![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
15 Cards in this Set
- Front
- Back
|
molecular geometry
|
general shape of a molecule determined by the relative positions of the atomic nuclei
|
|
|
VSEPR (valence shell electron pair repulsion)
|
define
|
|
|
2 valence pair form and angle
|
Linear
180 degrees |
|
|
3 valence electron pairs and no lone pairs form and degree
|
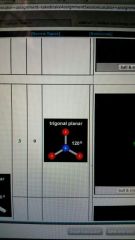
trigonal planar
120 degrees |
|
|
2 valence electron pairs and 1 lone pair form and degree
|
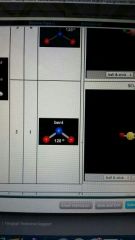
bent
120 degrees |
|
|
4 valence electron pairs and 0 lone pairs form and angle
|
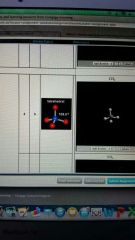
tetrahedral
109.5 degrees |
|
|
3 valence electron pairs and one lone pair form and degree
|
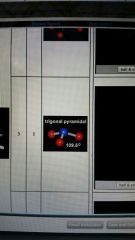
trigonal pyramidal
109.5 degrees |
|
|
2 valence electron pairs and two lone pairs form and degree
|
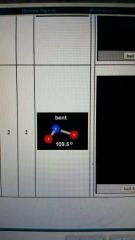
Bent
109.5 degrees |
|
|
5 valence pairs and 0 lone pairs form and angle
|

trigonal bipyramidal 90 and 120
|
|
|
4 valence electron pairs and 1 lone pairs form and angle
|
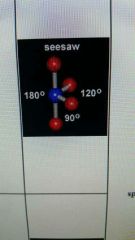
|
|
|
3 valence electron pairs and 2 lone pairs
|
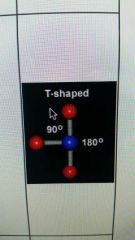
|
|
|
6 valence electron pairs and 0 lone pairs
|
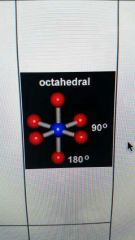
|
|
|
5 valence pairs and 1 lone pairs
|
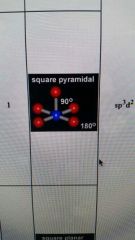
|
|
|
4 valence pairs and 2 lone pairs
|

|
|
|
de broglie wavelength
|
lambda=h/(mv)
|

