![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
12 Cards in this Set
- Front
- Back
|
Unsaturation Number
|
U = C + 1 - ½ (H) + ½ (N)
U = unsaturation number H = number of hydrogens + halogens N = number of nitrogens + phosphorus |
|
|
Interpretation of Unsaturation Number-----0
|
no rings, no double bonds, or no triple bonds
|
|
|
Interpretation of Unsaturation Number-----1
|
1 ring or 1 double bond
|
|
|
Interpretation of Unsaturation Number-----2
|
2 double bonds, 2 rings, 1 double bond + 1 ring, or 1 triple bond
|
|
|
Interpretation of Unsaturation Number-----3
|
3 double bonds, 3 rings, 2 double bonds + 1 ring, 2 rings + 1 double bond, 1 triple bond + 1 double bond, or 1 triple bond + 1 ring
|
|
|
Interpretation of Unsaturation Number-4
|
benzene
|
|
|
Interpretation of Unsaturation Number-5
|
benzene + 1 ring or 1 double bond (4 + 1)
|
|
|
Interpretation of Unsaturation Number-6
|
benzene + 2 rings, 2 double bonds, 1 double bond + 1 ring, or benzene + 1 triple bond
|
|
|
Nomenclature of Alkenes
|
The longest continuous carbon chain containing the carbon-carbon double bond is the parent compound.
-Change “-ane” of alkane to “-ene.” -Number from whichever end that gives the double bond the lowest possible number. Give the location of the double bond. For cycloalkenes, the double bond is assumed to be in the 1 position. -Use –diene, -triene, and –tetraene, etc if multiple double bonds are present. -Name and number substituents. |
|
|
If alkenes are substituents, use the following
|
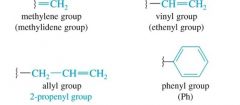
|
|
|
Cis and trans
|
cis – same group, same side
trans – same group, different sides |
|
|
E and Z – according to Cahn-Ingold-Prelog rules.
|

Z – high priority same side
E – high priority different sides |

