![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
49 Cards in this Set
- Front
- Back
|
An orbital with l = 2 is called a _____ orbital. These orbitals always occur in groups of _____ orbitals of equal energy. |
d
5 |
|
|
Which of the following statements correctly describe the principal quantum number, n? Select all that apply. |
n can have any positive whole-number value and the lower the value of n, the greater the probability that the electron is closer to the nucleus. |
|
|
Which of the following is a consequence of the Heisenberg uncertainty principle? |
It is not possible to assign fixed paths for electrons. |
|
|
An orbital with l = 0 is called a(n) _____ type orbital and has a(n) _____ shape. There is always a total of _____ orbital(s) of this type for each principal energy level. |
s spherical 1 |
|
|
Which of the following statements correctly describe the angular momentum quantum number, l? Select all that apply? |
- The allowed values of l are determined by the value of n.
- The number of possible l values equals the value of n.
The value of l dictates the allowed values of m[l] |
|
|
Electromagnetic radiation consists of particles called ______, each of which has a discrete amount, or quantum, of energy. However, since electromagnetic radiation also has wave properties, each particle is also characterized by a specific _____ (m) and frequency (s-1). |
photons wavelength |
|
|
Place the following types of electromagnetic radiation in order of increasing wavelength (shortest at the top to longest at the bottom). Radiowaves, UV radiation, Visible light, X-ray, IR radiation. |
X-ray UV radiation Visible light IR radiation Radiowaves |
|
|
Waves that are in phase... |
experience constructive interference. |
|
|
Amplitudes will add... |
if waves are in phase. |
|
|
Waves that are out of phase... |
experience destructive interference. |
|
|
Amplitudes will cancel... |
if waves are out of phase. |
|
|
Energy is not continuous, but is quanitized or divided into "packets" each of which contains a definite amount of energy. An energy "packet" is called a(n) _____ and the energy of each "packet" is directly proportional to its_____. |
quantum frequency |
|
|
Which of the following statements correctly describe probability density and electron probability density diagrams? |
- The probability of finding an electron generally decreases with increasing distance from the nucleus.
- These diagrams show the probability of finding an electron in a particular region of the atom. |
|
|
All matter behaves as though it moves in a(n) _____. The motion of any particle can be described by the de Brogile equation, which relations the wavelength of a particle to its ______ and speed. |
wave
mass |
|
|
The speed of a wave is determined by multiplying the ______ of the wave by its wavelength. In a vacuum all electromagnetic radiation travels at a constant speed, the speed of ______. |
frequency
light |
|
|
An atomic orbital in terms of the mechanical model of the atom... |
is a mathematical function that describes the position of the electron-wave in three dimensions. |
|
|
The principal quantum number n indicates the... |
principal energy level. |
|
|
The angular momentum quantum number l indicates the... |
sublevel of the orbital. |
|
|
The magnetic quantum number m[l] indicates the... |
orientation of an orbital in space. |
|
|
The relative size of the orbital is related to the value of... |
the quantum number n. |
|
|
The shape of the orbital is related to the value of... |
the quantum number l. |
|
|
For a principal quantum number n=3, what values of the angular momentum quantum number l are allowed? |
0, 1, & 2 |
|
|
The _____ momentum quantum number has the symbol l and can take values from _____ to (n-1). The number is related to the _____ of the orbital. |
angular 0 shape |
|
|
The phenomenon where an electric current is produced by shining a light on a metal plate is called the ____effect. The minimum frequency required to cause a current to flow is called the ______ frequency. This frequency depends on the identity of the metal used. |
photoelectric
threshold |
|
|
Which of the following statements correctly describe the Bohr model of the hydrogen atom? Select all that apply. |
- The atom is in its lowest energy state when the electron is in the orbit closest to the nucleus.
- Each energy state of the hydrogen atom is associated with a fixed circular orbit of the electron around the nucleus.
- Only certain energy levels are allowed within the hydrogen atom. |
|
|
Using the Schrodinger wave equation we can determine the probability of finding an electron in a particular region of the atom. The probability density _____ with distance from the nucleus, meaning that the farther one gets from the nucleus the _____ likely it is to find an electron. |
decreases
less |
|
|
Which of the following statements correctly describe the magnetic quantum number, m[l]? Select all that apply. |
- The allowed values for m[l] range from -l to +l.
- This value indicates the orientation of an orbital in the space around the nucleus. |
|
|
A p orbital has _____ regions or lobes of high electron probability, on either side of the nucleus. This gives the orbital a dumbbell shape with a(n) _____ or region of zero electron density at the nucleus. |
two
node |
|
|
The distance a wave travels during one cycle is called its _____. This is usually measured as the distance between two _____ or two troughs. |
wavelength
peaks |
|
|
All waves in the electromagnetic spectrum travel at the same ______ through a vacuum, but differ in their frequency and wavelength. A wave with a long (large) wavelength will have a(n) ______ frequency. |
speed low |
|
|
Electrons, like all other matter, exhibit the dual behavior of both _____ and waves. Since electrons travel like waves their energy is restricted to certain energy _____, each of which is associated with a specific wavelength. |
particles
levels |
|
|
Which of the following statements correctly describe photons? |
-A photon can transfer momentum to another particle in a collision.
|
|
|
The apparent bending of a wave around the edge of an object is called_____. If a wave passes through a slit that has a width of the same order of magnitude as its______ it will bend around both edges of the slit to give a semicircular wave. |
diffraction
wavelength |
|
|
Which of the following statements correctly describes how energy is absorbed by an atom? |
The energy of the absorbed radiation must match the difference between the two energy states of the atom |
|
|
Which of the following statements correctly describe wave-particle duality? |
-matter and energy are different forms of the same entity.
|
|
|
In Bohr's model of the atom, the quantum number n is associated with the _____ of an electron orbit. The lower the n value, the ____the electron is to/from the nucleus and the ____ energy level. |
Energy
closer
lower |
|
|
Planck's constant equation... |
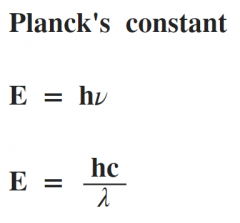
|
|
|
Each element has its own atomic line spectrum, consisting of fine lines of individual wavelengths that are characteristic for the element. This occurs because of the atom contains specific _______ levels, and an atom can only absorb or emit radiation that corresponds to the ________ between these levels. |
energy
difference |
|
|
Visible light is one type of _____ radiation, which consists of energy propagated by eletric and _____ fields that alternately increase and decrease in intensity as they move through space. |
electromagnetic
magnetic |
|
|
The _____ of a wave is the number of wave cycles per second. The quantity is given the symbol 𝝂 and has units of s⁻¹ or _____. |
frequency
Hz |
|
|
In the quantum mechanical model of the atom, an electron is viewed as a wave-particle that occupies a three-dimensional space near the nucleus. The movement of the electron is described by a _____ function, which is also called an atomic _____. |
wave
orbital |
|
|
The amplitude of an electromagnetic wave relates to _____ and _____. |
Intensity and brightness (for visible light) |
|
|
In order for a current to flow in a photoelectric cell, a photon of light must be _____ by an electron in the metal. For this to occur the____ of the proton must be equal to or greater than the energy needed to remove the electron from its energy state. It is the _____ of the light that is important, not its intensity. |
absorbed
energy
frequency |
|
|
An equation that reflects the relationship between energy and frequency is... |
E = h𝝂 |
|
|
These statements correctly describe p orbitals... |
- p orbitals always occur in groups of three
- a p orbital has a dumbbell shape with two lobes, one on either side of the nucleus.
- The p orbitals of a group are perpendicular to each other. |
|
|
An atom is in its lowest energy state, or ____ state, when its electrons are in the lowest possible energy levels. If an electron in the atom absorbs a photon of radiation that is equal in energy to the difference between two energy states it will move to a higher energy state and the atom is said to be in the ______ state. |
ground
excited |
|
|
What determines the color of visible light? |
Its frequency and wavelength |
|
|
According to the Bohr model for the hydrogen atom, the energy of the atom is not continuous but has certain discrete energy ______, each of which is related to a fixed circular _____ of the electron around the nucleus. The father the electron is from the nucleus, the ____the energy of the system. |
-levels
-orbit
-higher |
|
|
Describe the atom in terms of quantum mechanics. |
- Electrons exhibit behavior of both waves and particles.
- The movement of each electron in the atom can be described by a wave function.
- Each electron occupies a three-dimensional space near the nucleus. This space is described by a wave function. |

