![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
65 Cards in this Set
- Front
- Back
|
Mass number and Atomic number of an element
|
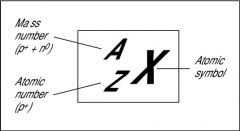
|
|
|
quantum #s: electron address
n l m(l) m(s) |
n: 1...infinity. aka energy level
l: 0...(n-1) subshell (e.g. s,p,d,f) m(l): -l....+l orientation m(s): +1/2 or -1/2 up or down |
|
|
Quantum Numbers
|

|
|
|
Electron Configuration exceptions
|
those elements located at d4 and d9 want to make orbitals half-full or full.
e.g. Cr [Ar]4s1 3d5 Cr, Cu, Mo, Ag, Au |
|
|
Paramagnetic
|
when there is unpaired electrons
odd # e- always paramagnetic attracted to magnetic field |
|
|
Diamagnetic
|
when electrons are paired at each orbital
very slight repulsion to magnetic field even # e-....either para or diamagnetic. |
|
|
How many electrons per shell?
|
2n^2 e-/shell
|
|
|
alpha decay
|
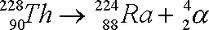
alpha = helium
only Z>83 |
|
|
beta- decay
|
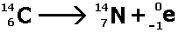
n to p...because n/p >> 1
|
|
|
beta+ (positron) decay
|
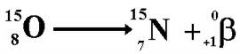
p to n...N/Z << 1
|
|
|
Electron capture
|
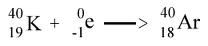
p to n...N/Z << 1
|
|
|
gamma decay
|
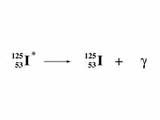
|
|
|
mass defect
|
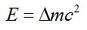
mass is less than the sum. Iron 56 has highest Nuc binding energy.
E/nucleon is proportional to ∆mass/nucleon |
|
|
Unstable Nuclei
|
Odd # of protons and/or neutrons
|
|
|
Stable Nuclei
|
N/Z ~ 1 and Z < 20
magic #s for N or Z: 2, 8, 20, 50, 82, 126 |
|
|
Effective Nuclear Charge (Zeff)
Zeff = group # |
Generally speaking, effective nuc. charge is charge felt by the valence electrons after you have taken into account the number of shielding electrons that surround the nucleus.
Example: Lithium, three protons and electron config. 1s2 2s1. The e- in 2s orbital is shielded from full attraction of the protons by the e- in the 1s orbital. Thus, Z* felt by the 2s electron should be one rather than three. |
|
|
Atomic Radius
|
down and to the left.
Na > Na+ Cl- > Cl |
|
|
Ionization Energy
|
up and right.
energy required to remove 1e-. 2nd e- is much harder to remove. (Endothermic) d5 and d10 Elements are more stable. |
|
|
Electron Affinity
|
increase from left to right.
Energy change associated with gaining an e- (Exothermic) |
|
|
Electronegativity
|
up and right.
smaller radius = higher e- neg. |
|
|
Bonding
|
Ionic
(strongest, meta/nonmetal, high m.p., high b.p.) Metallic (metal/metal, malleable, ductile) Covalent (nonmetal/nonmetal, lower m.p, b.p.) |
|
|
Covalent
|
sharing pairs of e-
molecular: h2O network solid: diamond, graphite |
|
|
Formal Charge
|
Normal # V.E. - Actual # V.E.
|
|
|
Intermolecular Forces
|
Hydrogen Bonding (strongest of Inter's)
Dipoles London Dispersion/Van der Waals |
|
|
Branching of alkanes
|
Sandwiches
decreases b.p. increases m.p. |
|
|
Longer Alkane (effects on b.p.)
|
longer carbon chain increases SA, greater london dispersion force
therefore increases b.p. |
|
|
Phase Diagram
|
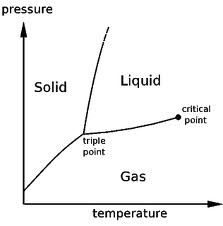
|
|
|
Phase Diagram for CO2
|
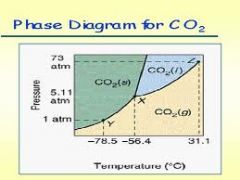
1atm is lower than the triple point. This is why it sublimes (solid to gas)
|
|
|
Phase Diagram for H2O
|
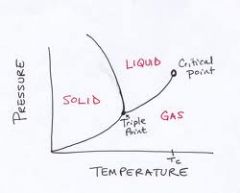
This explains why ice floats (ice is less dense than liquid water).
|
|
|
Calorimetry
|
q = mc∆T
q = heat change m = mass c = specific heat ∆T = Tf - Ti |
|
|
Phase Transitions
|
s-l-g (H>0, S>0)
fusion, sublimation, vaporization g-l-s (H<0, S<0) condensation, deposition, crystallization |
|
|
Ideal Gas
|
Each gas molecule has no volume.
No intermolecular forces. All collisions are elastic. KE ∝ T 1atm = 760 torr = 760 mmHg STP = 0∘C, 1atm 1 mole gas = 22.4L |
|
|
Ideal Gas Law
|
PV = nRT
|
|
|
Dalton's Law of Partial Pressure
|

P1 = X1 Ptot
|
|
|
Graham's Law of Effustion
|
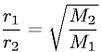
Rate is proportional to velocity, lighter gas has faster veloctiy
M = molar mass |
|
|
Combined Gas Law
|
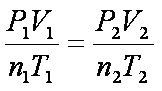
P ∝ 1/V
P ∝ T V ∝ T |
|
|
Solubility Rules
|
1) All Group I metal, NH4+, and NO3- salts are soluble
2) Most Ag+, Pb2+, and Hg₂2+ salts are insoluble |
|
|
Freezing Pt. Depression
|
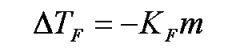
m is number of ions, Ba(OH)2 has 3 ions when dissociated
|
|
|
Vapor Pressure Depression (Raoult's Law)
|
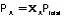
|
|
|
Boiling pt. Elevation
|
|
|
|
Boiling pt. Elevation
|
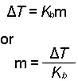
|
|
|
Osmotic Pressure
|
water moves across membrane from (low solute) high water conc. to low conc.
pie = MRT or P = (n/V)RT |
|
|
Catalyst
|
Speeds up reaction by lowering Ea.
Does not get used up. starts as reactant and ends up as product. (an intermediate is the reverse) |
|
|
Rxn Coordinate Diagram
Identify Transition State, Intermediates, delta H |
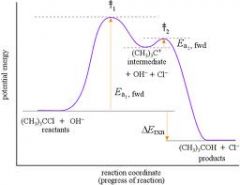
delta H < 0 because final is less than initial
|
|
|
Rate Law
|
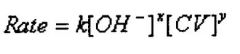
only reactants in rate law.
coefficients do not determine order. x, y exponents indicate order |
|
|
Common Ion Effect
|
increasing concentration of ion will decrease solubility
|
|
|
Le Chatelier's Principle
|
Add something to a rxn, the rxn will adjust to regain equilibrium.
In Exothermic (T is product), increasing T will cause a shift to LEFT increase in P = decrease in V increase V then shift to the side with more moles of gas |
|
|
Strong Acids
|
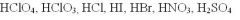
perchloric, chloric, hydrochloric....
|
|
|
Strong Bases
|
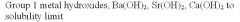
|
|
|
Binary Acid Trend (anything with H)
|
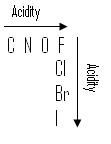
more e.n. the atom, stronger pull on H and makes it easier to give up H.
bigger the atom, longer and weaker the bond, therefore, easier to give up. |
|
|
OxyAcid Trend
|
everything else being equal, more Oxygen, more acidic.
more e.n., more acidic |
|
|
[H+] of WEAK acids
|
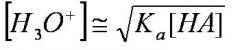
this equation is only for weak acids
|
|
|
3 LAWS of Thermodynamics
|
1. Conservation of Energy
2. ∆Suniv. > 0 3. S = 0 at 0K |
|
|
Systems: open, closed, isolated
|
open: exchange of heat, work, matter
closed: exchange of heat and work isolated: no exchange |
|
|
Neutral Cations
|
Conjugates of strong bases e.g. group 1A metals
all other cations are acidic |
|
|
Neutral Anions/Bases
|
Conjugates of strong acids e.g. Cl-, Br-, I-, NO3-, ClO4-, ClO3-
1 strong conjugate: HSO4- almost all other anions are bases |
|
|
Henderson-Hasselbalch equation (or buffer equation)
|
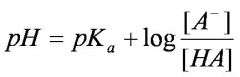
|
|
|
Bond Enthalpies
|
∆H = ∑Dbroken - ∑Dformed
D = bond bond breaking is endo bond making is exo (think alphabetical b to endo, m to exo) |
|
|
Enthalpies of Formation (Hess's Law)
|
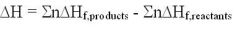
products or reactants in elemental state have ZERO formation values. e.g. Cl2, O2 etc.
|
|
|
∆G = ∆H - T∆S
(Get Higher Test Scores) |
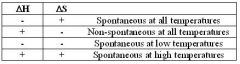
remember trends:
postive ∆S helps to go spont. negative ∆H helps to go spont. T amplifies S |
|
|
Electrochemical Cells (for all cells)
|
AN-ode OX-idation
RED-uction CAT-hode e- always flow from anode to cathode (because e- are lost at anode, gained at cathode, cathode gains mass) salt bridge: anions flow to anode, cations to cathode |
|
|
Galvanic/Voltaic Cell
|
cathode +
anode - |
|
|
Electrolytic Cell
|
cathode -
anode + consumes energy: e- goes to neg. cathode (not spontaneous) |
|
|
Oxidation States
|
1) Elements in their elemental form are in the zero oxidation state.
2) Group 1 metals are +1 and Group 2 metals are +2. 3) Hydrogen is +1 except when bonded to metals (when it’s –1). 4) The most electronegative elements get their typical ox state. 5) The last element not assigned balances the charge of the compound/ion. |
|
|
Spontaneous Rxn's
|
∆G < 0
Ecell > 0 Q < K |

