![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
16 Cards in this Set
- Front
- Back
|
Acid base titration: what's in the burette? |
Base |
|
|
Acid-base titration: which graphs have the flick? |
Weak acid |
|
|
What affects the redox potential for a transition metal ion changing from a higher to a lower oxidation state? |
pH The ligand |
|
|
Salt bridge salt |
Saturated potassium nitrate |
|
|
Test for sulfate ions |
Add acidified barium chloride White ppt of BaSO4 |
|
|
Haem is a ______ complex with a _______ ligand. What is haemoglobin? How does it work and what is the problem? |
Iron(II) Multidentate A nitrogen in a globin complex acts as a ligand with the iron. Oxygen forms a coordinate bond to the iron (II) which allows it to be transported in the the blood. Bond is weak so oxygen can be given up to cells CO replaces the oxygen as it's a better ligand. Bonds irreversibly so destroys the haemoglobin's oxygen-carrying capacity |
|
|
Colour of solid sulfur |
Yellow |
|
|
Hydrogen sulfide observation |
Bad egg smell |
|
|
HI observation |
Steamy fumes |
|
|
Iodine observation |
Black solid |
|
|
Sulfur dioxide colour |
Colourless |
|
|
Bromine observation |
Brown fumes |
|
|
Method for silver nitrate test |
Dilute nitric acid to remove soluble carbonate or hydroxide impurities A few drops of silver nitrate solution |
|
|
Alternative to direct chlorination of a swimming pool |

|
|
|
Chlorine and cold, dilute NaOH |
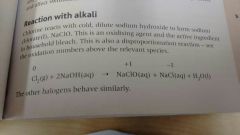
|
|
|
Tollens' Reagent formula What is it reduced to? |
[Ag(NH3)2]+ Metallic silver |

