![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
121 Cards in this Set
- Front
- Back
- 3rd side (hint)
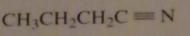
name:
|
butanenitrile
propyl cyanide |
16.2.a
|
|
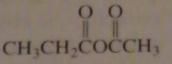
name:
|
ethanoic propanoic anhydride
acetic propionic anhydride |
16.2.b
|
|
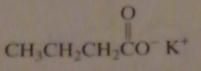
name:
|
potassium butanoate
potassium butyrate |
16.2.c
|
|
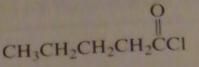
name:
|
pentanoyl chloride
valeryl chloride |
16.2.d
|
|
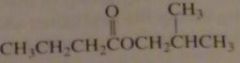
name:
|
isobutyl butanoate
isobutyl butyrate |
16.2.e
|
|
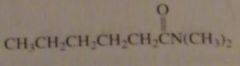
name:
|
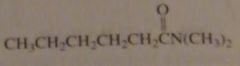
N,N-dimethylhexanamide
|
16.2.f
|
|
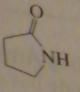
name:
|
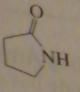
gamma-butyrolactam or
2-azacyclopentanone |
16.2.g
|
|
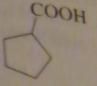
name:
|
cyclopentanecarboxylic acid
|
16.2.h
|
|
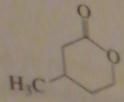
name:
|
Beta-methyl-delta-valeroactone or
|
16.2.i
|
|
|
structure of phenyl acetate
|
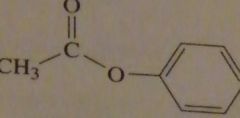
|
16.3.a
|
|
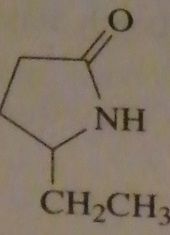
|
structure gamma-caprolactam
|
|
16.3.b
|
|
|
structure butanenitrile
|
.
|
16.3.c
|
|
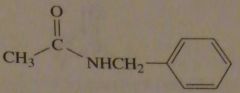
|
structure N-benzylethanamide
|
|
16.3.d
|
|
|
structure gamma-methylcaproic acid
|
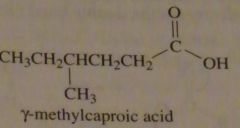
|
16.3.e
|
|
|
structure ethyl 2-chloropentanoate
|
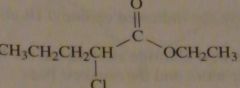
|
16.3.f
|
|
|
structure beta-bromobutyramide
|
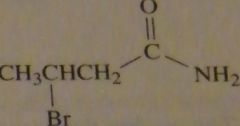
|
16.3.g
|
|
|
structure propanoic anhydride
|
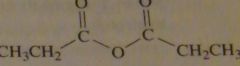
|
16.3.h
|
|
|
structure cycloheznecarbonyl chloride
|
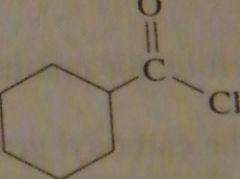
|
16.3.i
|
|
|
Which is longer, the carbon-oxygen single bond in a carboxylic acid or the carbon-oxygen bond in an alcohol? Why?
|
The carbon-oxygen and the methyl group is the longest because it is a pure single bond, whereas the other two carbon-oxygen bonds have some double-bond character.
|
16.4
|
|
|
There are three carbon=oxygen bonds in methyl acetate. What are their relative lengths? What are the relative IR stretching frequencies of these bonds?
|
The bond between oxygen and the methyl group is the longest because it is a pure single bond, whereas the other two carbon-oxygen bonds have some double-bond character. The bond between carbon and the carbonyl oxygen is the shortest because it has the most double bond character. Notice that the longer the bond the lower its IR stretching frequency. ADD PICTURE.
|
16.5
|
|
|
match the following compound with the carbonyl IR absorbtion band
acyl chloride 1800 & 1750 acid anhydride 1640 ester 1730 amide 1800 |
acyl chloride 1800
acid anhydride 1800 & 1750 ester 1730 amide 1640 |
16.6
|
|
|
The pK_a oh HCl is -7; the pK_a of H2O is 15.7. What is the product of the reaction of acetyl chloride with HO-?
|
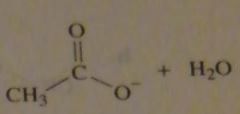
.
|
16.7.a
|
|
|
The pK_a oh NH_3 is 36; the pK_a oh H2O is 15.7. What is the product of the reaction of acetamide with HO-?
|
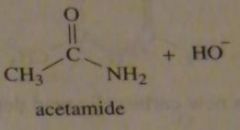
|
16.7.b
|
|
|
T/F If the newly added grouop in the tetrahedral intermediate is a stronger base than the group attached to the acyl group in the reactant, formation of the tetrahedral intermediate is the rate-limiting step of a nucleophile acyl substitution reaction.
|
true, long explanation
|
16.8
|
|
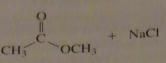
use the pK_a values to predict the product. (maybe put the value)
|
no reaction
|
16.9.a
|
|
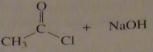
use the pK_a values to predict the product. (maybe put the value)
|
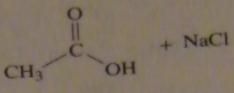
|
16.9.b
|
|
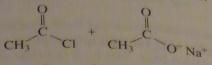
use the pK_a values to predict the product. (maybe put the value)
|
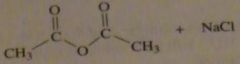
|
16.9.c
|
|
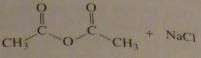
use the pK_a values to predict the product. (maybe put the value)
|
no reaction
|
16.9.d
|
|
|
What will be the product of a nucleophilic acyl substitution reaction- a new carboxylic acid derivative, a mixture of two carboxylic acid derivatives, or no reation - if the new group in the tetrahedral intermediate is the following?
a) a stronger base than the group that was already there b) a weaker base was already there than the group that was already there c)similar in basicity to the group that |
a) a new carboxylic acid derivative
b) NR c) a mixture of two carboxyl acid derivatives |
16.10
|
|
|
We have seen that it is necessary to use excess amine in the reaction of an acyl chloride with an amine. Explain why it is not necessary to use excess alcohol in the reaction of an acyl chloride with an alcohol
|
A protonated amine has a pK_a ~11. Therefore, the amine will be protonated by the acid that is produced in the reaction, and a protonated amine is not a nucleophile. Excess amine is used in order to have some unprotonated amine available to react as a nucleophile.
|
16.12
|
|
|
what is the mechanism for the reaction of acetyl chloride with water to form acetic acid.
|

|
16.13.a
|
|
|
what is the mechanism for the reaction of acetyl bromide with excess methylamine to form N-methylacetamide
|
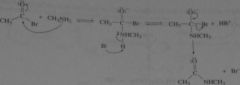
|
16.13.b
|
|
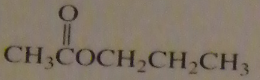
If you were to start with acetyl chloride what nucleophile would you use to make this compound?
|
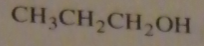
|
16.14.a
|
|

If you were to start with acetyl chloride what nucleophile would you use to make this compound?
|
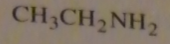
|
16.14.b
|
|
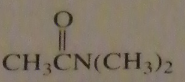
If you were to start with acetyl chloride what nucleophile would you use to make this compound?
|
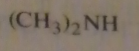
|
16.14.c
|
|
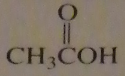
If you were to start with acetyl chloride what nucleophile would you use to make this compound?
|
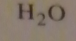
|
16.14.d
|
|
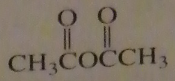
If you were to start with acetyl chloride what nucleophile would you use to make this compound?
|
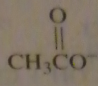
|
16.14.e
|
|
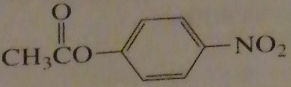
If you were to start with acetyl chloride what nucleophile would you use to make this compound?
|
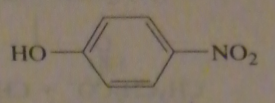
.
|
16.14.f
|
|
|
Propose a mechanism for the reaction of acetic anhydride with water. How does this mechanism differ from the mechanism for the reaction of acetic anhydride with an alcohol
|

same.
|
16.15
|
|
|
We have seen that acid anhydrides react with alcohols, water, and amines. In which one of these three reactions does th tetrahedral intermediate not have to lose a proton before it eliminates the carboxylate ion? explain
|

When an acid anhydride reacts with an amine, the tetrahedral intermediate does not have to lose a proton before it eliminates the carboxylate ion, because the carboxylate ion is a weaker base ( conj acid ~5) than an amine (conj acid ~10)
|
16.16
|
|
|
State three factors that contribute to making the noncatalyzed hydrolyssis of an ester a slow reaction. Which is faster, hydrolysis or aminolysis of an ester?
|
1. the carbonyl group of an ester is a weak electrophile
2.Water is a weak nucleophile 3. -OCH3 is a strong base and, therefore, a poor leaving group aminolysis is faster becasue an amine is a better nucleophile than water |
16.18
|
|

List the following esters in order of decreasing reactivity toward hydrolysis:
|

|
16.19
|
|
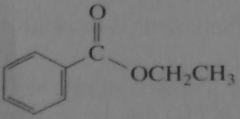
product of acid-catalyzed hydrolysis of the ester
|
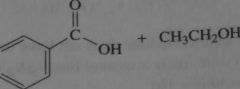
|
16.20.a
|
|
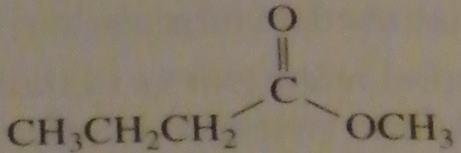
product of acid-catalyzed hydrolysis of the ester
|
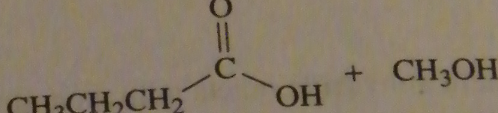
|
16.20.b
|
|
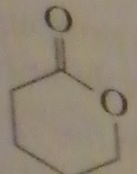
product of acid-catalyzed hydrolysis of the ester
|
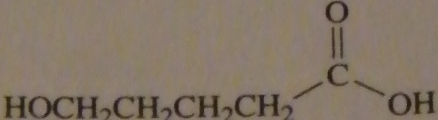
|
16.20.c
|
|
|
mechanism for the acid catalyzed reaction of acetic acid and methanol to form methyl acetate. Use HB+ and B to represent proton-donating and proton-removing species, respectively.
|
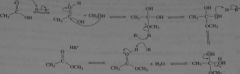
opposite of acid-catalyzed hydrolysis of methyl acetate
|
16.21
|
|
|
In the mechanism for the acid-catalyzed hydrolysis of an ester:
a) what species could be represented by HB+? b) what species could be represented by B? c) what species is HB+ most likely to be in a hydrolysis reaction? d) what species is HB most likely to be in the reverse reaction? |
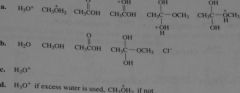
|
16.22
|
|
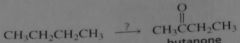
synthesis of butanone from butane
|
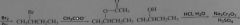
|
16.23
|
|
|
write the mechanism for the acid-catalyzed transesterification reaction of methyl acetate with ethanol
|

|
16.24
|
|
|
What species other than an acid can be used to increase the rate of a transesterification reaction that converts methyl acetate to propyl acetate? explain why the rate of aminolysis of an ester cannot be increased by H+, OH-, or RO-
|
The conjugate base of the reactant alcohol can be used to catalyze the reaction. If h+ is used as a catalyst, the amine will be protonated in the acidic solution and, therefore, will not be able to react as a nucleophil.
If HO- is used as a catalyst, HO- will be the strongest nucleophile present in solution, so it will attack the ester, and the product of the reaction will be a carboxylic acid rather than an amide If RO- is used as a catalyst, RO- will be the nucleophile, and the product of the reaction will be an ester rahter than an amide. |
16.25
|
|
|
An oil obtained from coconuts is unusual in that all three fatty acid components are identical. The molecular formula of the oil is C45 H86 O6. What is the molecular formula of the carboxylate ion obtained when the oil is saponified?
|
When the oil is saponified, it forms glycerol and three equicalents of carboxylate ion. In losing glycerol, the fat loses three carbons and five hydrogens. Thus, the three equivalents of carboxylate ion have a combined molecular formula of C42 H81 O6. Dividing by three gives the mol formula C14 H27 O2
|
16.29
|
|
|
Show how methyl butyrate (odor of apples) could be prepared using carboxylic acid as one of the starting materials.
|
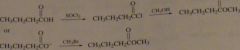
.
|
16.30.a
|
|
|
Show howoctyl acetate(odor of oranges) could be prepared using carboxylic acid as one of the starting materials.
|
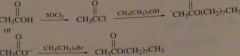
|
16.30.b
|
|
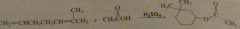
Propose a mechanism for the following reaction.
|
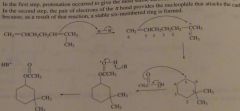
|
16.31
|
|
|
What acyl chloride and what amine would be required to synthesize N-ehtylbutanamide.
|
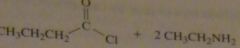
|
16.32.a
|
|
|
What acyl chloride and what amine would be required to synthesize N,N-dimethylbenzamide
|
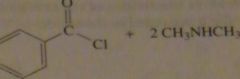
|
16.32.b
|
|

will this lead to the formation of an amide?
|
no
|
16.33.a
|
|
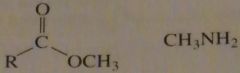
will this lead to the formation of an amide?
|
If the nucleophile were -CH3NH instead of CH3NH the better nucleophile will increase the rate of amide formation
|
16.33.b
|
|
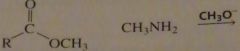
will this lead to the formation of an amide?
|
no
|
16.33.c
|
|
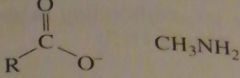
will this lead to the formation of an amide?
|
no
|
16.33.d
|
|
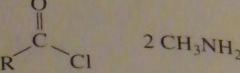
will this lead to the formation of an amide?
|
use a large excess of methylamine to force the equilibrium to the right
|
16.33.e
|
|
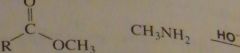
will this lead to the formation of an amide?
|
no
|
16.33.f
|
|
|
Explain why methicillin is effective in treating patients infected with bacteria resistant to penicillin G.
|
Because methicillin is missing the methylene group, its benzene ring is closer to the rou-membered ring. This plus the bulky methoxy groups may prevent penicillinase from being able to bind it at its active site, therefore, it would not be able to be used as a substrate. Alternatively, the extra steric hindrance associated with methicillin may prevent water from attacking the carbonyl group in order to hydrolyze.
|
16.35
|
|
|
List the following amides in order of decreasing reactivity toward acid-catalyzed hydrolysis. EDIT PICTURE
|
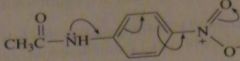
C>B>D>A
The relative reactivities of the amides depend on the basicities of their leaving groups: the weaker the base, the more reactive the amide. A p-nitro-substitued aniline is less basic than a m-substituted aniline because when the nitro group is in the para position, electrons can be delocalized onto the nitro group. |
16.36
|
|
|
what alkyl bromide would you use in a gabriel synthesis to prepare pentylaime
|
pentyl bromide
|
16.37.a
|
|
|
what alkyl bromide would you use in a gabriel synthesis to prepare isohexylamine
|
isohexyl bromide
|
16.37.b
|
|
|
what alkyl bromide would you use in a gabriel synthesis to prepare benzylamine
|
benzyl bromide
|
16.37.c
|
|
|
what alkyl bromide would you use in a gabriel synthesis to prepare cyclohexylamine
|
cyclohexyl bromide
|
16.37.d
|
|
|
which alkyl halide will form butyric acid after reaction with sodium cyanide followed by heating the product in an acidic aqueous solution?
|
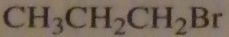
|
16.39.a
|
|
|
which alkyl halide will form isovaleric acid after reaction with sodium cyanide followed by heating the product in an acidic aqueous solution?
|
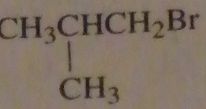
|
16.39.b
|
|
|
which alkyl halide will form cyclohexanecarboxylic acid after reaction with sodium cyanide followed by heating the product in an acidic aqueous solution?
|
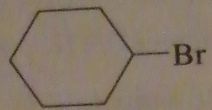
|
16.39.c
|
|
|
synthesis using an intramolecular reactoin
|

|
16.40.a
|
|
|
synthesis using an intramolecular reactoin
|

|
16.40.b
|
|
|
synthesis using an intramolecular reactoin
|

|
16.40.c
|
|
|
synthesis using an intramolecular reactoin
|
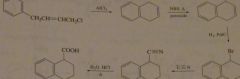
|
16.40.d
|
|
|
synthesis using an intramolecular reactoin
|

|
16.40.e
|
|
|
synthesis using an intramolecular reactoin
|

|
16.40.f
|
|
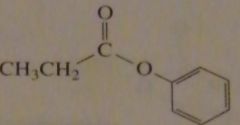
synthesize the following compound from a carboxylic acid
|

|
16.41.a
|
|
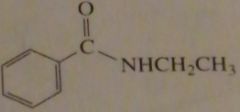
synthesize the following compound from a carboxylic acid
|

|
16.41.b
|
|
|
structure of N,N-dimethylhexanamide
|
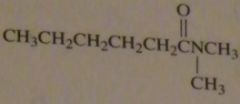
|
16.45.a
|
|
|
structure of 3,3-dimethylhexanamide
|
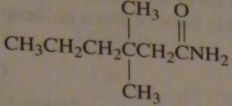
|
16.45.b
|
|
|
structure of cyclohexanecarbonyl chloride
|
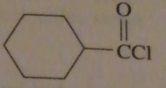
|
16.45.c
|
|
|
structure of propanenitrile
|
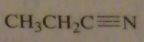
|
16.45.d
|
|
|
structure of propionyl bromide
|
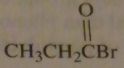
|
16.45.e
|
|
|
structure of sodium acetate
|
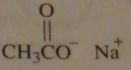
|
16.45.f
|
|
|
structure of benzoic anhydride
|
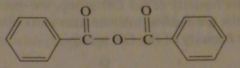
|
16.45.g
|
|
|
structure of Beta-Valerolactone
|
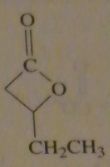
|
16.45.h
|
|
|
structure of 3-methylbutanenitrile
|
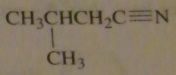
|
16.45.i
|
|
|
structure of cycloheptanecarboxylic acid
|
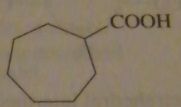
|
16.45.j
|
|
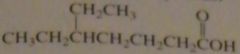
name:
|
5-ethylheptanoic acid
|
16.46.a
|
|
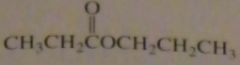
name:
|
propyl propanoate
propyl propionate |
16.46.b
|
|
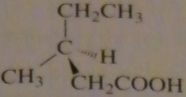
name:
|
(s)-3-methylpentanoic acid
(s)-beta-methylvaleric acid |
16.46.c
|
|
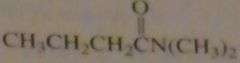
name:
|
N,N-dimethylbutanamide
N,N-dimethylbutyramide |
16.46.d
|
|
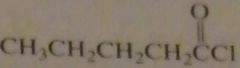
name:
|
pentanoyl chloride
valeryl chloride |
16.46.e
|
|
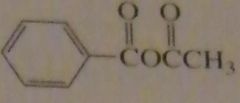
name:
|
acetic benzoic anhydride
|
16.46.f
|
|
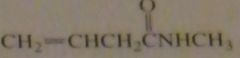
name:
|
N-methyl-3-butenamide
|
16.46.g
|
|
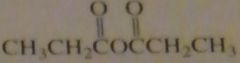
name:
|
propanoic anhydride
propionic anhydride |
16.46.h
|
|
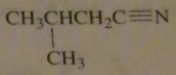
name:
|
(R)-3-methylhexanenitrile
|
16.46.i
|
|
|
products of reaction between benzoyl chloride with sodium acetate
|
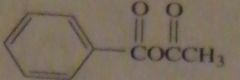
|
16.47.a
|
|
|
products of reaction between benzoyl chloride with water
|
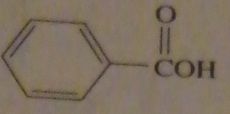
|
16.47.b
|
|
|
products of reaction between benzoyl chloride with excess dimethylamine
|
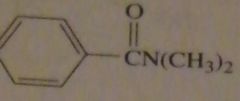
|
16.47.c
|
|
|
products of reaction between benzoyl chloride with aqueous HCl
|
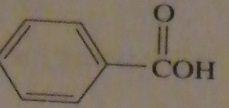
|
16.47.d
|
|
|
products of reaction between benzoyl chloride with aqueous NaOH
|
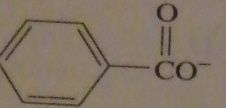
|
16.47.e
|
|
|
products of reaction between benzoyl chloride with cyclhexanol
|
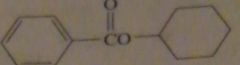
|
16.47.f
|
|
|
products of reaction between benzoyl chloride with excess benzylamine
|

|
16.47.g
|
|
|
products of reaction between benzoyl chloride with 4-chlorophenol
|
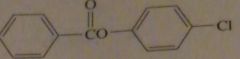
|
16.47.h
|
|
|
products of reaction between benzoyl chloride with isopropyl alcohol
|
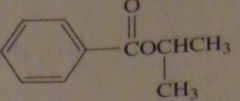
|
16.47.i
|
|
|
products of reaction between benzoyl chloride with excess aniline
|
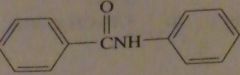
|
16.47.j
|
|
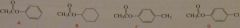
List the following esters in order of decreasing reactivity in the first slow step of a nucleophilic acyl substitution reaction (formation of intermediate)
|

The weaker the base attached to the acyl gropu, the easier it is to form the tetrahedral intermdiate. (p-chlorophenol is a stronger acid than phnol so the conjugae base of p-chlorophenol is a weaker base than the conjegate base of phenol, etc)
|
16.48.a
|
|
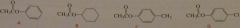
List the following esters in order of decreasing reactivity in the second slow step of a nucleophilic acyl substitution reaction (collapse of intermediate)
|
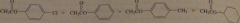
The tetra hedral intermediate collapses by eliminating the OR group of the tetrahedral intermediate. The weaker the basicity of the OR group, the easier it is to eliminate it. Thus, the rate of both formation of the tetrahedral intermediate and collapse of the tetrahedral intermediate is decreased by increasing the basicity of the OR group.
|
16.48.b
|
|
|
Because bromocyclhezen is a secondary alkyl halide, both cyclhexanol and cyclohezene are formed when the alkyl halide reacts with hydroxide ion. Suggest a method to synthesize cyclhexanol from bromocyclohexane that would form little or no cyclohexene.
|
Because acetate ion is a weak bease, the SN2 reaction will form only a substitution product. The product of the SN2 reaction is an ester which, when hydrolyzed, forms cyclohexanol and acetic acid
|
16.49
|
|

Which compound would you expect to have a higher dipole moment, methyl acetate or butanone?
Which would you expect to have a higher boiling point? |
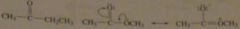
Methyl acetate has a resonance contributor that butanone does not have, and this resonance contributor cause methyl acetate to be more polar than butanone. Because butanone is less polar, it has the lower dipole moment.
Because it is more polar, the intermolecular forces holding methyl acetate molecules together are stronger, so we would expect methyl acetate ot have a higher boiling point. |
16.50
|
|

How could you use HNMR spectroscopy to distinghuish between the following esters?
|
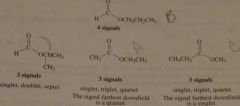
Propyl formate is easy to distinguish because it is the only ester that will show four signals. The other three esters show three signals. Isopropyl formate can be distinguished by its unique splitting pattern: a singlet, a doublet, and a septet. The splitting patterns of the other two esters are the same: a singlet, a triplet, and a quartet, They can be distinguisdhed because the highest frequency signal in ethyl acetate is a quartet, whereas the highest frequency signal in methyl propionate is a singlet.
|
16.51
|
|
|
If propionyl chloride is added to one equivalent of methylamine, only a 50% yield of N-methylpropanamide is obtained. If however acyl chloride is added to two equivalents of methylamine, the yield of N-methylpeopanamide is almost 100%. Explain these observations.
|
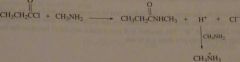
The reaction of methyl amine with propionyl chloride generates a proton that will protonate unreacted amine, thereby destroying its nucleophilicity. It two equivalents of CH3NH2 are used, one equivalent will remain unprotonated and be able to react with propionyl chloride to form N-methylpropanamide.
|
16.52
|
|

When a carboxylic acid is dissolved in isotopically labeled water, the label is incorporated into both oxygens of the acid. Propose a mechanism to account for this.
|
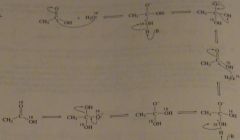
|
16.53.a
|
|

If a carboxylic acid is dissolved in isotopically labeled methanol and an acid catalyst is added, where will the label reside in the product?
|
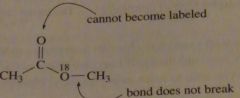
The carboxyl oxygen will be labeled. Only one isotpically labeled ocygen can be incorporated inthe ester because the bond between the methyl group and the labeled oxygen does not break, so there is no way for the carbonyl oxygen to become labeled.
|
16.53.b
|
|

If an ester is dissolved in isotopicall labeled water and an acid catalyst is added, where will the label reside in the product?
|
In the presence of an acid catalyst, the ester will be hydrolyzed to a carboxylic acid and an alcohol. Both oxygen atoms of the carboxylic acid will be labeled for the same reason that both oxygen atoms of the carboxylic acid are labeled in "a". The alcohol will not contain any label because the bond between the methyl group and the oxygen does not break.
|
16.53.c
|
|
|
synth propyl acetate (odor of pears) using 1) an alcohol 2) using an alkyl halide
|
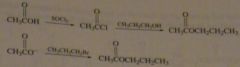
|
16.54.a
|
|
|
synth isopentyl acetate (odor of bananas) using 1) an alcohol 2) using an alkyl halide
|

|
16.54.b
|
|
|
synth ethyl butyrate (odor of pineapple) using 1) an alcohol 2) using an alkyl halide
|
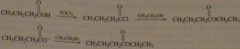
|
16.54.c
|
|
|
synth methyl phenylethanoate (odor of honey) using 1) an alcohol 2) using an alkyl halide
|
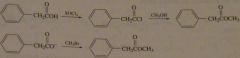
|
16.54.d
|

