![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
87 Cards in this Set
- Front
- Back
|
matter |
-organisms are composed of matter -matter is anything that takes up space and has mass -matter is made up of elements in pure form and in compound form |
|
|
element |
-what matter is made up of -substance that cannot be broken down to other substances by chemical reactions |
|
|
compound |
substance consisting of two or more elements in a fixed ratio |
|
|
essential elements |
-elements an organism needs to live and reproduce - only 20-20% of elements are essential elements -96% of living matter is made up of C,H,O,N -remaining 4% is Ca,P,K,S |
|
|
trace elements |
- elements required by organism to live in minute quantities -iron is a trace element needed by all forms of life -other trace elements are only required for certain species |
|
|
atom |
- smallest unit of matter that retains the properties of an element -atoms are composed of subatomic particles: neutrons, protons, electrons |
|
|
neutrons |
-no electrical charge -in nucleus |
|
|
protons |
-positive charge
-in nucleus |
|
|
electrons |
negative charge
-surround nucleus in electron cloud -attraction between opposite charges keep e- in vicinity of nucleus |
|
|
atomic nucleus |
made up of protons and neutrons
|
|
|
daltons |
-unit used to measure mass of atoms and subatomic particles
-neutrons and protons have masses close to 1 dalton -dalton is the same as atomic mass unit |
|
|
atomic number |
-# of protons in nucleus
-written as a subscript to the left of symbol |
|
|
mass number |
-sum of protons and neutrons in nucleus
-written as superscript to the left of element symbol |
|
|
atomic mass |
-atoms total mass, can be approximated by the mass #
|
|
|
isotopes and radioactive isotopes |
-two atoms of an element that differ in the number of neutrons
- decay spontaneously and leads to a change in the # of protons which transforms atom into an atom of a different element |
|
|
energy |
-capacity to cause change by doing work - |
|
|
potential energy |
-energy that matter has because of its location |
|
|
electron shell |
-an energy level of electrons at a characteristic average distance from the nucleus of an atom |
|
|
valence electrons |
e- in outermost shell of an atom
|
|
|
orbitals |
three dimensional space where an electron is found 90% of the time |
|
|
chemical bonds |
interactions that result in atoms staying close together |
|
|
covalent bonds |
bond that involves the sharing of valence electrons by two atoms |
|
|
molecule |
two or more atoms held together by covalent
|
|
|
single bond |
-sharing of one pair of valence e- |
|
|
double bond |
-sharing of two pairs of valence e-
|
|
|
structural formula |
notation used to represent atoms and bonding |
|
|
molecular formula |
abbreviated form |
|
|
electronegativity |
an atoms attraction for the e- in a covalent bond |
|
|
cation |
positively charged ion
|
|
|
anion |
negatively charged ion
|
|
|
ionic bond and ionic compounds |
-attraction between an anion and a cation -compounds formed by ionic bonds |
|
|
hydrogen bond |
forms when a hydrogen atom is covalently bonded to an electronegative atom. the attraction between the hydrogen and electronegative atom is a hydrogen bond |
|
|
reactants |
starting molecules of chem reaction |
|
|
products |
final molecules of a chem reaction
|
|
|
chemical equilibrium |
reached when the forward reaction and reverse reaction occur at the same time relative concentrations of reactants and products dont change |
|
|
nonpolar covalent bond |
atoms share the electrons equally |
|
|
polar covalent bond |
one atom is more electronegative, the atoms do not share the electrons equally |
|
|
ion |
charged atom or molecule |
|
|
van der waals interactions |
attractions between molecules that are close together |
|
|
water |
molecule that supports all life |
|
|
adhesion |
attraction between different substances, helps transport water in plants |
|
|
surface tension |
a measure of how hard it is to break the surface of a liquid, water has a high surface tension
|
|
|
kinetic energy |
the energy of motion. anything that moves has kinetic energy
|
|
|
specific heat |
the amount of heat that must be absorbed or lost for 1g of that substance to change its temperature by 1 degree Celsius |
|
|
solution |
liquid that is a complete homogeneous homogeneous mixture of substances |
|
|
solvent |
the dissolving agent of a solution
|
|
|
solute |
substance that is dissolved
|
|
|
aqueous solution |
solution where water is a solvent
|
|
|
hydrophillic |
any substance that has an affinity for water
|
|
|
hydrophobic |
any substance that repels water. substances that are nonionic and nonpolar like oil
|
|
|
molecular mass |
sum of all masses of all atoms in a molecule |
|
|
mole |
number of molecules usually measured in mols.
1 mol=6.02X10^23 molecules (daltons) |
|
|
molarity |
number of mols of solute per liter of solution (mol/L)
|
|
|
hydrogen ion |
its a single proton. the hydrogen atom thats transferred to another water molecule
|
|
|
hydroxide ion |
the water molecule that lost the proton
|
|
|
hydronium ion |
water molecule the hydrogen transferred to. the water molecule with the extra proton. often represented as H+
|
|
|
acid |
any substance that increases the H+ concentration of a solution
|
|
|
base |
any substance that decreases the H+ concentration of a solution
|
|
|
ph |
scale used to describe how acidic or basic a solution is. acidic- ph less than 7 neutral- has ph of 7 basic-ph greater than 7 |
|
|
four properties of water |
1. cohesive behavior 2. ability to moderate temperature 3. expansion upon freezing 4. versatility as a solvent |
|
|
hydrocarbons |
organic molecules consisting of only carbon and hydrogen |
|
|
structural isomers |
have different covalent arrangements of their atoms. |
|
|
cis-trans isomers |
cis- atoms connected to carbon on same side
trans- atoms connected to carbon on different sides |
|
|
functional groups |
the chemical groups directly involved in chemical reactions
|
|
|
what is ATP? |
adenosine triphosphate. an organic phosphate used as a source of energy for cellular processes. |
|
|
carbohydrates |
serve as fuel and building material for cells. made up of sugars and the polymers of sugars |
|
|
monosacchrides |
simplest carbohydrates |
|
|
disaccharides |
forms when dehydration reaction joins two monosaccharides
|
|
|
polysaccharides |
the polymers of sugars. have storage and structural roles |
|
|
phospholipids |
two fatty acids and a phosphate group attached to a gylercol. major component of cell membranes
|
|
|
steriods |
lipids characterized by a carbon skeleton consisting of four fused rings
|
|
|
catalysts |
chemical agents that speed up reactions
|
|
|
amino acids |
organic molecules with amino and carboxyl groups
|
|
|
DNA |
type of nucleic acid. deoxyribonucleic acid. has a shape of a double stranded helix
|
|
|
RNA |
type of nucleic acid. ribonucleic acid. single stranded. variable forms |
|
|
proteins |
biological functional molecule that consists of one or more polypeptides
|
|
|
gene expression |
the process of DNA synthesizing proteins |
|
|
nucleotides |
the monomers of nucleic acids. composed of a pentose sugar, nitrogenous base and one or more phosphate groups |
|
|
What is a chemical reaction? Why is a chemical reaction important? |
Its the making and breaking of chemical bonds. This leads to changes in the composition of matter |
|
|
Draw hydroxyl group. |

|
|
|
Draw a carbonyl group |

|
|
|
Draw a carboxyl group |
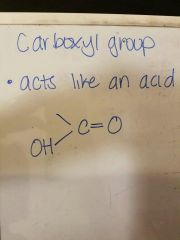
|
|
|
Draw an amino group |
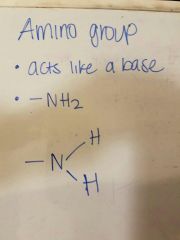
|
|
|
Draw a sulfhydryl group |
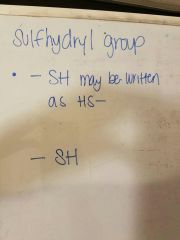
|
|
|
Draw a phosphate group |
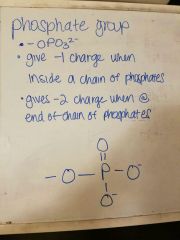
|
|
|
Draw a methyl group |

|
|
|
What are the four classes of macromolecules. What are they used for? |
Carbohydrates - serve as fuel and building material Lipids- has phospholipids which are important for cell membranes Proteins- variety of functions like enzymes, hormones, receptor, motor, and defense Nucleic acid- store, transmit, and express hereditary information |

