![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
19 Cards in this Set
- Front
- Back
|
What is the radius of an atom and a nucleus, size of an animal cell? |
O.1nm and 1 x 10^-14m 10-30 micrometers |
|
|
What is the mass and charge of proton, neutron and electron? |
Proton mass- 1 Neutron mass-1 Electron mass- 0 Proton charge- +1 Neutron charge - 0 Electron = -1
|
|
|
What is the atomic number and what is the mass number? |
The mass number is at the top and the atomic is at the bottom |
|
|
What is an element and what is a molecule? |
E- a substance made up of only one type of atom M- A small number of atoms chemically bonded together |
|
|
What is a isotope |
Is an atom with the same number of protons and electrons but a different number of neutrons (same atomic number, different mass number) |
|
|
How do you figure out the number of shells and the number of electrons on the outer shell in an element |
Count down to find the number of electrons on the outer shell count across for number of shellsq |
|
|
Mixture definition |
2 or more compounds or elements not chemically bonded together. Chemical properties of each component remain uncharged |
|
|
What is the formula for working out the relative mass |
(Number of atoms x mass) (number of atoms x mass) ------------------------------------------------------- Total number of atoms |
|
|
Definition of pure |
A single element or compound not mixed with any other substance |
|
|
What is the stationary medium and the active medium in chromatography? |
Stationary- chromotography paper Solvents + pigments - active medium |
|
|
What happened in chromotography |
Molecules have to pass through the stationary medium. Smaller molecules manage to go further In an allotted amount of time as the larger molecules are restrained |
|
|
Who was the plum pudding model by/nuclear model/planetary/quantum? |
PP- J.j. thomson N- ernest Rutherford P- Neil's bohr Q- Erwin shrödinger |
|
|
What happens as you move down the alkali metals? |
They become more reactive meaning easier to lose outer electron |
|
|
What happens as we move down the halogens? |
They become less reactive Boiling points and melting points increase |
|
|
Which halogens are gases liquids and solids? |
G- Chlorine and fluorine Liquids- bromine S- iodine and astatine |
|
|
Metal halide compound? What is this reaction called? What other gas could react with a halogen? |
●Sodium chloride ●Displacement ●Hydrogen |
|
|
How are elements ordered in the periodic table? Why is it called the periodic table? |
By atomic number Its arranged in periods |
|
|
What are ions? What happens to metal/ non-metals? |
Charged particles Non-metals become negatively charged Metals become positively charged |
|
|
What types of bonding are there? |
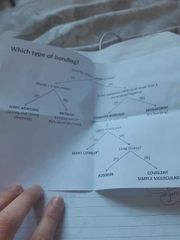
Covalent bonding, monatomic bonding, polymer, covalent simple molecular, giant covalent, metallic, ionic bonding |

