![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
17 Cards in this Set
- Front
- Back
|
Synthesis and function of NO?
|
No is synthesized from Arginine via the enzyme NO synthase (NOS).
It goes on to bind to the Iron on cGMP which causes it's activation, activating cGMP-dependant Protein Kinases (PKGs). This phosphorylates substrates. |
|
|
Synthesis and function of CO?
|
This is generated via heme oxygenase (HO), which catalyzes the conversion of heme to biliverdin (bilirubin precursor).
It goes on to bind to the Iron on cGMP which causes it's activation, activating cGMP-dependant Protein Kinases (PKGs). This phosphorylates substrates. |
|
|
Synthesis and function of growth factor neurotransmitters (BDNF & NGF)?
|
These are produced from the soma and dendrites and are released from soma, dendrites, and nerve terminals.
They not only play a neurotransmitter roll, they can also have a response on neighboring neuronal cells. |
|
|
What are the major Cholinergic neuronal pathways?
|
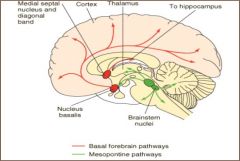
1. Basal Forebrain neurons
important for cortical activation & memory processing; degenerate in Alzheimer's Disease; basis for the use of cholinesterase inhibitors and nicotinic agonists for A.D. 2. Intrastriatal neurons control of posture and movement; activity inhibited by nigrostriatal DA input; basis for the adjunctive use of anticholinergics for some individuals with parkinson's disease. 3. brainstem (mesopontine tegmental) neurons activate thalamocortical circuits involved in arousal & REM; innervate cerebellum |
|
|
What are the major dopaminergic neuronal pathways?
|
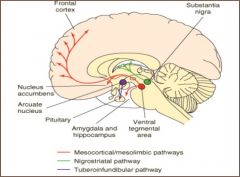
nigrostriatal neurons - control of posture and movement; degenerate in Parkinson's Disease; basis for the use of l-dopa for the treatment of P.D.
VTA projections (mesolimbic/mesocortical) - important for complex target-oriented behaviors; thought to be hyperactive in psychotic behaviors, basis for the use of DA receptor antagonists for treatment; VTA-NA pathway represents reward pathway, important for drug abuse/reinforcement tuberoinfundibular (hypothalamic) neurons - controls pituitary function; stimulation decreases prolactin secretion |
|
|
How does amphetamines affect the uptake-1 transporter of norepinephrine?
|
It reverses the transporter causing Norepinephrine to be pumped put of the axon terminal at a faster rate then vesicle fusion with presynaptic membrane.
|
|
|
What are the major noradrenergic neuronal pathways?
|
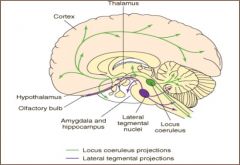
cell bodies originate in locus coeruleus in dorsal pons - widespread innervation to brain and spinal cord; controls responses to external sensory and motor stimuli; arousal & attention; may play role in learning & memory; important role in depression, basis for use of NE reuptake inhibitor antidepressants
cell bodies originate in lateral tegmental nucleus - extensive autonomic and neuroendocrine connections/regulation |
|
|
What are the major serotonergic neuronal pathways?
|
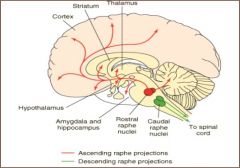
cell bodies originate in raphe nucleus - telencephalic & diencephalic pathways; broad influences throughout brain
projections to cerebral cortex important for sensory processing; possible role in schizophrenia; involved in the action of hallucinogens; basis for the use of 5-HT receptor antagonists for treatment of psychotic disorders input to hypothalamus important for homeostasis role in depression, eating behaviors and obsessive- compulsive disorder (OCD); basis for use of 5-HT reuptake inhibitors for depression and OCD |
|
|
How is histamine synthesized and inactivated?
|
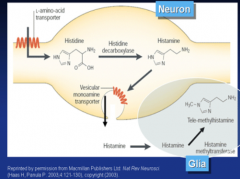
Histamine is synthesized from histidine via the enzyme histidine decarboxylase.
Histamine is catabolized via the enzyme histamine methyltransferase, which puts a methyl on the histamine ring. post-synaptic receptors are GPCRs: H1-H4 |
|
|
What are the major histaminergic neuronal pathways?
|
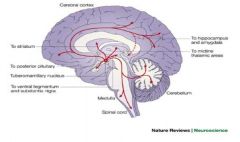
cell bodies originate in the lateral tuberomammillary nucleus of the hypothalamus - widespread pathways throughout the CNS; may play important role in arousal mechanisms, in coupling neuronal activity with cerebral metabolism, and in neuroendocrine regulation; basis why histamine receptor antagonists (antihistamines) lead to sedation
|
|
|
How is Gaba synthesized and inactivated?
|
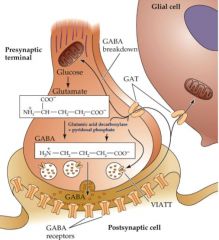
Gaba is synthesized from glutamate to Gaba by the enzyme glutamic acid decarboxylase (GAD) and pyridoxal phosphate.
terminated by reuptake by Na+ dependent Gaba transporters (GAT), then catabolized in neurons/astrocytes by GABA-T (GABA transaminase). GABAa & GABAc are LGIC GABAb is a GPCR and is coupled to Gi/o (inhibitory). |
|
|
What are the major GABA neuronal pathways?
|
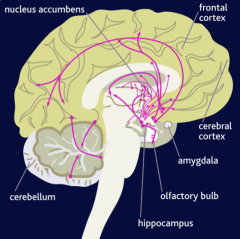
widespread throughout the CNS
represent nearly 40% of all neurons in the brain inhibitory in nature play important role in seizures, anxiety, & insomnia; basis for use of drugs that enhance GABA activity for the treatment of these disorders plays a role in pain |
|
|
How is Glutamate synthesized and inactivated?
|
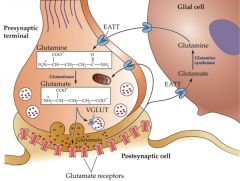
synthesized from glucose via Krebs cycle or from glutamine via glutaminase.
action terminated by reuptake in neurons & astrocytes by excitatory amino acid transporter (EATT) NMDA, AMPA, and kainate receptors are LGIC (ionotropic). NMDA receptor may mediate the toxic effect of glutamate. mglu1 - mglu8 are GPCR (metabotropic) |
|
|
What are the major Glutamate neuronal pathways?
|
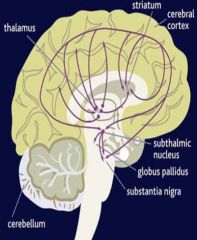
widespread throughout the CNS
mediate fast excitation in CNS; important role in seizures role in synaptic plasticity, learning and memory NMDA receptor antagonists may be neuroprotective, basis for use in A.D. & P.D.; may play role in schizophrenia |
|

|

|
|

|

|
|
|
Symptoms / Disorders chart
|

|

