, membrane remodeling, active metabolites
Notice it leaves the glycerol skeleton with the Os |
|
|
Sphingolipids: basic structure
|
Derivatives of Sphingosine
<img src = "http://upload.wikimedia.org/wikipedia/commons/a/a7/Sphingolipid.png"> |
|
|
What do sphingolipids do?
|
play important roles in signal transmission and cell recognition
|
|
|
Sphingomyelin
|

type of sphingolipid found in animal cell membranes, especially in the membranous myelin sheath that surrounds some nerve cell axons. It usually consists of phosphorylcholine and ceramide. In humans SPH represents ~85% of all sphingolipids.
Resembles phosphatidylcholine |
|
|
Gangliosides
|
Important membrane constituents which are responsible for both general (neg charge) and specific characteristics of the cell surface. Involved in receptor-ligand interaction. Composed of many modified sugar residues connected by glycosidic bonds.
<img src = "http://www.cyberlipid.org/images/pict100.gif"> |
|
|
Sphingolipidoses
|
Disorders resulting from mutations in specific lysosomal enzymes which hydrolyze specific bonds during normal turnover of gangliosides or sphingomyelin. Substrates then accumulate in organs. Eg. Gaucher's disease
|
|
|
Structure of cholesterol
|
<img src ="http://www.coe.drexel.edu/ret/personalsites/2004/murerian/web/images/cholesterol.JPG">
|
|
|
Physiological significance of cholesterol
|
<b>Steroid hormones are synthesized from cholesterol.</b>
Bile acids are major form in which it's removed from body. Excess cholesterol is esterified to fatty acid. Cholesterol ester is main constituent of atherosclerotic plaques. Unesterified cholesterol is <b> important constituent of cell membranes </b>, including modifying membrane properties. <b> Elevated blood cholesterol is a major risk factor for atherosclerosis and coronary artery disease </b> |
|
|
Fat Soluble Vitamins
|
A, D, E, K
|
|
|
Vitamin A
|
Retinol
Important constituent of visual system as well as molecules involved in growth regulation. Found in liver. Can be poisonous if too much is eaten. Fat Soluble. |
|
|
Vitamin D
|
Important to calcium and phosphorous absorption and transport. Exposure to sun causes synthesis.
|
|
|
Vitamin E
|
Important natural antioxidant/free radical scavenger.
|
|
|
Vitamin K
|
Cofactor in synthesis of important components of the blood clotting cascade. Found in leafy green veggies.
|
|
|
Where do fatty acids come from?
|
Dietary lipids, intracellular stores (adipose tissue), Circulating serum lipoproteins
|
|
|
Lipases
|
Hydrolyzes glyercol esters to release fatty acids from storage during<b> lipolysis </b>.
|
|
|
Pancreatic Lipase
|
Hydrolyzes dietary triglycerides in intestinal lumen during digestion
|
|
|
Lipoprotein Lipase
|
Hydrolyzes triglycerides being transported by chlyomicrons and VLDLs in blood
Activated by insulin, signal for fed state |
|
|
Hormone sensitive lipase
|
Hydrolyzes triglycerides store in adipose tissue; activated by glucagon or epinephrine.
Inhibited by insulin |
|
|
Fatty acid transport: how and why?
|
WHY: Unesterified fatty acids are detergent-like poisons to the body, so must be transported bound to a protein or in esterified state.
HOW: Bound to albumin, lipoproteins, specific transport systems (eg., carnitine shuttle) |
|
|
Fatty acyl CoA synthetase and reaction
|
Makes CoA esters from fatty acyls. First step of Beta oxidation
<img src = "http://www.tc.umn.edu/~zhan0260/boxidation_files/image003.gif"> |
|
|
pantothenic acid
|
a vitamin/essential nutrient required for production of CoA.
|
|
|
pyrophosphatase
|
hydrolyzes a pyrophosphate PPi group
|
|
|
Carnitine shuttle
|
Transport of <b> long chain </b> fatty acyl CoA molecules from cytosol to mitochondrial matrix.
1) Carnitine acyltransferase I exchanges carnitine for CoA to generate fatty acylcarnitine 2) Transporter shuttles acylcarnitine across inner membrane and carnitine in the other direction. 3) Carnitine acyltransferase II exchanges CoA from an internal pool for carnitine to regenerate fatty acyl-CoA in the mitochondrial matrix. <img src = "http://upload.wikimedia.org/wikipedia/commons/5/50/Acyl-CoA_from_cytosol_to_the_mitochondrial_matrix.gif"> |
|
|
Regulation of carnitine shuttle
|
CATI inhibited by malonyl-CoA, an intermediate in fatty acid biosynthesis, prevent futile cycling between β-oxidation and fatty acid synthesis.
Fatty acids in high concentration up-regulate CATI at a transcriptional level. |
|
|
What kinds of fatty acids don't use the carnitine shuttle and why?
|
Medium chain fatty acids. They can get across the mitochondrial membranes without getting made into CoA derivatives.
|
|
|
Carnitine
|
Carries fatty acids via shuttle system across mitochondrial inner membrane, esterified to its hydroxyl group.
|
|
|
Where does Beta oxidation happen?
|
Mitrochondrial matrix
|
|
|
Fatty acyl CoA dehydrogenase
|
Chain length specific. Product FADH2 from each round donates its reducing equivalents to the electron transport chain, with potential to generate 1.5 ATP via OXPHOS
|
|
|
Three major, general steps in beta oxidation
|
The beta oxidation of fatty acids involve three stages:
1. Activation of fatty acids in the cytosol 2. Transport of fatty acids into mitochondria (carnitine shuttle) 3. Beta oxidation proper in the mitochondrial matrix |
|
|
Beta oxidation: Total
|
<img src = "http://themedicalbiochemistrypage.org/images/betaoxidation.jpg">
|
|
|
Hydratase
|
catalyses 2nd reaction in beta oxidation
|
|
|
Beta OH AcylCoA dehydrogenase
|
Generates 1 NADH
catalyses 3rd reaction in B-oxidation |
|
|
thiolase
|
catalyzes 4th reaction in B-oxidation
Uses a S-CoA group to generate an acetyl CoA, and an acyl CoA that has 2 fewer carbons than when B-oxidation started |

