![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
21 Cards in this Set
- Front
- Back
|
Normal K values
|
• Plasma K 3.5-5.0
• ICF 150 mmol/L • ECF 5 mmol/L • ICF 25L • ECF 15L • Intracellular: 150 x 25 = 3750 mmol (98%) • Extracellular: 5 x 15 = 75 mmol (2%) |
|
|
factors influencing cellular potassium uptake
|
hormonal influences,
acid-base homeostasis, insulin, glucagon and ß and alpha adrenergic activity |
|
|
Why is potassium so tightly
regulated? |
• Resting membrane potential depends on ratio
between intracellular and extracellular potassium (Nernst equation) • Changes in extracellular potassium profoundly affect this membrane potential • Changes in membrane potential lead to changes in polarisation and excitability of nerve/muscle cells |
|
|
Distribution affected by
|
• Na/K ATPase vs K channels
• Insulin • Catecholamines – beta 2 • alkalosis |
|
|
Balance of intake and output
|
• Intake
– Dietary K approx 1 mmol/kg (39 mg = 1 mmol) – 80 (30-150) mmol/day (3200 mg/day) • Output – 90% urine – 10% stool |
|
|
K transport in the nephron
|
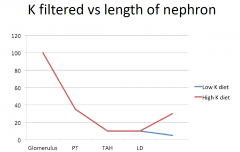
• Site 1:PT 65% K filtered is reabsorbed
• Site 2:TAH 25% • Site 3:ED nil • Site 4: LD/CCD K secreted 0-20% equivalent of filtered load • Site 5: MCD 5% reabsorbed (normally doesn’t happen) |
|
|
Cortical collecting duct- K
|
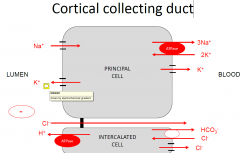
|
|
|
Factors regulating LD/CCD K secretion
|
• Blood
– Aldosterone* – Plasma [K] – Increased pH • Luminal – Na delivery e.g. diuretics at sites 1,2,3 – Flow rate by reducing luminal K concentration – Negative lumen potential difference (note amiloride) |
|
|
Potassium and aldosterone
|
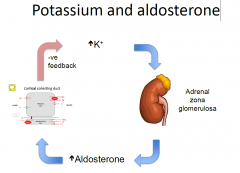
• Adrenal gland – increased plasma K increases
aldosterone secretion from zona glomerulosa (outer segment of adrenal) • Kidney – aldosterone increased K secretion at site 4, causing increased K excretion • This causes plasma K to fall, feeding back on adrenal gland |
|
|
Regulation of Renin
|
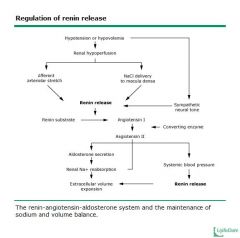
• Reduced ECF V and lower blood pressure ->
increased renin -> AI -> AII -> zona glomerulosa – increased aldosterone secretion • Aldosterone acts on kidney to increase sodium reabsorption and reduce sodium excretion • These effects on sodium negatively feedback on ECF and BP |
|
|
Aldosterone MOA
|
Aldosterone acts primarily in the distal nephron to increase the reabsorption of Na+ and Cl- and the secretion of K+ and H+. As with other steroid hormones, aldosterone acts by diffusing into the tubular cell and then attaching to a specific cytosolic receptor, The hormone-receptor complex then migrates to the nucleus, where it interacts with specific sites on the nuclear chromatin to enhance messenger RNA and ribosomal RNA transcription. This in turn is translated into the synthesis of new proteins called aldosterone-induced proteins (AIPs). The time required for these processes to occur accounts for the 30 to 90 minute latent period before electrolyte excretion is affected
How these proteins act is not well understood. Aldosterone increases the abundance of the alpha subunit and promotes the phosphorylation of the beta and gamma subunits of the luminal membrane Na+ channel through which luminal Na+ enters the cells. - the AIPs help INC channel activity and stability |
|
|
When is aldosterone maladaptive?
|
Dehydration with hypokalaemia – aldosterone
response is maladaptive |
|
|
Hypokalaemia K < 3.5 mmol/L
|
– Effect on transmembrane potential rendered more
electronegative, leading to hyperpolarisation and enhanced membrane excitability due to enhanced sodium channel activation – Skeletal muscle weakness – Cardiac effects ECG – T wave height falls, U wave, ectopics (ventricular – VT/VF) – GIT – smooth muscle effects – ileus (pseudo-obstruction) – Kidney – long term hypokalaemia causes polyuria – nephrogenic diabetes insipidus and renal fibrosis |
|
|
Hypokalaemia - causes
|
• Redistribution e.g. pH changes
• Reduced intake e.g. diet (must be extreme due to kidney’s ability to adapt) • Excess loss – Kidney • diuretics (sites 1,2,3) • Excess aldosterone (Conn’s syndrome) – GIT • Vomiting – GI loss, volume contraction with increased aldosterone • Diarrhoea – as above • Purgatives – Use urine potassium unless hypovolaemic when aldosterone will increase urine K despite GI loss |
|
|
Urine Potassium
|
– Use urine potassium unless hypovolaemic when aldosterone will increase urine K despite GI loss
high= kidney low = GIT but its not that simple |
|
|
Conn’s syndrome
|
• A cause of hypertension
• Usually associated with adrenal adenoma or bilateral adrenal hyperplasia • Hypokalaemia is a common (but not universal) accompaniment -30% |
|
|
Hypokalaemia - treatment
|
• Reverse cause
• Replace with KCL – oral (slow K) or IV • amiloride & spironolactone |
|
|
Hyperkalaemia
|
• > 5 mmol/L
• Effects – Skeletal muscle – paralysis – Heart – T wave peaking, QRS, cardiac arrest depending on K level • Cell membrane potential rendered less electronegative, leading to excessive depolarisation and reduced membrane excitability due to reduced sodium channel activity |
|
|
Hyperkalaemia - causes
|
• Spurious – e.g. haemolysed – pink blood (sample error)
• Redistribution – e.g. low pH, no insulin, beta-blockade • Increased intake in individuals with kidney disease • Renal retention – Reduced GFR (AKI and CKD) – not enough K to distal tubule in filtrate and not enough Na as well – Normal GFR – tubular secretory failure e.g. Addisons, amiloride, diseases of tubules |
|
|
Addison’s disease
|
• Classically an autoimmune disease
• Adrenal insufficiency • Lack of aldosterone • Hyperkalaemia • Hyponatraemia • Hypotension |
|
|
Hyperkalaemia - treatment
|
• Stabilise cell membrane – Ca gluconate
• Shift K into cells – insulin + glucose – B2 agonist – Bicarbonate • Remove potassium – Diuretic – Resin in gut – Dialysis |

