![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
43 Cards in this Set
- Front
- Back
|
Matter |
Anything that takes up space and has mass |
|
|
Mass |
Amount of material in matter, equals weight |
|
|
Atoms |
Smallest stable units of matter; building blocks of all living and non-living things |
|
|
Proton |
In nucleus, positive charge, provides mass & identity |
|
|
Neutron |
In nucleus, no charge, provides mass |
|
|
Electron |
In electron shell, negative change, allows interaction with other atoms (bonding) |
|
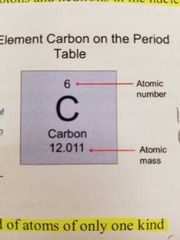
Atomic number |
Number of protons in an atom (number of protons equals number of electrons) |
|

Mass number |
Total number of proto s and neutrons in nucleus |
|
|
Element |
Pure substance composed of atoms of only one kind |
|
|
Valence shell |
Outermost energy level forming "surface" of atom |
|
|
Valence electrons |
Electrons in outermost energy level |
|
|
Ionic bond |
Involves loss/gaining of one or more electrons, creating bond by electrical attraction |
|
|
Ion |
Atoms that has unequal number of protons/electrons creating charge |
|
|
Cation |
Ion with positive charge |
|
|
Anion |
Ion with negative charge |
|
|
Ionization |
Dissociation of bonds that separate cations from anions that can now take part in other chemical reactions |
|
|
Covalent bonds |
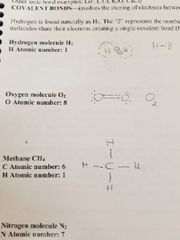
Involve sharing of electons between atoms creating a bond |
|
|
Antioxidant |
Molecules that safely interact with free radicals, neutralizing them before vital molecules are damaged |
|
|
Free radical |
Destructivd ion or molecule containing unpaired electrons in the valence energy level |
|
|
Element |
Pure substance made up by only one kind of atom |
|
|
Hydrogen bond |
When hydrogen shares its electron with other atoms its proton keeps slight positive charge which allows it to form weak bonds with other slightly negative atoms |
|
|
Acid |
Substance that increases hydrogen ion concentration in a water solution when dissociation occurs, strong acids dissociate completely |
|
|
Base |
Substance that decreases hydrogen concentration in a water solution by releasing hydroxyl ions to form water molecules with the hydrogen, strong bases dissociate completely |
|
|
Surface tension |
Water uses hydrogen bonds to create a barrier the keeps small objects from entering |
|
|
Reactants |
Reacting substances in a chemical reaction |
|
|
Products |
Substances formed during chemical reaction |
|
|
Kinetic energy |
Energy of motion that can be transferred to another object and do work |
|
|
Potential energy |
Stored energy that has the potential to do work |
|
|
Enzyme |
Special proteins that promote chemical reactions by lowering their required activation energy |
|
|
Catalyst |
Compounds that speed up chemical reactions without being permanently changed or consumed; enzymes belong to this class of compounds |
|
|
Hydrophobic |
Compounds that do not readily interact with water |
|
|
Hydrophilic |
Compounds that readily interact with water |
|
|
Specificity |
Every enzyme catalyzes only one type of reaction |
|
|
Cofactor |
Helper molecule that has to bind an enzyme before a substrate can bind to it (regulation process) |
|
|
Adenosine Triphosphate |
ATP = created by phosphorylation of ADP, consists of adenine, ribose and three phosphate groups. It's the high energy compound derived from nucleotides |
|
|
Nucleotides |
Building blocks of nucleic acids, made of 3 components: nitrogenous base, penrose sugar, phosphate group |
|
|
RNA |
Ribonucleic acid, performs protein synthesis as directed by DNA |
|
|
DNA |
Deoxyribonucleic acid, stores genetic information that controls protein synthesis |
|
|
Magnesium |
Mg |
|
|
Calcium |
Ca |
|
|
Chlorine |
Cl |
|
|
Zinc |
Zn |
|
|
Copper |
Cu |

