![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
133 Cards in this Set
- Front
- Back
|
base peak |
the highest peak in the mass spectrum where the ion has been cleaved at the benzylic carbon |
|
|
sodium acetylide reactions with carbonyls |
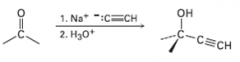
forms new carbon carbon bond |
|
|
Grignard reaction products |
he Grignard Reaction is the addition of an organomagnesium halide (Grignard reagent) to a ketone or aldehyde, to form a tertiary or secondary alcohol, respectively. The reaction with formaldehyde leads to a primary alcohol. |
|
|
Grignard Reaction with Nitriles |
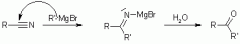
|
|
|
Grignard Reactions with CO2 |
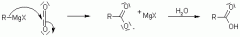
|
|
|
Grignard reactions with oxiranes |
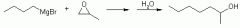
|
|
|
Smith Simmons Reagent |
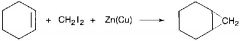
Reactions with alkenes to give cyclopropanes
stereochemistry is conserved |
|
|
Preperation of iodomethylzinc |
CH2I2 + Zn in diethyl ether with copper=ICH2ZnI |
|
|
Which way is R? |
Clockwise |
|
|
Which way is S? |
Counterclockwise |
|
|
Olefin metathesis |
redistribution of alkene fragmenets |
|
|
Applications of olefin metathesis |
ring opening metathesis ring closing metathesis cross metathesis |
|
|
Grubbs catalyst |
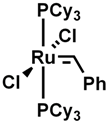
used in olefin metatheisis
|
|
|
RX + ZN(in diethyl ether)-RZnX |
Organozinc reactions |
|
|
What is iodomethylzinc used for? |
react w alekens to give cyclopropanes
takes double bond, splits and adds new bond to form triangle |
|
|
MgI |
Grignard reagent |
|
|
MgBr |
Grignard reagent |
|
|
Why are organometallic compounds useful? |
Because they have carbanionic character which makes them a source of nucleophilic carbons |
|
|
Reactivity of Grignard Reagents |
MgI>MgBr>MgCl |
|
|
lithium divinyl cuprate |
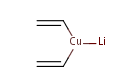
|
|
|
Gilman reagents |
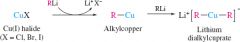
copper halide with 2 equivalents of alkyl or aryllithium
only one of the alk compounds stays on product |
|
|
benzyl group |
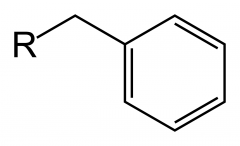
|
|
|
amine |
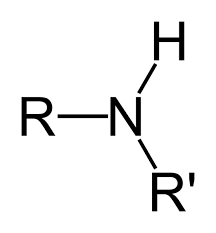
|
|
|
Organolithium/Magnesium reaction |
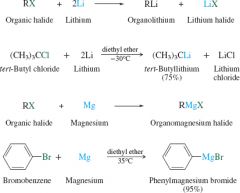
|
|
|
Reactivity of types of halides |
alkyl>aryl>vinyl |
|
|
What affect do double and triple bonds have on polarity? |
increase polarity |
|
|
What are the three pieces of information obtained from an H NMR |
1. Location tells environment of carbon 2. Area(intergration) tells ratio of hydrogens 3. Shape indicates number of neighboring protons. |
|
|
When will two hydrogens on the same carbon not be equivalent? |
When there is a stereocenter! |
|
|
When will a methyl group have unequivalent hydrogens? |
NEVER! |
|
|
What is the larger number area in an NMR called?? |
Downfield |
|
|
Alkane chemical shift |
1-2ppm |
|
|
What is the inductive effect? |
Withdrawl of electron density from one atom towards another, produces deshielded atoms |
|
|
When several halogens are present what happens to deshielding? |
Additive effect, more halogens=more deshielding |
|
|
methyl chemical shift |
0.9ppm |
|
|
methylene chemical shift |
1.2ppm |
|
|
methine chemical shift |
1.7ppm |
|
|
alcohol or ether effect on alpha proton shift |
+2.5 |
|
|
oxygen of ester on alpha proton shift |
+3 |
|
|
carbonyl group on alpha proton shift |
+1 |
|
|
Why are aromatic hydrogens shift so strong? |
the net magnetic field of the aromaticity is very strong |
|
|
What causes multiplicity? |
The magnetic effect of neighboring hydrogens |
|
|
N+1 rule |
Neighboring carbons are n and 1 and that will give you the multiplicity of the given hydrogen |
|
|
coupling constant |
stronger the closer the chemical shifts are together
(J) the distance between the individual peaks |
|
|
Steps to analyzing proton nmr |
1. Degree of unsaturation 2. number and intergration of signal 3. analyze signals and draw fragments 4. assemble fragments |
|
|
What is the important aspect of C13 NMR? |
Chemical shift |
|
|
enantiotopic hydrogens |
end up with enantiomers when doing z substitution(with stereocenter present adjacent)
not equivalent but chemical shift is the same |
|
|
diastereotopic hydrogens |
different chemical shifts, not equivalent
diastereomers when z substitution |
|
|
can carbons be enatiotopic and diastereotopic? |
Yes |
|
|
Preparation of acetlyenic orgnolithium reagents |
terminal alkyne with alkyllithium |
|
|
Palladiun Cross Coupling |
mostly involves synthesizing biaryls
electronegative positive atoms removed and new carbon carbon bonds formed
Pd(0) reagent |
|
|
replace alcohol with halide |
add strong acid |
|
|
hydroboration |
breaks double bond adds OH anti M's rule syn addition god for regio/stereoselective reactions |
|
|
oxidizing agent |
accepts electron from another species |
|
|
reducing agent |
donates electron to another species |
|
|
oxidation of primary alcohols and secondary alochol reagents |
PCC, C5H5NH, ClCrO3-, PDC Cr2O7
replaces alcohol with aldehyde for primary replaces alcohol with ketone for seconday |
|
|
epoxide with grignard reagent |
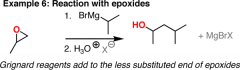
creates primary alcohol
have two more carbons in product then grignard reagent |
|
|
Why can you not use a protic solvent in a grignard reaction? |
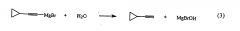
It destroys the reagent |
|
|
What does a ketone + Grignard reagent yield? |
Tertiary alcohol |
|
|
What does an aldehyde with a grignard reagent yield |
secondary alcohol |
|
|
What does formaldehyde with grignard reagent produce? |
primary alcohol |
|
|
Acid catalyzed reaction of alkenes + H2O |
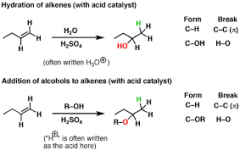
water adds to double bond M's rule |
|
|
hydroboration-oxidation of alkenes |
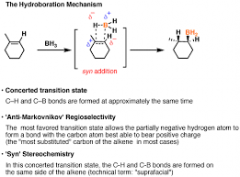
no rearrangement H and OH add syn
opposite M's rule |
|
|
hydrolysis of alkyl halide |
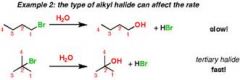
|
|
|
reduction of aldehydes by hydrogenation |
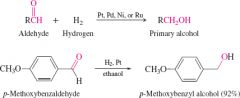
form primary alcohols
|
|
|
reduction of ketones by hydrogenation |
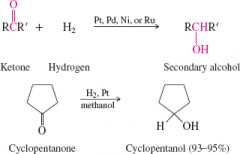
forms secondary alcohols |
|
|
redution of ketone and aldehyde by metal hydride that can be used with protic solvents |
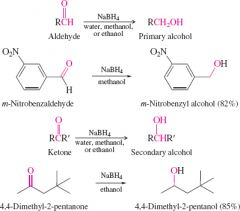
ketons still form secondary
aldehydes still form primary
doesnt require additional adding of water |
|
|
reduction of ketone and aldehyde by metal hydride with aprotic solvents |
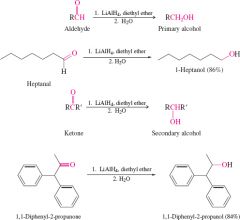
must have water added as second step due to volitality with water |
|
|
What is the reducing agent when using NaBH4 |
BH4- |
|
|
Which reagents can you use to add an alcohol to an alkene with a carbonyl group? |
LiAlH4 or NaBH4 they will not reduce the alkene double bonds |
|
|
reduction of carboxylic acid |
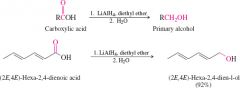
ONly LiAlH4! uses the same number of carbons in reactant and product |
|
|
general epoxide + grignard reagent mechanism |
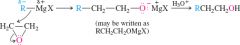
|
|
|
vicinal alcohol |
a compound with alcohols on adjacent carbons |
|
|
alkenes + osmiumtetraoxide |
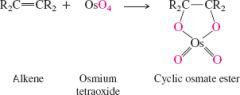
stable but easily cleaed with tertbuytl alcohol |
|
|
dihydroxylation |
syn, vicinal addition of alcohols on double bond of alkene
-Reagents OsO4 with tert butyl alcohol in tert butyl alcohol |
|
|
products of dihydroxylation on alkenes |
if cis reagent 1 product if trans 2 enatiomers(R and S) |
|
|
meso compounds |
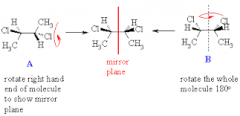
a molecule with multiple stereocenters that is superimposable on its mirror image |
|
|
alcohol + hydrogen halide |
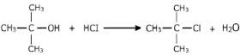
yields alkyl halide |
|
|
alcohol and thionyl chloride |
produces alkyl chlorides |
|
|
alcohol and phosphorous tribromide |
produces alkyl bromide |
|
|
acid catalyed dehydration of alcohol |
produces alkene
OH group removed and forms water
alkene formed where hydroxyl group was |
|
|
alcohol conversion to p=toulenesulfonates |
kicks off hydrogen adds R and SO2 group |
|
|
conversion of alchol to ether |

2 equivalents of the alchol + acid yeild RCH2 group on both sides of original oxygen
primary alcohols only |
|
|
REACTION: Diol + H2SO4 |

will form water and 5 or 6 member either when it can
primary, secondary, tertiary |
|
|
fischer esterification |
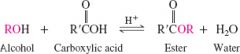
reversible, equilibrium lies slightly toward products |
|
|
Method to shift equilibrium of fischer esterification |
remove water add more reactants |
|
|
Reaction to form ester(anhydride |
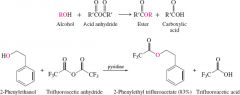
acid annhydride and alcohols in weak base(pyridine) |
|
|
reaction to form ester(protic) |
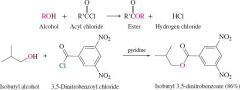
acyl chloride and alcohol |
|
|
oxidation of primary alcohol |

will form carboyxlic acid if in excess |
|
|
Which reagents can be used to synthesize aldehydes by oxidation |
pyridium chlorochromate PCC pyridium dichlorochromate PDC with CH2Cl2
both stop at aldehyde before going to carboxylic acid |
|
|
alcohol oxidation to ketone |
secondary alcohol + PCD or PCC in CH2Cl2 |
|
|
how would you know you havent formed a secondary or primary alcohol in an oxidation involving chromium? |
No color change if it changes to green then most likely you have formed secondary or tertiary |
|
|
tertiary alcohol oxidation |
no reaction |
|
|
why avoid oxidation with chromium |
extremely toxic |
|
|
what nontoxic reagent can be used in oxidation of alcohols? |
chloro dimethylsulfonium |
|
|
formation of chloro dimethylsulfonium ion |
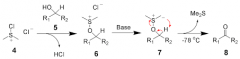
|
|
|
swern oxidation mechanism |
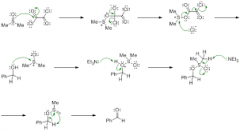
|
|
|
swern oxidationreactants and products |
primary alcohol produces aldehyde secondary alcohol produces ketone
reagenet is DMSO+ NEt3
|
|
|
REACTION:
Oxidative cleavage of 1,2 diols |
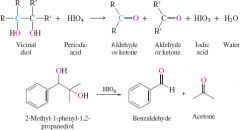
uses iodic acid NaIO4 or HIO4 to cleave the molecule in two
|
|
|
REACTION:
Cyclic diol cleavage |
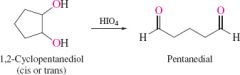
cleaves ring and add carbonyl groups where OH groups were with carbonyls on opposite ends |
|
|
thiol group structure and behavior |
R-SH called cercapto group larger better nucleophle no hyrogen bonding due to decreased polarity |
|
|
thiol preperation |

SN1 |
|
|
production of disulfides |

|
|
|
add alkane group in leaving groups position |
gilman reagent Dialkyl copper lithium1 |
|
|
where must the double bond be when identifying the area of hydrogenation? |
Where the chirality center is |
|
|
molecular formula rule of olefin cross metathesis |
original reactants - C2H4 |
|
|
synthesis of primary alcohol using grignard reagents |
1. ethylene oxide 2. formaldehyde |
|
|
why cant tertiary alcohols be oxidized? |
No hydrogen on the carbon that the hydroxyl group is attached to to be removed |
|
|
List 7 Reactions to synthesize alcohols
|
1. Reaction of alcohols with hydrogen halides
2. Reaction of alcohols with thionyl chloride 3. Reaction of alcohols and phosphorus tribromide 4. Acid catalyzed dehydration of alcohols 5. Conversion of alcohols to mesylates, tosylates, and triflates 6. Converting primary alcohols to ethers. (Condensation reaction) 7. Fischer Esterification |
|
|
PROPERTIES OF: |
Reactivity: 3* > 2* > 1*
Sn1 |
|
|
PROPERTIES OF:
Reaction of alcohols with thionyl chloride |
1* and 2* alcohols
Stereochemistry inverted Produces alkyl chloride |
|
|
PROPERTIES OF:
Reaction of alcohols and phosphorus tribromide |
1* and 2* alcohols
Sn2 |
|
|
PROPERTIES OF:
Acid catalyzed dehydration of alcohols |
E1
3* > 2* > 1* rearrangement possible |
|
|
Why do we convert alcohols to mesylates, tosylates, and triflates
|
makes OH a good leaving group
|
|
|
PROPERTIES OF:
Converting primary alcohols to ethers. (Condensation reaction) |
2 Eq.
Protonation and Substitution pathway |
|
|
PROPERTIES/REAGENTS OF:
Fischer Esterification |
– Reversible reaction
Reagents: 1. Always 1 alcohol & 1 carboxylic acid 2. Acid (H2SO4) |
|
|
PROPERTIES/PRODUCTS OF:
Reaction of alcohols and acyl chlorides |
Removal of proton from alcohol, removal of Cl ion from acyl chloride
Products: 1. Ester formation 2. HCl formation |
|
|
PROPERTIES/PRODUCTS OF:
Reaction of alcohols and acid anhydrides |
Anhydride is cleaved at middle oxygen, acylate part is protonated, other part gets the R–O portion of alcohol
Products: 1. ester formation 2. carboxylic acid also formed |
|
|
REAGENTS FOR:
Oxidation of primary alcohols to carboxylic acids |
K2Cr2O7/K2CrO4/KMnO4
|
|
|
REAGENTS FOR: |
PDC/PCC |
|
|
REAGENTS FOR : |
PDC/PCC in CH2I2 |
|
|
REAGENTS FOR:
Oxidation and Cleavage of Vicinal Diols |
Periodic Acid (HIO4) |
|
|
PROPERTIES/PRODUCTS OF:
Oxidation and Cleavage of Vicinal Diols |
Vicinal Diols Only
Can be used to open cyclic diols Cleavage between two OH bearing carbons Carbonyl is formed on each carbon bearing the OH Product: Ketone or Aldehyde, depending on substituents |
|
|
REAGENTS FOR: |
H2SO4
Produces ROR |
|
|
Describe an Acid Anhydride |
An organic compound with 2 acyl groups bound to the same oxygen. |
|
|
REACTION: Addition of primary alcohol with two additional carbons |
epoxide, H3O+ |
|
|
REACTION:
Preparation of Diols |
Reduction of existing ketones or aldehydes with H2 and catalyst
|
|
|
REACTION:
Preperation of Vicinal Diols |
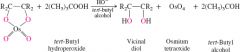
Alkene + OsO4, Tertbutyl alcohol Adds alcohols to both carbons of double bond |
|
|
REAGENT:
Add Chlorine |
SOCl2 |
|
|
REAGENT: Add Chlorine or Bromine to benzene |
FeCl3, Cl2 FeBr3, Br2 |
|
|
Why use PDC/PCC instead of NaCr2O7 |
-PCC/PDC will stop at ketone or aldehyde but Na2Cr2O7 will reduce all the way to carboxyli acid |
|
|
REACTION:
Cleavage of alkene by oxidizing reagent |
MnO4 in H3O or O3 in Zn
Aldehydes will be oxidized all the way to carboxylic acids
|
|
|
REACTION:
Sodium Amide + Alcohol |
NaNH2 + ROH
Oxygen grabs Na NaOR formed + NH3 |
|
|
Fischer esterification reagent that adds a methyl group |
CH3OH in H2SO4 |

