![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
272 Cards in this Set
- Front
- Back
|
Growth factors |
-are peptides/proteins which stimulate cells to divide or increase in size (hypertrophy), thus regulate cell growth. -Most growth factors have autocrine or paracrine actions; they are synthesized, secreted and |
|
|
Trophic factors |
-are which regulate cell differentiation,, survival, phenotype expression and plasticity as well as growth, which can be growth factors. |
|
|
Neurotrophic factors |
-stimulate the growth of neural cells |
|
|
Somatotrophic factors |
-stimulate the growth of somatic (body) cells. |
|
|
Cytokine growth factors |
-regulate growth and multiplication of |
|
|
Neurotrophic Factors: Nerve Growth Factor (NGF) |
-promotes axon growth and survival of neurons in the SNS and some sensory CNS neurons. |
|
|
Neurotrophin 3 (NT-3) |
-Promotes growth of hippocampal neurons, somatosensory neurons, and cerebellar neurons. |
|
|
Neurotrophin 4/5 (NT-4/5) |
-promotes growth of sensory and motor neurons |
|
|
Neurotrophin 6 (NT-6) |
-Similar to NGF, but less potent; promotes growth of sensory and SNS nerves. |
|
|
Brain Derived Neurotrophic Factor (BDNF) |
-Promotes growth of peripheral sensory neurons and CNS neurons which do not respond to NGF. |
|
|
Ciliary Neurotrophic Factor (CNTF) |
-Promotes growth of SNS, PNS and sensory |
|
|
Glial Cell Line Derived Neurotrophic Factor (GDNF) |
-enhances survival of midbrain dopaminergic neurons |
|
|
Somatotrophic Factors: Epidermal Growth Factor (EGF) |
-Stimulates cell division in epidermal and certain other cells. |
|
|
Fibroblast Growth Factors (FGFs) |
-Stimulates proliferation of fibroblasts, endothelial cells and other cell typesincluding blood vessels, neurons and adrenal cells. Found in the pituitary gland. |
|
|
Platelet-Derived Growth Factor (PDFG) |
-Stimulates proliferation of connective |
|
|
Transforming Growth Factor Beta (TGF-b) |
-Potentiates or inhibits the responses |
|
|
Insulin-like Growth Factors (IGF-I and IGF-II) |
-Somatomedins - stimulate proliferation of fat and connective tissue cells. |
|
|
Cytokine Growth Factors: Interleukin 3 (IL-3)
|
-stimulates stem cell differentiation. |
|
|
Granulocyte Colony Stimulating Factor (G-CSF) |
-Stimulates granulocyte/macrophage progenitor cells and neutrophils |
|
|
Macrophage Colony Stimulating Factor (M-CSF) |
-Stimulates granulocyte/macrophage progenitor cells and macrophages. |
|
|
Granulocyte/Macrophage Colony Stimulating Factor (GM-CSF) |
-stimulates granulocyte/macrophage progenitor cells. |
|
|
Other cell growth stimulating factors |
Neurotransmitters |
|
|
Anabolic |
-construction of molecules from smaller units --building up organs or tissues |
|
|
Catabolic |
-large molecules are broken down into smaller units
|
|
|
Metabolic |
-shaping one molecule into another |
|
|
Effect of NGF on SNS growth |
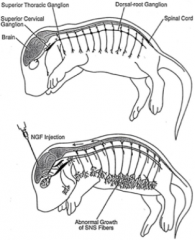
Injection of NGF in the brain causes abnormal growth in the dorsal-root ganglion extending from the spinal cord, in the SNS fibers. |
|
|
More Examples of Growth Factors |
Mast Cell Growth Factor (MGS) |
|
|
Extracellular matrix/adhesion factors |
Nerve Cell Adhesion Molecules (NCAMs) |
|
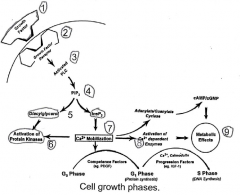
|
The example in Figure 2.4 shows the activation of membrane associated phospholipase C that results in the formation of diacylglycerol (DAG) and inositoltriphosphate (IP3) from phosphatidylinositol-diphosphate (PlP2). These events lead to the activation of protein kinases and calcium mobilization in the cell, resulting in |
|
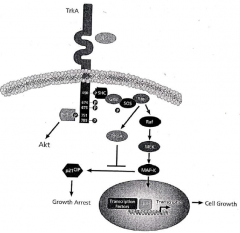
|
Figure 2.2 shows NGF binding to a tyrosine kinase (TrkA) receptor and activating the MEK- MAP-kinase intracellular second messenger system. Epidermal Growth Factor can bind to four different types of tyrosine kinase receptors to activate a number of different intracellular messengers, including PlP2, JAK-STAT, MEKERK and JNK signal transduction pathways, as shown in Figure 2.3. |
|
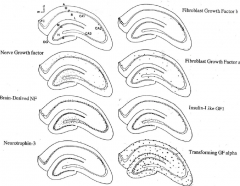
|
Growth factor receptors in the hippocampus. The distribution of growth factor receptors in the hippocampus, as shown in coronal sections on the right side of the rat brain. All distributions are in neurons, with the exception the TGFa precursor, which is localized in glia. The top left panel indicates the regions of the hippocampus, including the dentate gyrus (DG) with its molecular (m), granule cell (G) and hilar cell (H or CA4) layers. Ammon's horn indicates the CA1, CA2, and CA3 regions, each with an alveus (A), stratum oriens (O), pyramidal layer (P), stratum radiatum (R), and stratum lacunosum (L). The fasciola cinerium (FC) is also illustrated. |
|
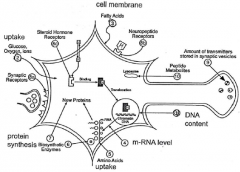
Ten ways that growth factors can activate biochemical changes in cells. |
1) The DNA content of the developing brain gives a measure of cell number, reflecting the difference between cell formation and cell death. 2) The metabolic activity of the cell can be measured by its uptake of glucose, oxygen or other ions. 3) The development of cell membrane can be measured by the level of fatty acids taken up as membrane components. The biosynthetic activity of the cell can be measured by: 4) the level of RNA synthesized; 5) the uptake of amino acids; 6) the increase in specific biosynthetic enzymes; or 7) the increase in protein synthesis. The readiness of the nerve cell for functional activity can be determined by measuring: 8a) the number of synaptic receptors for neurotransmitters; 8b) the number of receptors for neuropeptides and non-steroid hormones, or 8c) the number of steroid hormone receptors. Likewise, the amount of neurotransmitter stored in the vesicles 9) and the level of transmitter and other hormone and neuropeptide metabolites 10) in the cell can be used as measures of the functional readiness of the nerve cell |
|
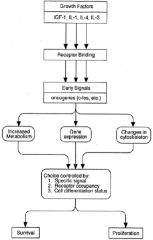
|
How growth factors stimulate cell survival and proliferation. Insulin-like growth factor-1 (IGF-1) and interleukin-4 (IL-4) promote cell survival by binding to their receptors, activating second messenger systems which stimulate the production of early onset oncogenes (c-fos, c-myc, c-jun). These oncogenes then stimulate increased metabolism, the expression of genes which stimulate protein synthesis, or changes to the cytoskeleton. Depending on the specific growth factor, its receptor and differentiation status of the cell, the growth factors may stimulate cell survival or proliferation. For example, growth factors may promote survival of immature cells and proliferation of mature cells. |
|
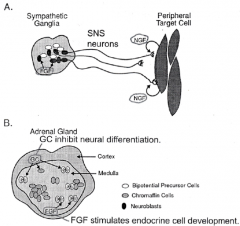
|
The role of growth factors in cell differentiation. The bipotential precursor cells of the neural crest are plastic in their development and may differentiate into neural or endocrine cells. A) In the sympathetic nerve ganglia, fibroblast growth factor (FGF) induces the development of neurons whose axonal projections extend toward peripheral target cells. The release of nerve growth factor from these target cells promotes synapse formation and the survival and maturation of these neurons. B) In the adrenal cortex, glucocorticoids (GC) inhibit the neural differentiation of the progenitor cells, which are then stimulated by GFG to develop into mature endocrine gland cells. |
|
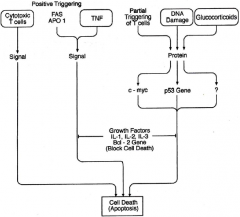
|
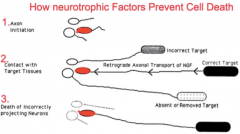
Mechanisms for promoting cell death (apoptosis). There are a number of mechanisms for deleting cells. Cytotoxic T cells, Tumor Necrosis Factor (TNF) and cell surface antigens (Fas, APO 1) can cause cell death directly. Glucocorticoids may stimulate apoptosis by activating genes which trigger cell death. Likewise, T cell receptor stimulation and agents which cause damage to the cellular DNA can stimulate apoptosis by activating cell death genes. The tumor suppressor gene (p53) and the proto-oncogene c-myc, whic regulates cell growth, may both be involved in triggering apoptosis when activated by external modulators such as glucocorticoids. BUT, activation of the "survival" oncogene bcl-2, results in the expression of proteins which may inhibit apoptosis and facilitate cell survival. Certain growth factors, such as interleukins IL-1, IL-2, and IL-3 may also inhibit apoptosis in their target cells. |
|
|

|
|
|
Apoptosis:
|
-Programmed cell death -Deleting unwanted cells (pruning) -Can be triggered by cytotoxic T cells and tumor necrosis factor (TNF) -DNA damage and excessive glucocorticoid hormone stimulation - cell death genes (p53)
Growth factors: -block cell death by blocking actions of p53 -cell survival genes BCL-2 -IL-1, 2, and 3 may also inhibit apoptosis
|
|
|
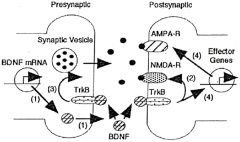
A model for the role of BDNF in learning and memory. 1) Neuronal activity increases BDNF gene expression and stimulates BDNF release from presynaptic sites in an activity-dependent manner. BDNF binds to TrkB receptors located on presynaptic and postsynaptic sites, leading to the activation of signal transduction pathways including MAPK and PI3-K. 2) Activation of BDNF/TrkB signaling causes phosphorylation of NMDA receptors. 3) Increase in neurotransmitter release from presynaptic sites, and 4) enhancement of protein synthesis. In addition, BDNP-induced modulation of growth and complexity of dendrites may participate in long-term memory. |
|
|
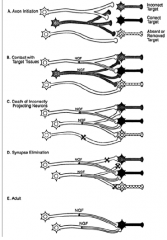
How neurotrophic factors prevent cell death |
|
|
|
Internal genitals of males or females |
Males: vas deferens Females: Fallopian tubes, uterus |
|
|
Hormonal gender -type of hormone secreted by gonads |
Male: Mainly androgens Female: Mainly estrogens and progestins |
|
|
Accessary sex hormones -depends on hormone |
Male: Prostate gland, seminal vesicles, ejaculatory ducts Female: Skene's gland, Bartholin's gland |
|
|
External genitals |
Male: Penis, scrotum Female: Vagina, clitoris, labia |
|
|
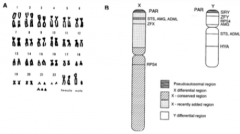
The human karyotype. Humans have 23 pairs of chromosomes for a total of 46 chromosomes; 22 pairs of autosomes and one pair of sex chromosomes. Females have two X sex chromosomes and males have an X and Y chromosome. B. The X and Y chromosomes differ in size and in gene content, although they evolved from a homologous pair of genes. The Y chromosome is much smaller than the X chromosome and the sex chromosomes have very few homologous genes. The shared genes are the STS, AMG, ADMI, ZFX/Ym and RPS4 genes and the genes in the pseudoautosomal region (PAR). The three genes on the Y chromosome which are specific to the male are the HYA, ZFY, and SRY genes. |
|
|
Genotypic sex determination |
-One of two mechanisms that determine whether ovaries or testes develop. -This is the mechanism in which genes on the sex chromosomes determine testes/ovaries determination
|
|
|
Environmental sex determination |
-One of two mechanisms that determine whether ovaries or testes develop. -Mechanism in which environmental factors, such as temperature, determine testes/ovary development -Environmental sex determination or -In fish, some species have genotypic sex determination, some have temperature dependent sex determination and some species which can change from one sex to the other, depending on environmental conditions. Sex change in fish is regulated primarily by size and social factors: the largest females change to males in all female groups or in groups with few males. The "trigger" for sex change may be |
|
|
Sex determination |
The undifferentiated embryonic vertebrate gonad is bipotential: it can develop into |
|
|
Chromosomes in rodents |
There are, however, some species of rodents (eg., wood lemmings, collared lemmings and the Oregon vole) in which females have a Y chromosome and males do not. |
|
|
Chromosomes in birds |
In birds, sex is also determined genetically, with males having ZZ sex chromosomes and females having ZW sex chromosomes. |
|
|
Chromosomes in reptiles/amphibia/fish |
Many reptiles, amphibia and fish also have genotypic sex determination using either the XX/XY or ZW/ZZ sex chromosomes. |
|
|
Regions of the Y chromosome |
In mammals, the Y chromosome is much smaller than the X chromosome and contains three unique gene regions: the male-specific histocompatibility antigen (HYA) genes; the zinc finger (ZFY) gene; and the sex-determining region (SRY) genes. |
|
|
What is needed for masculization to occur? |
The Y chromosome contains the testes-determining factor (TDF) genes which must be present for masculinization to occur (Graves, 1994). Page et al. (1987) claimed that the ZFY gene was the TDF gene, but masculinization can occur in the absence of the ZFY gene. Sinclair et al. (1990) identified the SRY gene, which is now believed todetermine testes development (Sultan, et al., 1991; Graves, 1994; Schafer and Goodfellow, 1996). If a 46XY genotypic "male" is missing the top of the Y chromosome carrying the SRY gene, they develop as an infertile phenotypic 46XY female, with streak gonads. Likewise, if a 46XX genotypic "female" carries the SRY gene on one X chromosome, they develop as a phenotypic male. |
|
|
How many genes are needed for proper sexual differentiation? |
At least 19 genes may be necessary for proper sexual differentiation and these genes are on the autosomes as well as the X and Y chromosomes. For example, three genes on the Y chromosome (WT1, SF1 and SOX9) are necessary for the development of functional testes and at least one gene on the X chromosome (DSS) is necessary for the |
|
|
Barr bodies |
Only one X chromosome is necessary for female development and the second X |
|
|
The undifferentiated gonad is composed of... |
...cells derived from four different types of |
|
|
Development of Sertoli cells |
The SRY gene codes for a protein messenger (as yet unknown) that switches on the development of the Sertoli cells from the supporting cells in the undifferentiated gonad. The developing Sertoli cells stimulate the production of sperm from the germ cells, stimulate the differentiation of the steroid-producing cells into Leydig cells which secrete testosterone, and produce Müllerian inhibiting substance which inhibits the development of female internal genitalia. Without the SRY gene, the supporting cells develop into follicular cells, the germ cells produce oocytes, and the steroid-producing cells become theca cells which produce estrogen. The pathway of testes development from the undifferentiated gonad occurs in a number of steps and Figure 3B shows some of the disorders of testes development that can occur in mutant mice. |
|
|
The testes determining region of the mouse Y chromosome (Tdy)... |
...is homologous with the TDF region of the human Y chromosome and the sex determining gene of the mouse (Sry) is homologous with the human SRY gene. Female mice having the Sry gene on their |
|
|
Disorders of sex determination |
A 46XX female having the SRY gene is |
|
|
Turner's Syndrome |
Turner described 7 females with sexual |
|
|
Multiple X Chromosome |
About 1 in 900 girls (1.1/1000) are born with |
|
|
Klinefelter's Syndrome (47XX) |
Klinefelter et al. (1942) described 9 men with |
|
|
XYY Syndrome |
About 1 in 850 (1.18/1000) boys born have extra Y chromosomes, resulting in a 47XYY or 48XYYY karotype. These males have masculine |
|
|
Genetic true hermaphrodites |
A true hermaphrodite has both ovarian and |
|
|
Sexual differentiation |
Sexual differentiation is the mechanism through which genetically determined sex |
|
|
Differentiation of the internal genital organs |
Before sexual differentiation begins |
|
|
Gonadal hormone secretion |
As the testes develop, they produce two |
|
|
Differentiation of the external genitalia |
The external genitalia begin to differentiate between 2.5 and 5 months of fetal age (Table 6.3). In the male, androgens cause |
|
|
Differentiation of the hypothalamic-pituitary-gonadal feedback system |
From the |
|
|
Sexual differentiation of the brain and spinal cord |
Testosterone acts to |
|
|
Alterations in nerve number |
(a) There are steroid-induced sex differences in neural |
|
|
Steroids influence neurite growth |
(a) Testosterone and estrogen both stimulate neurite |
|
|
Sex steroid influence on neuronal size and form |
(a) Neonatal steroids produce larger RA |
|
|
Sex steroid influence on biochemical processes |
In both neonatal and adult animals, |
|
|
Steroid-induced changes in steroid binding and accumulation in cells |
(a) Neonatal hormones alter the sensitivity of neurons to hormones. (b) Levels of steroid receptors can be influenced by steroids in adulthood. A. By influencing the number of receptor proteins produced in the cells and thus the |
|
|
Freemartin |
Farmers who had twins in an animal, one male one female, had a sterile female. This may be because they're in the same environment and some of the blood mixed, the female is exposed to the same chemical environment that the male is, and the female brain develops more of a male pattern. Although she looks reasonably normal, her ovaries are atropic, and her gonads are not present. |
|
|
Why do 2M males show more aggression? |
2M males show more aggression, due to a higher proportion of estrogen. Males surrounded by two females actually show more sexual behaviour than other types of males. |
|
|
Levels of steroid receptors can be |
....By influening the structure of neural development. This includes (1) modulation of |
|
|
Perinatal manipulation of sexual differentiation |
Manipulation of gonadal hormone levels during the critical period of sexual |
|
|
Castration of males on the first day after birth... |
...results in the brain retaining its female |
|
|
Demasculinization |
Following the time of puberty, males castrated in infancy show less mounting of females (demasculinization). Males castrated at birth show less rough and tumble play in infancy, are less aggressive, scent mark less, show increased activity levels and more parental behaviour than control males. |
|
|
Feminization |
If castrated and given estrogen and progestin injections in adulthood, will show female pattern lordosis when mounted by another male (feminization). Control males show masculine behaviour if castrated in adulthood and given either testosterone or estrogen injections. |
|
|
Masculinized females |
Show more rough and tumble play, are more |
|
|
Neonatal cyproterone acetate |
Neonatal cyproterone acetate administration "feminizes" males, by inhibiting hypothalamic organization. Also, cyproterone acetate prevents penis and accessory sex gland development. This requires huge cyproterone acetate doses over long periods of pre- and post-natal development (10 days prenatal, three weeks postnatal). |
|
|
Females masculinized by receiving testosterone |
If a female has been "masculinized" by receiving testosterone, her clitoris may be enlargd to form a small penis. Male rats given cyproterone acetate and estrogen develop a vagina, show cyclic gonadotrophic secretion system and are treated like females by other males -- yet they are genetically male! |
|
|
When newborn female rats are given testosterone injections... |
...they have small ovaries, no ovulatory cycle and no ovulation (no formation of corpora lutea) i.e. no LH surge. Castrated newborn male rats given ovarian implants as adults show ovulation (formation of a corpora |
|
|
Aromatiziation of testosterone |
Testosterone is converted or aromatized to estradiol by the enzyme aromatase. Thus, in |
|
|
Testosterone can act at its target cells in three ways |
By directly stimulating testosterone receptors; by aromatization to estrogen and stimulation of estrogen receptors or by reduction to DHT and stimulation of DHT receptors. Aromatization and reduction can only occur in cells which contain the appropriate enzymes. Thus, if aromatase or reductase enzymes are lacking, some aspects of masculinization may fail to occur, while other aspects will occur normally. There are a wide variety of disorders of sexual differentiation which occur because of enzyme deficiences. |
|
|
Prevention of female masculinization |
There are three reasons why females are not |
|
|
Alphafetoprotein |
Alphafetoprotein is present in both males and females, but only binds to estrogen, not testosterone, so does not prevent |
|
|
Three examples of sexually dimorphic neural circuits |
There are three examples of sexually dimorphic neural circuits which have been well studied: (1) the sexually dimorphic nucleus of the pre-optic area of the brain of rodents; (2) the spinal nucleus of the bulbocavernosis in rodents, and, (3) the brain regions controlling birdsong. |
|
|
Sexually dimorphic behaviours |
These sex differences in neural organization of |
|
|
What do the brain and neurotransmitter system begin as? |
The brain and neurotransmitter system, like the genitals, begins as "undifferentiated" |
|
|
Masculinization appears to function like a rheostat... |
...you start with a neutral (female) |
|
|
Most sexually dimorphic behaviours do not emerge until after puberty... |
...when they are "activated" by the gonadal hormones in adulthood. These sex differences in behaviour include sexual behaviour (proceptive and receptive); aggression; scent-marking; ultrasonic vocalizations; parental behaviour and activity levels. |
|
|
Adrenogenital syndrome |
There are some people who have an enzyme deficiency in the adrenal cortex, and instead of cortisol being produced, adrenal androgens are synthesized. Progesterone is thus converted to androgens in the adrenal cortex rather than cortisol. Adrenogenital syndrome, or congentital adrenal hyperplasia, has little effect on developing males, who already have high testicular androgen levels, but the high levels of adrenal androgens can partly masculinize females. Thus, a 46 XX genotype with ovaries and fallopian tubes, may have her external genitals fully or partially masculinized, depending on the amount of adrenal androgens produced. In extreme cases, the female is completely masculinized and the gender of rearing is male. In less extreme cases, the genitals can be surgically feminized and the gender of rearing is female. In these less extreme cases, the female may experience a late menarche, but will usually be able to conceive, give birth and lactate (see chapter 18 on gender role and identity). Of course, these females must be given corticosteroid treatment immediately after birth as they produce little or none from their own adrenal cortex. If cortisone therapy is given neonatally, excess androgen production ceases and female puberty and reproductive functions are normal - but menstruation may start late. |
|
|
Testicular Feminizing Syndrome |
This syndrome, also called androgen |
|
|
Reifenstein's Syndrome |
This is incomplete androgen insensitivity or partial feminization. |
|
|
Imperato-McGinley Syndrome |
In this condition, the male lacks the enzyme 5 |
|
|
Receptor positive resistance |
The androgen receptor protein has a defect so that the H-R complex can not bind to the chromatin in the nucleus. - or, the hormone-receptor (H-R) complex fails to be accepted on the chromatin. |
|
|
Pseudohermaphrodites |
Pseudohermaphrodites are more common and have the internal genitals of one sex and |
|
|
Hormone treatments in pregnancy |
If a pregnant female takes external hormone or drug treatments, these external chemicals may interfere with the pregnancy. Those chemicals which alter gonadal steroid hormone levels may cause abnormalities in sexual differentiation in the offspring. If estrogens or androgens are elevated during pregnancy, the body will be masculinized. If DES is taken, still other complications arise in female babies, related to uterine cancer etc. If progestins or anti-androgens are taken during the pregnancy, the baby will be feminized. |
|
Activation of the H-P-G system |
Gonadal steroids reach a peak during the |
|
|
Onset of Puberty Postulated Ontogeny of Hypothalamic-Pituitary-Gonadotropin-Gonadal Circuit: Fetus |
a. Secretion of pituitary FSH and LH by 80 days gestation |
|
|
Onset of Puberty Postulated Ontogeny of Hypothalamic-Pituitary-Gonadotropin-Gonadal Circuit: Infancy and Childhood |
a. Negative feedback control of FSH and LH secretion highly sensitive to sex |
|
|
Onset of Puberty Postulated Ontogeny of Hypothalamic-Pituitary-Gonadotropin-Gonadal Circuit: Late Prepubertal Period |
a. Decreasing sensitivity of hypothalamic gonadostat to sex. Increased secretion |
|
|
Onset of Puberty Postulated Ontogeny of Hypothalamic-Pituitary-Gonadotropin-Gonadal Circuit: Puberty |
a. Further decrease in sensitivity of negative feedback mechanism to sex steroids |
|
|
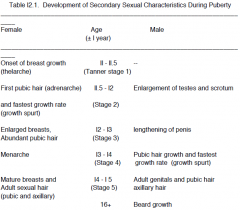
There are a number of terms related to puberty that should be defined: |
Adenarche is the beginning of secretion of adrenal androgens at puberty; |
|
|
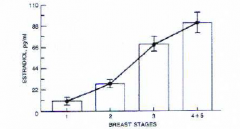
Tanner breast stages by estradiol stages |
|
|
|
Variables influencing the timing of puberty |
The timing of puberty can be influenced by nutrition, so that overweight people reach puberty sooner than underweight people and anorexics show a delay or inhibition of puberty. Stress is a second variable which influences the timing of puberty. Stress causes a delay in reaching puberty as it inhibits gonadotrophin secretion. Thus, victims of abuse (psychosocial) dwarfism often show delayed puberty (Chapter 16). Light cycles are a third variable which influence the timing of puberty. Animals kept in long days (16 hr light) will reach puberty earlier than animals kept in the dark or long night (8 hr light) environments. Social odours or "pheromones" from other |
|
|
Critical weight theory |
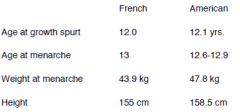
The earliest theory of what controls |
|
|
Changes occurring at puberty in females |
In females, changes begin to be noticed around ll years of age with the onset of breast development (thelarche). Adrenarche is associated with the initiation of pubic hair growth due to adrenal androgens and this is followed by the growth spurt due to a surge of GH. As growth continues, the "critical body weight" occurs and gonadarche begins, with breast enlargement and more abundant pubic |
|
|
Changes occuring at puberty in males |
Male pubertal development is l-2 years behind that of females (why?) In males, puberty begins with the gradual rise in androgen secretion from both adrenal cortex and gonads, resulting in an increase in testes, scrotum and penis size . This is followed by the growth of pubic hair and the growth spurt, 2 years behind the |
|
|
Activation of the H-P-G |
Activation of the H-P-G system involves increased release of FSH-RH and LH-RH from the hypothalamus, FSH and LH from the adenohypophysis and increased levels of gonadal steroids. Figure l7.4 shows the pattern of pituitary-gonadal hormone release during puberty in males and females, superimposed on a graph of growth rate (i.e. height gain in |
|
|
FSH in puberty |
Taking a monthly "window" on the hormone changes in puberty, these figures show that prepubertally, there are low levels of FSH, LH estradiol and progesterone. FSH rises first at thelarche and maintains this level at menarche, |
|
|
LH and estradiol in puberty |
LH remains low at thelarche, begins to rise at menarche and shows adult cyclic fluctuations by a 6-9 months after menarche. Estradiol remains low at thelarche (except for one girl, W), shows some surging at menarche, and adult |
|
|
Progesterone in puberty |
Progesterone is low through thelarche and menarche and shows adult cyclic patterns by 6-9 months after menarche. It is not until the late stages of puberty (after menarche) that the surges in LH occur which are necesesary for follicle release and ovulation to occur. Note |
|
|
GH in puberty |
The changes in GH secretion over a 24 hour |
|
|
Effect of LH on sleep |
Like GH, LH shows a day-night rhythm. In pre-pubertal females, LH is low in both day and night and in early puberty LH shows sleep-related surges or pulses about every 90 min. In later puberty, the nocturnal LH surges begin to |
|
|
Adult females with anorexia nervosa... |
...show amenorrhea (inhibition of the |
|
|
The metabolic changes caused by increased |
These include the development of the external |
|
|
Estrogens |
Estrogens increase uterine, vaginal and breast development and influence fat deposition and distribution. There are sex differences in how people get fat - men get beer bellies, women get fat asses and thighs. Estrogen stimulates |
|
|
Progesterone |
Progesterone often depends on the "priming" action of estrogen for its effect. The estrogen acts to prime the target cells, by initiating cell growth, synthesis of enzymes or progesterone receptors, and then progesterone acts on |
|
|
Gonadal Steroids |
The gonadal steroids act like growth hormones to promote somatic growth in a number of ways. Gonadal steroids also interact with the hypothalamic-pituitary feedback system and activate neural steroid receptors establishewd during the organizational period. The neural receptors for gonadal steroids are located |
|
|
Neuroendocrine activation at puberty: Changes in feedback regulation |
The H-P-G feedback system. In males, gonadotrophins are secreted tonically, or continuously, whereas, female secretion is cyclic. This is controlled LH-RH and FSH-RH release and the sensitivity of gonadal hormone sensitive by neurons of the median eminence, ventromedial and anterior hypothalamus, and |
|
|
Psychological and social changes at puberty |
Mood. First changes in puberty are probably LH-RH-LH and testosterone. May start the changes in sexual arousal. Ultrashort feedback loop - regulation of hypothalamic activity by hypothalamic releasing hormones (negative). |
|
|
Activation of Sex differences in behaviour |
After puberty, sex differences in behaviour increase. Discuss. (1) sexual arousal, (2) aggression, (3) IQ scores, (4) motivation. Are these hormonal or socio-cultural? |
|
|
What determines the timing of puberty? The Gonadostat and Opiostat theories. |
(a) The gonads. The gonads are fully developed at 25 days of age. If the ovaries |
|
|
The exact mechanism which "triggers" puberty is not yet understood and there may be three changes at puberty. |
(l) The secretion of FSH/LH-RH is controlled by the noradrenaline and dopamine |
|
|
Dual mechanism controlling puberty |
l. The hypothalamic-pituitary-gonadal negative feedback loop is the first factor that |
|
|
The Gonadostat Theory |
highly sensitive. Using the male as an example, if a 6 year old boy secretes some testosterone, it is picked up by neural receptors and the hypothalamic-pituitary system is very sensitive, so this testosterone causes a high amount of negative feedback, resulting in a severe inhibition of LH-RH release. Any androgens present in the circulation will shut down the LH-RH system completely. The second system, the intrinsic CNS firing system has a rhythm of cell firing in the arcuate nucleus of the hypothalamus (Figure 17.9). Before puberty, these cells fire at a low rate, releasing very little neurotrnasmitter (probably norepinephrine) As |
|
|
As puberty occurs, there are changes in these two mechanisms. |
During infancy and childhood, any gonadal hormone present causes |
|
|
The gonadostat |
"The gonadostat" has two |
|
|
What is changing at puberty is |
(l) the sensitivity of the hormone-sensitive cells of the hypothalamus and that interacts with (2) the firing rate of the cells of the arcuate |
|
|
The opioid theory |
At puberty, gonadal steroids inhibit opiate release and there are few opiate receptors, so NE release is maximal. Masculinization of opiate system perinatally alters its sensitivity or response to androgens, resulting in a masculine, continuous LH-RH secretion rather than the pulsatile female secretion. Other factors influencing the gonadostat. Figure l7.l1 shows that many other neural, hormonal, |
|
|
Input from the cortex of CNS may mediate stress effects on the timing of puberty |
The pineal gland mediates the effects of light; the amygdala may mediate effects of odours; the hippocampus the effects of adrenal corticosteroids; and the mesencephalon and MFB- may mediate other neural stimulation (e.g. sexual arousal via tactile or visual stimulation (? is this too far fetched?) |
|
|
A number of neural, hormonal and environmental factors can modulate the |
High melatonin |
|
|
Pheromones |
Olfactory stimuli from adult animals may accelerate or inhibit puberty (primer pheromones). High population density and social stress will delay puberty. The hypothalamus controls the timing of puberty. It is affected by catecholamines (dopamine and noradrenalin); indoleamines (serotonin); pineal |
|
|
Delayed puberty |
Can result from both low levels of gonadal steroids (hypogonadism) and low levels of gonadotrophins (hypo-gonadotrophic) or be associated with high levels of gonadotrophins (hyper-gonadotrophic). In some cases, people |
|
|
Advanced puberty |
(precocial puberty). Results from premature gonadotrophin release or from gonadal or adrenal humans, or from taking extraneous hormone treatment. Isosexual = development due to normal sex hormones. Heterosexual = development due to sex hormones of opposite sex. In advanced pubertry, there is precocious sexual development. Guiness Book - youngest mother = Linda Nadina, Peru, l939, 5 years 8 months old. |
|
|
Metabolic Activities of Androgens at Puberty |
l. Stimulates growth of penis, scrotum, seminal vesicle and prostate gland. |
|
|
Metabolic Activities of Estrogen at Puberty |
A. Estrogen. |
|
|
Metabolic Activities of Progestins at Puberty |
B. Progesterone. |
|
|
Chronological age |
Chronological age is how old you are, number-wise. |
|
|
Developmental (Tanner) Stage Visuals |
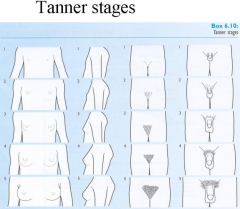
There are 6 tanner stages, and they correspond to level of development in males and females. |
|
|
Tanner stage males |
Tanner I prepubertal (testicular volume less than 1.5 ml; small penis of 3 cm or less) [typically age 9 and younger] Tanner II testicular volume between 1.6 and 6 ml; skin on scrotum thins, reddens and enlarges; penis length unchanged [9-11] Tanner III testicular volume between 6 and 12 ml; scrotum enlarges further; penis begins to lengthen to about 6 cm [11-12.5] Tanner IV testicular volume between 12 and 20 ml; scrotum enlarges further and darkens; penis increases in length to 10 cm and circumference [12.5-14] Tanner V testicular volume greater than 20 ml; adult scrotum and penis of 15 cm in length [14+] |
|
|
Tanner stages females |
Tanner I no glandular tissue: areola follows the skin contours of the chest (prepubertal) [typically age 10 and younger] Tanner II breast bud forms, with small area of surrounding glandular tissue; areola begins to widen [10-11.5] Tanner III breast begins to become more elevated, and extends beyond the borders of the areola, which continues to widen but remains in contour with surrounding breast [11.5-13] Tanner IV increased breast size and elevation; areola and papilla form a secondary mound projecting from the contour of the surrounding breast [13-15] Tanner V breast reaches final adult size; areola returns to contour of the surrounding breast, with a projecting central papilla. |
|
|
Tanner stages for pubic hair for both males and females |
Tanner I no pubic hair at all (prepubertal state) [typically age 10 and younger] Tanner II small amount of long, downy hair with slight pigmentation at the base of the penis and scrotum (males) or on the labia majora(females) [10–11.5] Tanner III hair becomes more coarse and curly, and begins to extend laterally [11.5–13] Tanner IV adult-like hair quality, extending across pubis but sparing medial thighs [13–15]| Tanner V hair extends to medial surface of the thighs |
|
|
Ontogenetic development |
Changes in growth and development shown in humans and animals from the time of conception to birth and throughout their lives until their death can all be viewed as "developmental changes". This life-span view focuses on ontogenetic development as a life-long process and examines the sources of behavioural variability in individuals over their life span. Because there is a high degree of |
|
|
Developmental changes |
Changes in growth and development shown in humans and animals from the time of conception to birth and throughout their lives until their death can all be viewed as "developmental changes". |
|
|
Lifespan Development |
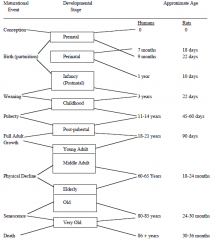
Life-long development consists of a series of patterns of change which differ in age of onset, duration, and termination. Different developmental functions have different times of onset over the life-span and different temporal parametersnd that some developmental changes are |
|
|
Genetic, biochemical and socio-cultural |
Because the development of each individual can be modified by a number of factors, each person or animal has the potential to develop differently if external or internal conditions are modified. Thus, genetic, biochemical and socio-cultural differences all influence the rate and level of development of an individual over |
|
|
Qualitative development changes |
Qualitative changes involve the onset or |
|
|
Quantitative development changes |
Quantitative changes involve the gradual growth or deterioration in the size of a body organ or the expression of a behaviour. |
|
|
Longitudinal research |
A longitudinal study examines one group of subjects at different ages. Thus, one group of children would be studied at 2, 4 and 6 years of |
|
|
Cross-sectional research |
By examining groups of individuals of different |
|
|
Measuring growth |
Growth is a complex process which involves a series of changes as well as increases, or decreases, in size. Growth may include cell |
|
|
Molecular biology and genetics of growth |
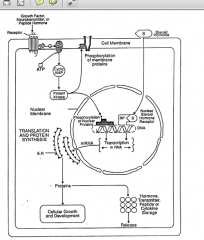
The neurochemical basis of cell growth and development. Growth factors, neurotransmitters, neuropeptides and steroid hormones bind to specific receptors in their target cells to stimulate protein synthesis by activating nuclear regulatory proteins. The steroids bind directly to their nuclear receptors, while the growth factors, neurotransmitters and peptides bind to membrane receptors and activate second messenger systems. The proteins synthesized by the cell may be regulatory proteins such as receptors, G-proteins, enzymes, etc. which serve intracellular functions, including cell growth and development or hormones, neurotransmitters, peptides or cytokines, which are stored in secretory granules and then released. |
|
|
Measuring normal and abnormal development |
Changes in neuroendocrine, physiological and behavioural development can |
|
|
Prenatal development - effects of teratoge |
Prenatal development involves primarily the differentiation and growth of tissues in the body and the brain. Neural and endocrine development are of particular interest in this book, but skeletal, physiological and behavioural development all begin during the prenatal period. Some behaviours, such as fetal movements can be recorded during the |
|
|
Teratology |
Teratology is the study of congenitally deformed fetuses and teratogens are agents which cause these deformations. Teratogens, can be hormones, drugs, environmental chemicals such as lead, polychlorinated biphenyls (PCBs), mercury, or other environmental agents, infectious diseases or radiation (Table1.6). Teratogens can alter neurochemical signals and thus alter |
|
|
Some teratogens known to cause human birth defects |
Hormones |
|
|
Five questions asked about critical (sensitive) periods |
1. Locus What molecular, physiological or behavioural aspect of the system has |
|
|
Examples of circus freaks and their developmental abnormalities |
Hormonal abnormalities |
|
|
The Cincinnati Developmental Test Battery for Rodents. |
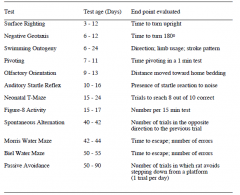
|
|
|
Organizational versus activational effects |
Hormones and other neurochemicals can influence whether developing cells live or die and regulate their rate of growth. Through these mechanisms, these chemical messengers modulate the developmental organization of the nervous, Neuroendocrinology of Development 1 - 13 endocrine and immune systems. Once these systems have developed fully, chemical signals modulate their rate of activity. The interaction of the |
|
|
Physical development |
- morphological measures (limb length, tooth growth, etc.). |
|
|
Reflexes |
- rooting reflex to a touch on the lips. |
|
|
Gross motor development |
- head and hand movement. |
|
|
Fine motor development |
- hand-eye co-ordination: reaching and grasping. |
|
|
Sensory development |
- orientation to visual, auditory, or olfactory stimuli. |
|
|
Cognitive development |
- habituation to repeated stimuli. |
|
|
Language development |
- prelinguistic speech. |
|
|
Moral development |
- Kohlberg's stages of moral development. |
|
|
Social-emotional development |
- age of emergence of emotional expressions (smiling, grimacing, etc.) |
|
|
Sexual Development |
- Freud's stages of psychosexual development. |
|
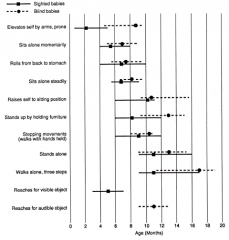
|
Comparative development of blind and sighted babies. The mean age and range in ages at which sighted babies (square) and blind babies (circle) reach each development milestone is shown. |
|
|
Organizational |
A mode by which steroids affect behaviour. - Organize neural system development during critical |
|
|
Activational |
A mode by which steroids affect behaviour. - Activate neural pathways that are already present, e.g. effect of estrogen and progesterone on female sexual behaviour. |
|
|
Two problems with this dichotomy |
1. How is this dichotomy defined and how do behavioural and |
|
|
How do organizational and activational effects differ? |
1. Organizational effects are permanent (irreversible); activational |
|
|
Changes in development in adolescence and adulthood |
Biological maturity occurs at puberty with the capacity to reproduce, but social, cognitive and emotional development continue well into adulthood. The neuroendocrine changes that occur at puberty, along with a person's prenatal development and psychosocial experiences during early development shape their adult behaviour. During adolescence and adulthood, hormone levels are altered by |
|
|
Developmental changes in old age |
As a person enters old age, they go through a number of biological, cognitive, social and personality changes. The neuroendocrine system shows a number of changes in function with age. Sensory abilities decline with age as do neuromuscular capabilities and the reproductive capacity. The menopause marks |
|
|
Internal and external influences on development: developmental |
The pattern of neural development has some plasticity and can be modified by the interaction of internal (genetic, hormonal, neurochemical) and external (sensory stimuli, nutrients, experiences) stimuli. Many aspects of postnatal development involve learning by experience. The capacity to learn is thus a |
|
|
Neuroendocrinology of development |
The brain, endocrine glands, and the cells of the immune system grow like |
|
|
The role of the immune system in development |
The thymus gland is essential for the maturation of T and B cells and without the thymus gland the developing organism suffers from massive infections and “wasting disease” |
|
|
Neuroendocrine theories of aging |
Many aspects of the neuroendocrine |
|
|
Some theories of Human Development |
Sigmund Freud - Psychosexual and personality development |
|
|
The neuroendocrine system as the mechanism of change in development |
“A unique feature of developmental theories is that they must specify some process or mechanism responsible for producing the changes that constitute development. The change mechanism constitutes a “motor” that power development. This element is crucial. How can one explain development without |
|
|
Theory that biological maturation is the cause of development |
Several theories (Freud, Bowlby, Gesell and Money & Ehrhardt) posit biological maturation as the cause of development, thereby implying that individual growth is relatively fixed and mostly immune to environmental stimuli. |
|
|
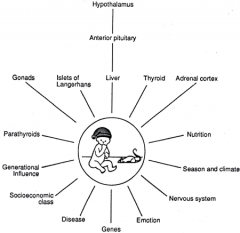
Factors affecting growth and development. Development is a process which is influenced by the complex interactions among numerous internal and external factors. Virtually all of these factors affect development through the neuroendocrine system. |
|
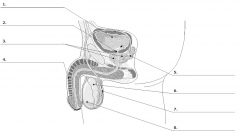
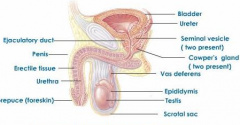
Male Reproductory System Labeled |
|
|
|
The testes form part of the third order hypothalamic-pituitary-gonadal feedback system. The testes are composed of: |
(1) seminiferous tubules and (2) interstitial or Leydig cells. Spermatogenesis occurs in the Sertoli cells of the seminiferous tubule, and this is stimulated by FSH and by testosterone (acting in a pracrine fashion (?). Androgens, particularly testosterone and androstenedione and produced in these Leydig or interstitial cells and the Leydig cells are stimulated by LH or interstitial cell stimulating hormone. |
|
|
Effects of Castration |
|
|
|
Feedback of testosterone on hypothalamic and pituitary hormones |
You know that in the H-P-G system, there is long-loop negative feedback of testosterone |
|
|
Testosterone rhythms |
Androgen secretion from the testes shows a daily and an annual rhythm, with some researchers claiming a lunar or monthly rhythm (Figure 2.3). The daily rhythm of testosterone secretion shows a peak in the early morning (around 8 a.m.) and a |
|
|
Testosterone dependent male behaviours |
There are a number of sexually dimorphic |
|
|
What happens when the testes is implanted or removed in roosters? |
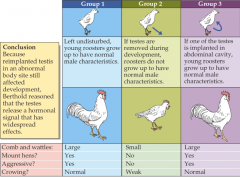
|
|
|
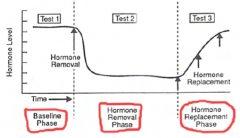
The three phases of a standard hormone removal and replacement experiment. Behavioural tests are conducted one or more times during the baseline, hormone removal (post-castration) and Hormone replacement phases. A series of hormone replacement injections (arrows) is usually given during the hormone replacement phase. Hormone removal: surgical pharmacological - drug (antagonist) Immunological (antibodies) Hormone replacement: whole glands purified hormones synthetic hormones |
|
|
Constraints on hormonal activation of behaviour |
In a standard experiment on the effects of |
|
|
Not all species respond the same way to gland removal (e.g. castration) and hormone Factors that affect an animal responding differently to castration. |
The age of the animal, its past experience and learning also influence the effects of the |
|
|
When an animal or human subject is tested, the stimulus situation or manner in which |
|
|
Male sexual behaviour |
Sexual behaviour of male mammals involves an appetitive or courtship phase, followed by a consummatory or copulatory phase. During the courtship phase, there is a change in sexual arousal and in male-female interactions which may begin as exploratory or aggressive in nature. During this period, the male is attentive to the odour, tactile responses, and auditory stimuli from the female and may respond by scent-marking; touching, nudging or licking the female, and producing sonic or ultrasonic vocalizations. During the copulatory phase the male mounts the female, gains penile intromission, shows a species typical pattern of penile thrusting and ejaculates. Males of some species mount and ejaculate only once while others have multiple ejaculations. During the copulatory phase, other behaviours such as aggression, olfactory investigation, scent-marking and vocalizations, may also occur. |
|
|
Sexual arousal |
Sexual excitement causes a "burst" of LH to be discharged and this stimulates a burst of testosterone. This may facilitate release of testosterone from Leydig cells and facilitate sexual activity. In bulls this can occur within about a half an hour. This response represents an increase in synthesis of testosterone. In mice, it has recently been shown that plasma testosterone levels increase within 30-60 min of exposure to a novel female, and does not depend on copulation (pheromones). |
|
|
Sexually experienced males castrated before puberty seldom indulge in sexual behaviour. Males castrated in adulthood show gradual decreases in sexual activity which depend on... |
...(1) the species and (2) the amount of previous sexual experience. Following castration of sexually experienced males, there are large species differences in the proportion which continue to mount and ejaculate. Notice that the decline in copulatory behaviour is gradual for all species (Figure 2.5) and that for rats, copulation is reduced almost to zero after nine weeks. Cats show a similar decline in sexual behaviour, but 20% may still copulate 15 weeks after castration. With sexually experienced rhesus monkeys, 60% still copulate 14 weeks after castration and almost all sexually experienced dogs continue to copulate. Thus, although castration inhibits sexual behaviour, there are large species differences in this and individual differences within species. (Effects of castration on sexually inexperienced males: prepubertal castration. |
|
|
Dose dependent effects for hormone-replacement therapy. |
When hormone-replacement therapy is given after castration to rats (Figure 2.6) there is a dose-dependent effect; so that a certain dose is required to return to pre-castration levels of performance. Lower doses will result in partial performance. In some cases (see below) high hormone doses result in enhanced performance.But the dose of hormone it takes to stimulate a certain behaviour is not fixed. It depends on the time after castration, immediately versus long term castration. Hormone injection immediately after castration requires lower doses of hormone than after long term gland ablation or hormone deficiency. This is because of down-regulation of receptors. |
|
|
Methods of hormone administration |
oral administration, injections, intramuscular at i.p into the interperitoneal cavity. A third method of giving a hormone is to use a capsule implanted under the skin. A capsule gives a constant, steady release of hormone while oral release pills give slow release and injections provide "pulses" of hormone, so different methods of hormone administration will result in different types of stimulation (constant vs pulsatile). If hormones are naturally secreted in pulses, injections provide a more natural treatment and you can use "osmotic mini-pumps" which can be programmed with mini-chips to administer mini-pulses of hormone release. They are now working on these for insulin for diabetics and other hormone treatments and these can be programmed to give certain doses of hormones at certain times to provide a normal circadian rhythm of hormone release (find data on this). |
|
|
Effects of other steroids on male sexual behaviour |
Estrogen injections also stimulate male sex behaviour in castrated adult males (Soderson, Hormones and Behavior,1973, 4, 247-256) and others. Soderson showed that adult male rats with sexual experience performed almost equally as well in sexual behaviour experiments given estrogen or testosterone, but estrogen doses had to be much larger than the testosterone doses. |
|
|
Humans have been castrated for two reasons: |
to remove testicular tumors, and, as treatment for sex offenders. Studies of castrated sex offenders, however, show that there is a great deal of variability in response to castration. In Heim's 1981 study, 39 sex offenders were castrated between 32 and 69 years of age and they answered questionnaires given about four years after their operation. This demonstrates a number of problems with certain studies on hormones and human behaviour. First, the data depend to a great extent on introspective self reports rather than objective behavioural data; it is retrospective; and the subjects must be relied on to be truthful and to return the questionnaires. |
|
|
Castration can cause a number of somatic and psychological changes in males. |
These include inreased body fat, breast development (gynecomasty), osteoporosis and bone pains as well as increased "calmness", or feelings of depression and isolation. |
|
|
Heim suggests that age and personality contribute to the effects of castration on humans and suggest a number of factors which may account for these differences. These include: |
1. irritation caused by castration. 2. differences in organizational effects and their activation at puberty 3. the patient's psychological attitude to castration whether they think it should influence their behaviour or not. 4. the stimulatory influence of adrenal androgens. 5. experience. 6. stimulus or situational factors, which enhance or inhibit sexual behaviour; i.e., marriage or not; wife's attitude, etc. |
|
|
Are testosterone levels lower in homosexual men than in "heterosexuals"? |
In a review of six such studies, however, Rose (19 ) found only one study where homosexuals had a lower testosterone level, one where they had a higher level and four with no difference. Meyer-Bahlburg (1977) reviewed much of the research on hormone levels and homosexuality and concluded that (1) hypogonadism (undeveloped testes) seldom result in homosexual orientation, (2) most homosexual men have normal testosterone, LH and FSH levels in adulthood, and (3) female sex hormone treatment (estrogen) does not lead to a change in sexual orientation. Thus, hormone levels in adulthood do not seem to differ between homo and heterosexual males and any hormonal influence on gender identity appears to occur prenatally. |
|
|
Aggressive behaviour |
There are two types of studies to look at with respect to hormones and aggressive behaviour: correlational and experimental . A correlational study first observes the aggressive behaviour of a group of animals and ranks them from most to least aggressive. Blood samples are then taken from each animal and analyzed for androgen levels to see if the aggressive, dominant males have a higher testosterone level than the subordinate males. |
|
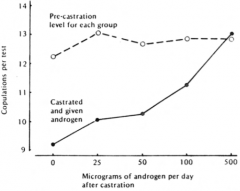
|
The effects of increasing doses of testosterone propionate on the frequency of copulations per test in male rats compared to pre-castration levels for each group. |
|
|
Correlation of testosterone levels and aggression |
In humans and primates, castration does not seem to reduce aggression. In one study, nine of 16 men castrated for sexual offences died as the result of fighting (Kling, 19 ). In prisoners, more violent ones were found to have higher testosterone levels and less anxiety than less aggressive prisoners. Castration results in a reduction in fighting in rats and mice; but little change in monkeys and dogs. In cats, Art(?) found a rapid decrease in fighting in 53%, slow decrease in 35%, and no change in 12%. In gerbils, castration increases aggressiveness. |
|
|
Androgen replacement therapy |
Testosterone injection into castrates reinstates aggressive responses but this often depends on individual precastration aggressive levels. Dihydrotestosterone and estrogen have been shown to increase aggressiveness in castrated male mice. |
|
|
As shown by Yahr (19 ) with gerbils, some may become more aggressive and this depends on prior experience and on the specific stimulus situation in which it was tested... |
If the male was dominant and very aggressive before castration, he is more likely to continue to be very aggressive. If the male is tested in its own home territory, and you place an intruder in, the castrated male is just as aggressive, if not more so, than an intact male. Thus, the stimulus situation and the effects of learning and/or experience may counteract the effects of castration. The age at castration also influences aggressive behaviour. Males castrated before puberty are more aggressive than males castrated in adulthood. |
|
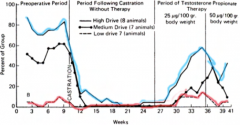
|
Effects of castration and daily treatment with 25 ug of testosterone on the mating behaviour of male guinea pigs that showed high, intermediate or low levels of sexual behaviour before castration. Note that when testosterone therapy was given, the individual differences seen in the baseline period returned. When mated, females given oil never fought males, but females given testosterone in infancy showed fights 50% of the time, females given estrogen showed fights 1/3 of the time. Males given estrogen had smaller accessory sex glands thus estrogen in infancy may inhibit masculinization in intact males. Testosterone injections in females interfered with normal sexual behaviour and increased aggressiveness. This suggests that the organizing effects of testosterone in infancy are widespread and may affect many neural areas. |
|
|
Hormones and aggression in humans |
Correlational studies of androgens and aggression in humans have often used pencil and paper tests to measure aggression and these results show very low correlations with androgen levels. In correlational studies looking at hormone levels of wrestlers after winning or losing matches. Also studies of men dying in aggressive encounters. Are there any experimental studies on humans? |
|
|
Responses of fighting. Adrenocortical levels (Bronson and Elefthenieu, 1965, Science). These authors reared mice in isolation and tested them at 85 days of age. Mice were placed in the home cage of a "trained fighter" in three groups: |
(1) fighter allowed to attack on each of 15 days; (2) fighter allowed six attacks on first five days only, (3) fighter never allowed to attack. Blood was collected and analyzed for corticosterone. Mice never attacked (Group three) had low corticosterone levels. Both of the exposed groups had high concentrations. This shows that in defeated mice there is a high corticosterone level. The peak occurs about one hour after exposure to the aggressive male. Subordinates were found to have heavier adrenal glands. |
|
|
Leshner has proposed a model of hormones and aggression. In simplified form it has three components: |
(1) baseline hormone levels, (2) stimulus processor and (3) response mechanism. (1) Baseline hormone levels. Testosterone and ACTH determine the animal's response to an intruder. High testosterone levels and low ACTH levels favor aggression or attack; low testosterone levels and high ACTH favor submission. (2) The C.N.S. response is determined by (1) neonatal hormones, (2) adult hormone levels and (3) past experience. These act to alter the shift of brain circuits to fear or aggression in novel situations. Little work has been done to test this proposition. (3) Hormonal feedback. We have seen that defeated males show increased adrenocortical activity, dominants see unaffected. Some studies have also found a depression of testosterone levels in defeated animals, and a decrease in LH. Dominant animals do not seem to show increases in testosterone or LH. Defeat appears to alter the amount of neurotransmitters in the limbic system of the brain; increasing catecholamine and serotonin levels (neurotransmitters). |
|
|
Lesher's model proposes... |
...that there is a gonadal-adrenal-neural interaction in aggression. A baseline testosterone-corticosteroid balance determines the fight-flee reaction. The consequences of fights serve to alter the testosterone-corticosteroid balance, producing less testosterone and more C in defeated animals. This serves to alter their initial response to new situations and their odour and pheromne production. |
|
|
Scent marking |
Most mammals have scent glands on their bodies, such as the ventral gland of the gerbil or the flank gland of the hamster, which are used for scent marking. Dogs, rats and many other mammals use urine for scent marking. Scent marking is the release of social odour or pheromones. It is also an androgendependent male behaviour. It is eliminated by castration and reinstated by testosterone injections. It probably depends on both neonatal and adrogenization and circulatory testosterone levels. |
|
|
Experiments on urine marking in male and female rats show that: |
1. males urine-mark about twice as often as females 2. castration eliminates marking in both sexes 3. testosterone increases marking in both sexes 4. estrogen increases marking, in males, not females 5. P alone does not increase marking in either sex |
|
|
Ultrasonic vocalizations |
During courtship and sexual behaviour, male rodents produce ultrasonic vocalizations. Castration reduces the number of ultrasounds and testosterone replacement increases the frequency of ultrasounds. |
|
|
Anabolic steroids and their effects on males |
The effects of anabolic steroids on muscle mass and muscle strength is a controversial topic. Anabolic actions of steroids are those that build skeletal and muscular mass and the anabolic effect of steroids, particularly synthetic androgens caus increasesd muscle mass during the growth spurt in males at puberty. |
|
|
Progesterone |
– Anti-androgenic actions |
|
|
Oxytocin/Vasopressin |
– Released during sexual behaviour (orgasm) – Effects on smooth muscles (vas deferens) – Effects on blood pressure (for erection) – Neuromodulatory effects in the brain |
|
|
Stress hormones |
– ACTH/Cortisol released during stress – Likely reduce sexual behaviour |
|
|
Haupt and Rovere (1984) have reviewed the studies on the effects of anabolic steroids and concluded that those studies in which anabolic steroids increase strength had four features: |
1. used trained weight lifters. 2. used a particular type of synthetic steroid called methandrosterolone (Dianabol) 3. test on single repetition maximal weight method 4. stay on a high protein diet |
|
|
The problem with taking these steroids are the side effects: |
both psychological and physical. The psychological side effects include a change in mood, increased aggression, and reduced sex drive. The physical side effects include gynecomostia (breast enlargement) and endocrine side effects because the synthetic steroid provides external negative feedback to the H-P-G third-order feedback loop. The steroid, thus reduces LH-RH, FSH-RH, LH and FSH levels and thus inhibits the synthesis of testosterone in the gonads. Because the synthetic steroid does not have all of the same effects of testosterone, fertility is reduced. Figure 2.13 shows that administration of an external injection of androgen dramatically reduces sperm count. Other side effects may involve liver and circulatory system damage, but these, are controversial (Haupt and Rovere). Most of the side effects are reversible after the use of steroids is stopped and return to normal. |
|
|
Female weight lifters who take steroids... |
...usually have a disrupted menstrual cycle, voice changes, and some masculinization (e.g. Russian women's swim team: we came to swim,not to sing). Neural sites of androgenic activation of male behaviour -write about this. Effects of testosterone metabolytes on behaviour and the aromatization hypothesis. Say a few words on this. Effects of hypothalamic and pituitary hormones in male socio-sexual behaviours. Effects of NT on male sociosexual behaviours. Effects of neuropeptides on male sociosexual behaviours. |
|
|
Sexually dimorphic behaviours |
Behaviours specific to, or shown primarily by, animals of one sex. They are often measured by how examining play behaviour and play preferences. |
|
|
Testes determining gene (SRY) |
In mammals, the Y chromosome contains the testes determining gene (SRY), which causes the bipotential gonads to develop as testes. Without a Y chromosome the gonads develop as ovaries. Fetal testosterone and adrenal androgens cause masculinization of the external genitals and the neural pathways in the brain. The external genitals determine the behaviour of other people and, as the child develops, he or she develops a self-image of their own body. Between l.5 and 4 years of age the child begins to develop a juvenile gender and role identity, including sex differences in play behaviour, toy preferences. |
|
|
Perinatal sex differences in androgen levels... |
...can result in sex differences in behaviour at very early ages. For example, very young rhesus monkeys (3.5-9 months old) show sex differences in play behaviour (Figure 13.3). Males have much higher levels of pursuit play (chasing) and rough-and-tumble play than females. Masculinized females (females given prenatal testosterone treatment) show an increase in chasing and rough and tumble play, indicating that this sex difference is mediated by prenatal hormones. |
|
|
The most important biological aspect of sexual differentiation in terms of the psycho-social environment... |
...is the sex of the external genitals. These determine how other people will behave towards a child, as a male or a female, from the moment of birth, and thus determine gender of rearing (see Table 6.1 in Chapter 6 on sexual differentaition). Parents and other adults have culturally determined expectancies about how males and females should differ and their perceptions of their newborn infants are "filtered" through these expectations. |
|
|
At puberty, the rise in gonadal hormone levels causes... |
...the development of physical, neuroendocrine and psychological changes which combine with juvenile gender identity and role to determine adult gender identity and role. All of these components interact to determine adult sexuality, which involves the hormonal activation of neural centres in the brain which cause sexual arousal and sexual motivation. Social learning and experience during development then interacts with the hormonal changes during puberty to provide a psychological interpretation of the physical changes at puberty. |
|
|
Theories in gender identity |
• Biological–Freud – Sex differences in genitals determined iden4ty – Stages of psychosexual development • Social Learning-‐ Bandura – Differen4al reinforcement of sex typed behaviour – Learn by watching same sex role models • Cogni4ve Development-‐ Kohlberg – Gender iden4ty develop along the same lines as cogni4ve development (think Piaget) |
|
|
Money and Ehrhardt's theory of gender identity and role development |
Money and Ehrhardt's (l972) combined biological-cognitive learning approach considers both the prenatal hormonal organization of behaviour and the influences of social learning on gender identity and role development (Table 13.3). According to Money and Erhardt, the development of gender identity and role depends upon "a prenatally programmed disposition coming into contact with postnatally socially programmed signals". This theory has six stages of psychosexual development which parallel those proposed by Freud.
|
|
|
The prenatal stage |
Prenatal hormones "predispose" the organization of the brain to develop a male or female sex identity and role. But this predisposition requires postnatal social conditioning (see below). The perinatal surge in testosterone levels in males (Figure 13.2) is the primary hormonal event in sexual differentiation as it masculinizes the developing fetus. |
|
|
Gender dichotomy |
Gender dichotomy concerns the social expectations and socialization process by which boys and girls are treated differently. The child's external genitals determine the behaviour of the parents and other people toward the child (gender dicotomy) resulting in differences in communication with boys and girls. Parents attitudes differ towards boys and girls from the time of birth (Figure 13.4). During infancy, parents allow boys to behave more independently than girls. Sex differences are shown in play behaviour, playmates and toy preferences. Boys show more rough and tumble play and this occurs in rats, monkeys (as shown in Figure 13.3) and humans. During play behaviour children take the appropriate male or female roles, prefer to play with sex-appropriate toys and prefer playmates of the same sex. This sex difference in early play behaviour occurs long before puberty and has been attributed to perinatal androgens, but may also be influenced by social factors. |
|
|
Core gender identity |
The interaction of biological factors and parental behaviour along with the child's self-perception of his/her body leads to the development of a core gender identity. This means that deep down, the child knows they are a male or female and not the opposite sex. Once the core gender identity develops, a child will not say "My name is Jimmy and I am a boy and when I grow up I am going to be just like mommy". The child has the concept that they are male or female and this has a number of implications for their future behaviour (i.e., to be like mommy or like daddy). The child's concept of male or female roles are thus based on the behaviour of other adults; whether parents, neighbours or characters in TV shows. Core gender identity develops between l l/2 and 4 years of age. |
|
|
Gender identification and role rehersal |
Juvenile gender identity and role rehersal develop from 4-7 years of age. Once children develop a core gender identity, they develop appropriate behaviours to go with that gender identity. During gender identification and role rehersal, children learn both male and female sex roles. The sex role of their own sex is learned by identification with the same sex parent and the role of the opposite sex by complementation. Children identify with the parent of the same sex and learn the complementary role of the parent of the opposite sex. |
|
|
Consolidation |
Consolidation occurs from the age of 7 to puberty (7-l2). This is a stage of more-or-less sexual segregation, in which boys and girls maintain separate "cultures". During the stage of consolidation, the child assimilates what he/she has learned about adult sex roles and behaviour and learns how to act as an adult. |
|
|
Puberty |
Finally, during the stage of puberty, there is activation of heterosexual interest, love and sexual arousal, romantic attachment and erotic imagery. |
|
|
Transvestite |
Men who dress in the clothes of women or who feel compelled to cross-dress, often in relation to sexual arousal. There is no gender conflict. Of course, a woman may wear a suit and tie or men's work clothing without being considered a transvestite, but a man in a pink dress or nylons and high-heels is immediately so labelled. The DSM-IV-TR (2000) defines this as "Transvestic Fetishism" and considers it a "disorder described only in heterosexual males" |
|
|
Homosexuality |
This involves erotic fantasy and sexual interaction with someone of the same sex. Homosexuality can be of two types: Obligative which is compelling or exclusive homosexuality, with no heterosexual interest; and Facultative, which is optional homosexuality, which occurs in situations where no heterosexual contact is possible, such as sexually segregated schools or prisons. Often a facultative homosexual is heterosexual in other situations, or bisexual. Obligative homosexuality may be more of a gender "conflict" than facultative homosexuality. |
|
|
Bisexual |
Sexual orientation toward both males and females. |
|
|
Transsexual |
Someone who believes that they are the opposite sex and takes to living, dressing, and playing the role of the opposite sex. This is often done by people who desire to have their sex changed. They may undergo social, hormonal and surgical sex reassignment. Many are people who believe that they belong to the opposite sex and desire to have the body, appearance and social role of the opposite sex. |
|
|
Causes of gender identity and role conflicts |
According to Money and Ehrhardt (1972) and Money (1994), gender identity and role conflicts and psychosexual dysfunction can develop when there are sex errors in any of the components of the development of gender identity and role. |
|
|
Genetic bases |
Disorders involving genes on the X chromosome or the testes determining gene or other genes on the Y chromosome can lead to errors in sexual determination. These include errors of sex chromosomes (45 X0 = Turner's syndrome; 47XXY = Kleinfelter's syndrome); testes determining gene errors (SRY); errors in genes for the synthesis of glucocorticoids (congenital adrenal hyperplasia) and genes for the synthesis of androgen receptors (androgen insensitivity syndrome). Since these genetic mutations may be inherited, gender identity and role conflicts may occur in families. |
|
|
Prenatal hormone disorders |
Failure of "normal" sexual differentiation of the brain and/or external genitals may predispose a person to gender conflicts. Both neural and genital differentiation can be affected by lack of androgens or androgen receptors in males or excessive androgens in females. Hormonal disorders may also occur in males because the enzymes aromatase or 5 alpha-reductase are absent, in which case the disorder may be inherited. Teratogens may also cause neonatal hormone disorders as can maternal hormone treatment in pregnancy with androgens or progestins. |
|
|
Ambiguous external genitals |
If a child is born a hermaphrodite or pseudo-hermaphrodite, with ambiguous external genitals, this may lead to ambiguous sex assignment at birth and ambiguous gender of rearing by the parents. This can occur in syndromes which result in incomplete closure of the urogenital sinus of a male or partial closure of the vagina in a female. Incomplete development of the penis in males or enlargement of the clitoris in females may also cause ambiguity of sex assignment. Thus, genital ambiguity can occur when a female has an enlarged clitoris or a male's scrotum has failed to fuse, resulting in a vaginal-like opening. This genital ambiguity causes problems in naming the baby, in the parents "concept" of the baby (Fig. l3.2) and in ambiguous treatment of the baby in terms of early social behaviour. |
|
|
Early Socialization |
Parental encouragement of cross-dressing may be a predisposing factor for transvestism, which is defined only for males. Thus, males who are encouraged to dress as females and take on a feminine role may develop conflicts of gender identity. Parental encouragement of cross-dressing and female gender role (in boys) may include close maternal contact, reduction of aggressive behaviour, and encouragement to play with female toys (dolls). Reinforcement of children for playing with opposite sex toys or cross-dressing may facilitate gender dysphoria (Hoyenga and Hoyenga 1979). Although such explanations are compelling, there seems to be little evidence for the influence of parents on gender identity disorders. |
|
|
Poor parental sex role models |
If the child has no 'normal' sex role models from which to learn sex roles he/she may develop gender conflicts. According to Money and Ehrhardt (1972), the most difficult situation for a young child's sexual identity and role development is to have a parent who "switches roles" e.g. a transvestite father (p.l5); a domineering mother and a weak father; a bisexual mother. |
|
|
Reversed identification and complementation |
A child may identify with the parent of the opposite sex and learn the role of the same sex parent as a complementary rather than primary role. A child could, therefore, desire to grow up and take on the role of the opposite sex parent, rather than the same sex parent. In their cross-cultural review, however, Whitam and Zent (1984) found that many homosexual and heterosexual men began cross-dressing before the age of 6, and suggested that gender identity and role reversal occured without any parental or formal influence and before the child began to identify with one parent or the other. |
|
|
Sexual segregation |
Sexual segregation during adolescent development, involving cultural injunctions against boy-girl relationships and exposure only to children of the same sex, may lead to a failure to learn complementary sex roles and a desire to be with members of the same sex due to ignorance or inexperience with members of the opposite sex (e.g. the Brtitish public school syndrome). This may give rise to facultative homosexual relationships, but not to gender identity disorders. |
|
|
Social learning at puberty |
With the populization of homosexuality as a life-style on television, gay-pride week and the gay and lesbian parade, people may choose homosexuality, transvestism or other "alternative" lifestyles as they develop their erotic interests. This may be a response to social pressure from friends, a desire for an anti-establisment lifestyle or a reaction against parental values. Many groups advertise the benefits of a homosexual lifestyle. |
|
|
Dropping out of the battle of the sexes |
Some teenagers may simply be terrified of approaching the opposite sex due to a lack of knowledge about social interactions, fear of rejection or traumatic experiences. For these people, avoiding the opposite sex may result in the formation of sexual relationships with the same sex or no sexual relationship at all. |
|
|
Andrenogenital syndrome |
Adrenogenital syndrome, or congenital adrenal hyperplasia (CAH), is the masculinization of a genetic (46XX) female by excessive androgen release from the adrenal cortex (Speiser and White 2003). The degree of masculinization can vary from very slight, in which case corrective surgery can be done and the child reared as a girl (Figure 13.6). In other cases, masculinization is complete and the child is thought to be a boy and reared as a boy (Figure 13.6) (Money and Ehrhardt 1972; Speiser and White 2003). In some cases CAH children are assigned as females at birth and reassign themselves as males later. |
|
|
Polycystic ovarian syndrome (PCOS) |
Polycystic ovarian syndrome causes infertility in up to 20% of women (Eggers and Kirchengast 2001). This disorder involves androgenization, insulin resistance, obesity, menstrual dysfunction and hirsutism (Setji et al 2007; Baldwin and Witchel 2006). The excess hair growth, lack of menstrual periods and infertility are stressful to these women, who complain of a "theft of womanhood" (Kitzinger and Willmott 2002). About half of the women seeking female-to-male sex reassignment have been found to have PCOS (Balen et al 1993; Baba et al 2007; Bosinski et al 1997). This suggests that the hyperandrogenization in PCOS women may masculinize them or that their unfeminine appearance may cause sufficient gender dysphoria for them to seek sex reassignment. |
|
|
Maternal androgen levels |
Mothers with high androgen levels may have slightly masculinized daughters. Hines et al (2002) found that maternal testosterone levels were correlated with masculinized gender role in 3.5 year old girls. Likewise women who are exposed to environmental androgens may have masculinized daughters. |
|
|
Progestins in pregnancy |
If a pregnant female takes progestins to prevent a miscarriage, these may have androgen-like properties and have a masculinizing effect on the fetus. If the fetus is female, these may increase masculinization (Table l3.8) but gender identity and sexual orientation are congruent with gender of rearing. |
|
|
Androgen insensitivity syndrome (testicular feminizing syndrome) |
Androgen insensitivity syndrome is an X-linked recessive trait in a genetic male (46XY) who lacks receptors for androgens, thus the target cells are insensitive (see Wilson 2001; Hughes 2006). There is a failure to masculinize the person's physical characteristics and they develop as feminine (Table 13.9). There is incomplete closure of the urogenital sinus and incomplete development of the penis. This results in a short vagina and an incomplete uterus, but the internal genitals (i.e., the gonads) are male - testes. Ovaries fail to develop. At puberty, breasts develop normally in response to adrenal and testicular estrogens, but there is no menarche, no menstrual cycle, and no fertility. Male secondary sex characteristics fail to develop; so there is no beard, voice change or male muscular development (Figure 13.8). As people with androgen insensitivity syndrome appear feminine, they are reared as females, and their gender identity and gender role develop as female. |
|
|
Imperato-McGinley Syndrome |
Some 46XY males have a deficiency in the enzyme 5 alpha-reductase, which converts testosterone to 5 alpha-dihydrotestosterone, or a deficiency in the enzyme 17 beta-hydroxysteroid dehydrogenase, which converts androstenedione to testosterone (see Figure 13.7). These males have a partial failure of the external genitals to masculinize during fetal development (see Table 13.10) and the infant is assigned a female gender at birth. At puberty, however, testosterone levels increase as androgens are synthesized by different pathways (see Chapter 6 on sexual differentiation) and the child is masculinized at puberty. |
|
|
Animal models of physchosexual differentiation |
If a female rat or monkey is given perinatal injections of androgens, they can be masculinized. This is a model of andrenogenital syndrome. Similarly, if a male rat or monkey is castrated or given anti-androgenic drugs during the perinatal period, they can be feminized. This is a model of androgen insensitivity syndrome (testicular feminization syndrome) (Phoenix et al 1959; Baum 1979). Studies with these animal models can help to understand the role of hormones and social experience on the development of sexually dimorphic behaviour and sexual orientation. |
|
|
The anti-hormone viewpoint |
Some people believe that gender identity and gender role development have nothing to do with hormones, but are due solely to "social pressure" (Rogers 1994; Ross, Rogers and Walsh 1982; McCulloch 1978) and are critical of Money and Ehrhardt's (1972) theory of the interaction between hormones and environment in the development of gender identity and role (Rogers and Walsh 1982; Rogers 1988). DeBold and Luria (1983) provide a critique of the Rogers and Walsh (1982) critique of Money's ideas. The debate is continued by Rogers (1983) and DeBold and Luria (1984). |
|
|
A biological basis for homosexuality? |
Dorner (l983) argued that there is a biological basis for homosexuality. He suggested that homosexuality was due to improper masculinization during prenatal development, possibly as a result of prenatal stress during pregnancy. He suggested that prenatal stress demasculinizes males and that a biological basis for male homosexuality could be found in the failure to masculinize the brain and hypothalamic-pituitatry-gonadal feeback system. |
|
|
Prenatal stress and homosexuality |
Dorner (l983) found that males born in Germany within a month of the big bombing raids of WWII had a higher percentage of homosexuality than males born before or after the bombing raids. In fact, the rate of homosexuality in these males was twice the average rate in Germany (Figure l3.11). Dorner argued that the prenatal stress suffered during the bombings inhibited masculinization of these males in the same way that Ward (1972) found prenatal stress caused feminization and demasculinization in male rats (see Chapter 8). Dorner's results were greeted with disdain, if not outright aggression by researchers who believed that homosexuality was a response to rearing or social-psychological influences during development. Bailey et al (1991) could not replicate Dorner's finding that homosexuality in human males was related to prenatal stresss, but these researchers did find a familial genetic link in homosexuality. |
|
|
Hypothalamic-pituitary-gonadal feedback system of homosexual males |
There are sex differences in the gonadal steroid hormone feedback mechanism. Testosterone or estrogen injections provide negative feedback on LH in normal males, causing reduced LH release, but positive feedback in females, causing an LH surge. Dorner (l983) found an LH surge in homosexual males after estrogen injection, suggesting failure of prenatal masculinization (Figure l3.12a). These suggestions were not received favorably, but a replication of Dorner's results by Gladue et al. (l984) (Figure l3.12b) has lent support to the hypothesis that in some homosexuals there has been a failure of masculinization and these males have a feminized neural development and hypothalamic-pituitary-gonadal feedback system. Gladue et al's (1984) results have also been criticized. Hendricks et al (1989) could not replicate these results nor could Gooren (1986a,b). Thus there is no final conclusion as to whether there are differences between homosexual and heterosexual men in their LH response to estrogen. |
|
|
Imperato-‐McGinley Syndrome |
• 46XY • Deficiency in 5 α-‐reductase OR deficiency in 17-‐β hydroxysteroid dehydrogenase • Assigned female at birth-‐ masculinized at puberty • ‘Guevedoces’ (testes at 12 years) • Many change from female sexual iden4ty to male! What if there was such a disorder where 5-a reductase was not present? 46XY testes develop, yet testes develop internally, then testes become external. Testes are not as visible and not as large. They are born as male but they look like girls, and they become masculinized.
|
|
|
What about homosexuality in females? |
Mustanski et al (2002) point out that much less work has been done on the biological basis of female homosexuality than on male homosexuality (Table 13.15). McFadden and Champlin (2000) and McFadden (2002) found that otoacoustic emissions and auditory evoked potentials differed between men and women. They showed that homosexual women had masculinized responses, suggesting that homosexual and bisexual females showed masculinization of the inner ear. But the same measures showed that the auditory system of homosexual males was hyper-masculinized, which is contrary to their argument of prenatal masculinization in homosexual females. Clearly these results need to be examined carefully (see Mustanski et al 2002). In studies of transsexuals, Bosinski et al (1997) found high rates of hyper-androgenic disorders in female-to-male transexuals, suggesting that these women were masculinized. |
|
|
The developmental instability theory suggests |
that some fetuses may be vulnerable to environmental and genetic stressors which cause disruptions of sexual differentiation independantly of sex hormones (see Chapter 3). These prenatal stressors could include maternal illness, infection, drug use or exposure to environmental chemicals (teratogens) such as pesticides. |

