![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
27 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
Matter |
Has volume and mass |
|
|
|
Pure Substance |
Always contains the same ratio of components. All one substance.
|
|
|
|
Element |
Cannot be broken down into anything simpler by chemical means. Atom=smallest chunk E.g. mercury, krypton |
|
|
|
Compound |
Chemical combination of two or more elements Molecule=smallest chunk E.g. pure water, salt, pure sand, magnesium oxide |
|
|
|
Solution |
-All parts are the same but can be made in any amount -Two or more substances -Homogeneous mixture -E.g. salt water, clear tea, bronze |
|
|
|
Mechanical mixture |
-Can identify different parts (phases) -Heterogeneous mixture -E.g. a beach, ocean water, milk, human blood, paper with ink |
|
|
|
Proton (location, charge) |
-in the nucleus -charge of +1 |
|
|
|
Neutron (location, charge) |
-in the nucleus -charge of 0 |
|
|
|
Electron (location, charge) |
-in orbit -charge of -1 |
|
|
|
Rutherford's model of the atom |
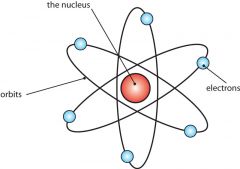
Protons and neutrons in the nucleus with electrons in orbit. Atom is mostly empty space. The Solar System model. |
|
|
|
Rutherford model vs. Thomson model |
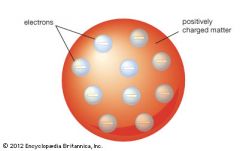
-Thomson has a solid atom with electrons scattered in positive stuff. Plum Pudding Model. -Rutherford put all the positive in the center and electrons in orbit. The atom is not solid. |
|
|
|
Bohr model of the atom |
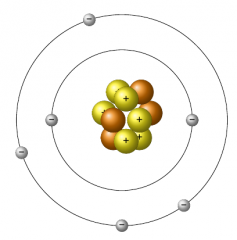
Had the atom as Rutherford did except electrons existed in a specific order with a maximum number of electrons in each orbit. |
|
|
|
Max. number of electrons in each level of a Bohr model |
2,8,8... (for first twenty elements) |
|
|
|
Hypothesis |
-Educated guess -Based on evidence from previous knowledge |
|
|
|
Theory |
-Has been scientifically proven -Has evidence to support it |
|
|
|
Properties of metals |
-Lustre -Silver except for gold and copper -Solid at room temperature except for mercury -conductors -malleable and ductile |
|
|
|
Noble gases |
-Helium, Neon, Argon, Krypton, Xenon, Radon -Last column on Periodic Table |
HNAKXR |
|
|
Halogens |
-Fluorine, Bromine, Iodine, Chlorine, Astatine -Second last column on Periodic Table |
|
|
|
Transition metals |
-Chunk in the middle of Periodic Table -Manganese, Cobalt, Iridium, Titanium, Zinc |
|
|
|
Charge on ions pattern |
+1, +2, +3, +/-4, -3, -2, -1, 0 |
|
|
|
Re activity in metals on the Periodic Table |
-Less reactive going right -More reactive going down |
|
|
|
Chemical Change |
-Producing a new substance -Colour change, solid or gas forming in liquid, light or heat produced, hard to reverse |
|
|
|
Physical Change |
Change in appearance only |
|
|
|
Physical properties |
Properties of a substance that we can observe or measure. |
|
|
|
Qualitative Properties |
-Describe a substance, observable E.g. colour, shape, taste, odour, size, state of matter, texture, hardness, brittleness, lustre, dull, solubility (does it dissolve), viscosity, optical clarity, malleability, ductility |
|
|
|
Quantitative Properties |
-Measured properties E.g. mass, size, density, volume, melting point, boiling point |
|
|
|
Diatomic Elements |
-Exist only as a pair of atoms together -Hydrogen (H2), Nitrogen (N2), Oxygen (O2), Fluorine (F2), Chlorine (Cl2), Bromine (Br2), Iodine (I2) |
|

