![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
14 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
Compared to the mass and the penetrating power of an alpha particle, a beta particle has |
Less mass and greater penetrating power |
Table O |
|
|
What forma when carbon -14 decays? |
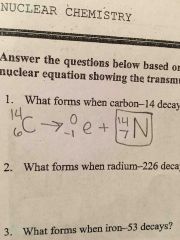
Back (Definition) |
Use table N for reference how long |
|
|
How long will it take for 30g of 222Rn to decay to 7.5g? |
30->15->7.5 2(3.823d) 7.646d |
How long= time and 222Rn= table N |
|
|
For any element, an unstable isotope is called |
Radioisotopes |
Radio |
|
|
When a nucleus is unstable it is a ________ substance that will ______ break down and release radiation |
Radioactive; spontaneously |
U listen to the ___ when in the car; kinda starts with the word spoon but its a compliment |
|
|
Beta decay |
Moves faster and are more penetrating then a alpha particle. |
Running fast |
|
|
The nuclear charge (the atomic number) is ___ by _ |
Increased by 1 |
The opposite of decrease;_234 |
|
|
Positron decay |
Is the anti-matter of an electron, so it is a "positive electron" |
|
|
|
Fission |
Spitting of a radioisotope (usually U-235); produces a great amount of energy; releases radiation |
When a couple breaks up with someone they ___;makes a good... ; another word for lets go |
|
|
Fusion |
Joining of 2 + nucleari; releases great amount of energy but no radiation |
|
|
|
During a nuclear reaction, mass is converted into |
Energy |
|
|
|
Which equation represents natural transmutation? |
14 14 0 6 C---> 7 N + -1 e |
|
|
|
Compare the atomic masses of nuclides used in fusion to the atomic masses of nuclides used in fission. |
Fusion particles are lighter |
|
|
|
State one potential benefit of using nuclear fusion instead of the current use of nuclear fission to produce electricity. |
The fusion process produces less radioactive waste |
|

