![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
76 Cards in this Set
- Front
- Back
|
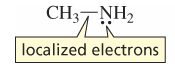
Localized Electrons |
electrons that are restricted to a particular locality |
|
|
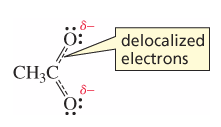
Delocalized Electrons |
electrons that are shared by more than two atoms (dashed lines in bonds) |
|
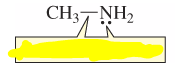
Are the above localized or delocalized electrons? |
|
|
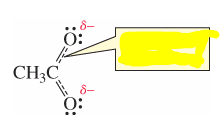
Are the above localized or delocalized electrons? |
|
|
|
Kekule proposed that benzene is a ring of ____ carbons with ___ double bonds, which oscillate between carbons so rapidly as to be indistinquishable. He was ultimately (correct/incorrect). |
Kekule proposed that benzene is a ring of six carbons with three double bonds, which oscillate between carbons so rapidly as to be indistinguishable. He was ultimately incorrect. |
|
|
Benzene is a (planar/nonplanar) molecule with (parallel/nonparallel) p orbitals that overlap on (one/two) side(s). |
Benzene is a planar molecule with parallel p orbitals that overlap on two sides. |
|
|
Benzene's carbons have a total of _____ p orbitals (___ above and ____ below the plane of the ring) in which each (pi/sigma) electron is shared by ____ carbons. |
Benzene's carbons have a total of 12 p orbitals (6 above and 6 below the plane of the ring) in which each pi electron is shared by six carbons. |
|
|
How should one draw benzene? |
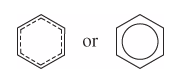
|
|
|
Resonance Contibutor |
AKA resonance structure or contributing resonance structure a structure with localized electrons that approximates the true structure of a compound with delocalized electrons |
|
|
Resonance Hybrid |
the actual structure of a compound with delocalized electrons; represented by two or more structures with localized electrons (resonance contributors) |
|
|
Electron delocalization is shown by ________ whereas equilibrium is shown by ________. |
Electron delocalization is shown by ↔whereas equilibrium is shown by ⇋. |
|
|
Resonance contributors are (real/imaginary) structures. Resonance hybrids are (real/imaginary) structures. |
Resonance contributors are imaginary structures. Resonance hybrids are real structures. |
|
|
Electron delocalization is most effective for (planar/nonplanar) molecules because this (maximizes/minimizes) the amount of overlap between p orbitals. |
Electron delocalization is most effective for planar molecules because this maximizes the amount of overlapt between p orbitals. |
|
|
Delocalized electrons result from a ___ orbital overlapping the ___ orbitals of ____ adjacent atoms. |
Delocalized electrons result from a p orbital overlapping the p orbitals of two adjacent atoms. |
|
|
List the rules for drawing resonance contributors. |
1. Only e- move; never atoms. 2. Only pi e- and lone-pair e- move. 3. Total number of e- does not change. Total charge does not change. |
|
|
To draw resonance contributors, move only ___ electrons or _____________ toward an sp__ carbon.
|
To draw resonance contributors, move only pi electrons or lone-pair electrons toward an sp2 carbon. |
|
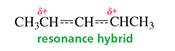
Draw the resonance contributors. |
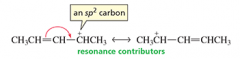
|
|
|
Delocalization makes a molecule (more/less) stable. |
Delocalization makes a molecule more stable. |
|
|
Electrons usually move (towards/away from) the most electronegative atom. |
Electrons usually move towards the most electronegative atom. |
|
|
All resonance contributors (do/do not) contribute equally to the resonance hybrid. |
All resonance contirbutors do not contribute equally to the resonance hybrid. |
|
|
The greater the predicted stability of a resonance contributor, the (more/less) it contributes to the structure of the resonance hybrid. |
The greater the predicted stability of a resonance contributor, the more it contributes to the structure of the resonance hybrid. |
|
|
The more a resonance contributor contributes to the structure of the resonance hybrid, the (more/less) similar the contributor is to the real molecule. |
The more a resonance contributor contributes to the structure of the resonance hybrid, the more similar the contributor is to the real molecule. |
|
|
Separated Charges |
a positive and negative charge that can be neutralized by the movement of electrons |
|
|
Resonance contributors with separated charges are relatively (unstable/stable) and thus (high/low) in energy. |
Resonance contributors with separated charges are relatively unstable and thus high in energy. |
|
|
Delocalization Energy |
the extra stability a compound achieves as a result of having delocalized electrons |
|
|
A resonance hybrid is (more/less) stable than any of its resonance contributors is predicted to be. |
A resonance hybrid is more stable than any of its resonance contributors is predicted to be. |
|
|
The greater the number of relatively stable resonance contributors, the (greater/smaller) the delocalization energy. |
The greater the number of relatively stable resonance contributors, the greater the delocalization energy. |
|
|
The more nearly equivalent the structures of the resonance contributors, the (greater/smaller) the delocalization energy. |
The more nearly equivalent the structures of hte resonance contributors, the greater the delocalization energy. |
|
|
Diene |
a hydrocarbon with two double bonds |
|
|
Isolated dienes have double bonds that are separated by (one/more than one) single bond. |
Isolated dienes have double bonds that are separated by more than one single bond. |
|
|
Conjugated dienes have double bonds that are separated by (one/more than one) single bond. |
Conjugated dienes have double bonds that are separated by one single bond. |
|
|
The most stable alkene has the (largest/smallest) heat of hydrogenation. |
The most stable alkene has the smallest heat of hydrogenation. |
|
|
An increase in delocalization energy means a(n) (increase/decrease) in stability. |
An increase in delocalization energy means an increase in stability. |
|
|
A conjugated diene is (more/less) stable than an isolated diene. |
A conjugated diene is more stable than an isolated diene. |
|
|
Allylic Carbon |
carbon adjacent to sp2 carbon of an alkene |
|
|
Benzylic Carbon |
carbon adjacent to sp2 carbon of benzene ring |
|
|
Allylic Cation |
carbocation with positive charge on allylic carbon |
|
|
Benzylic Cation |
carbocation with positive charge on benzylic carbon |
|
|
Allyl Cation |
an unsubstituted allylic cation |
|
|
Benzyl Cation |
unsubstituted benzylic cation |
|
|
Rank the following from most to least stable: Methyl cation Primary carbocation Tertiary carbocation Benzyl cation Secondary carbocation Allyl cation |
Benzyl cation > allyl cation > tertiary carbocation > secondary carbocation > primary carbocation > methyl cation |
|
|
The more stable the base, the (stronger/weaker) its conjugate acid. |
The more stable the base, the stronger its conjugate acid. |
|
|
A nearby electronegative atom stabilizes an anion by ___________________________________. |
A nearby electronegative atom stabilizes an anion by inductive electron withdrawal. |
|
|
Greater stability comes from a(n) (increase/decrease) in delocalization energy. |
Greater stability comes from an increase in delocalization energy. |
|
|
Which acids have a pK_a of the following: <0 : ~5 : ~10 : ~15 : |
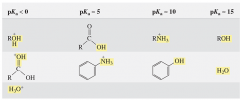
<0 : protonated alcohol, protonated water, protonated carbonyl groups ~5 : carboxylic acid, protonated aniline ~10 : protonated amine, phenol ~15 : alcohol, water |
|
|
Electron donation (increases/decreases) acidity. Electron withdrawal (increases/decreases) acidity. |
Electron donation decreases acidity. Electron withdrawal increases acidicty. |
|
|
Substituents with a lone pair on the atom attached to a benzene ring can localize electrons through __________________________________. |
Substituents with a lone pair on the atom attached to a benzene ring can localize electrons through donation of electrons by resonance. |
|
|
List the substituents that commonly donate electrons by resonance. |
NH2, OH, OR, Cl |
|
|
Substituents attached to a benzene ring that are doubly or triply bonded to a more electronegative atom can delocalize electrons through __________________________________________. |
Substituents attached to a benzene ring that are doubly or triply bonded to a more electronegative atom can delocalize electrons through withdrawal of electrons by resonance. |
|
|
List the substituents that commonly donate electrons by resonance. |
C=O, C≡N, SO3H, NO2 |
|
|
The more electron donating the substituent, the (more/less) it decreases the acidity of a COOH, an OH, or an +NH3 group attached to a benzene ring. |
The more electron donating the substituent, the more it decreases the acidity of a COOH, an OH, or an +NH3 group attached to a benzene ring. |
|
|
The more electron withdrawing the substituent, the (more/less) it increases the acidity of a COOH, an OH, or an +NH3 group attached to a benzene ring. |
The more electron withdrawing the substituent, the more it increases the acidity of a COOH, an OH, or an +NH3 group attached to a benzene ring. |
|
|
For electrophilic addition reactions, one can determine the major product by examining the (number of hydrogens/relative stabilities of individual carbocations). |
For electrophilic addition reactions, one can determine the major product by examining the relative stabilities of individual carbocations. |
|
|
1,2-Addition |
AKA direct addition addition to the 1- and 2-positions of a conjugated system |
|
|
A(n) (isolated/conjugated) diene undergoes only 1,2-addition. |
An isolated diene undergoes only 1,2-addition. |
|
|
1,4-addition |
AKA conjugate addition addition to the 1- and 4-positions of a conjugated system |
|
|
A(n) (isolated/conjugated) diene undergoes both 1,2- and 1,4-addition. |
A conjugated diene undergoes both 1,2- and 1,4-addition. |
|
|
What is the conjugated system? |
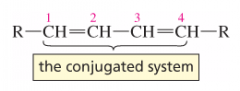
|
|
|
Diels-Alder Reaction |
![a [4+2] cycloaddition reaction in which a conjugated diene reactions with a carbon that contains a C=C](https://images.cram.com/images/upload-flashcards/96/31/65/20963165_m.png)
a [4+2] cycloaddition reaction in which a conjugated diene reactions with a carbon that contains a C=C |
|
|
Dienophile |
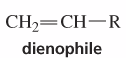
an alkene that reacts with a diene in a Diels-Alder reaction |
|
|
Carbons containing carbon-carbon (double/triple) bonds can be used as dienophiles to prepare compounds with two isolated double bonds. |
Carbons containing carbon-carbon triple bonds can be used as dienophiles to prepare compounds with two isolated carbon bonds. |
|
|
List the steps to determine the reactants needed to synthesize a Diels-Alder product. |
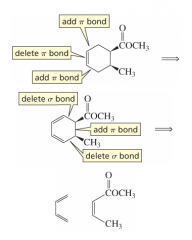
1. Locate double bond in project; delete that pi bond and add pi bonds on either side of it. 2. Delete the sigma bonds between the diene side of the cycloalkene and add a pie bond between the two carbons that just had a sigma bond deleted. |
|
|
Aromatic compounds (are/are not) particularly stable because they have (small/large) delocalization energies. |
Aromatic compounds are particularly stable because they have large delocalization energies. |
|
|
For a compound to be aromatic, it must be ____________ and __________, and it must have an (interrupted/uninterrupted) cloud of an (odd/even) number of pi electrons. |
For a compound to be aromatic, it must be cyclic and planar, and it must have an uninterrupted cloud of an odd number of pi electrons. |
|
|
Which sp-hybridized carbons have p orbitals? |
Only sp2 and sp carbons have p orbitals |
|
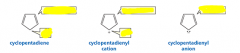
Which of these are aromatic? |
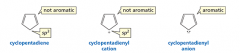
|
|
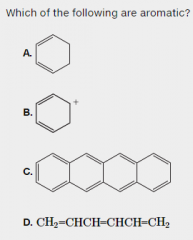
|
A is not: it does not have an uninterrupted cloud of pi electrons B is not: it does not have an odd number of pi electrons C is the only aromatic compound D is not: it is not cyclic |
|
|
Electrophilic Aromatic Substitution |
a reaction in which an electrophile substitutes for a hydrogen of an aromatic ring |
|
|
Aromatic substitution is (more/less) stable than nonaromatic substitution. |
Aromatic substitution is more stable than nonaromatic substitution. |
|
|
Describe the free energy graph of an electrophilic aromatic substitution and electrophilic addition. |
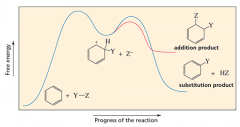
|
|
|
List the five most common electrophilic aromatic substitution reactions. |
1. Halogenation - Br, Cl, I 2. Nitration - NO2 3. Sulfonation - SO3H 4. Friedel-Crafts acylation - RC=O 5. Friedel-Crafts alkylation - R |
|
|
Describe the steps of the mechanism for electrophilic aromatic substitution.
|
1. Electrophile (Y+) adds to nucleophilic benzene ring, forming a carbocation intermediate. 2. Base in the mixture (:B) removes a proton (H+) from the carbocation intermediate and the electrons that held the proton move into the ring to re-establish aromaticity. H+ is always removed from the carbon that formed the bond with the electrophile. |
|
|
What substituents exhibit inductive electron withdrawal on alkene rings? |
Substituents which are more electron withdrawing (more electronegative) will exhibit inductive electron withdrawal. |
|
|
What substituents exhibit electron donation by hyperconjugation? |
Substituents that are alkyl substituents can donate electrons via hyperconjugation (donating electrons to empty p orbital(s)) |
|
|
What substituents exhibit electron donation by resonance? |
Substituents that have a lone pair on the atom direct attached to the benzene ring can delocalize that pair into the ring. |
|
|
What substituents exhibit electron withdrawal by resonance? |
Substituents that have the first atom doubly or triply bonded to a more electronegative atom than H can exhibit electron withdrawal by resonance. |

