![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
13 Cards in this Set
- Front
- Back
|
IUPAC name of CH3C(CH3)2CH2CH2NH2 |
1-amino-3,3-dimethylbutane |
|
|
IUPAC name of CH2=CHCH2CH2CH3 |
1-pentene |
|
|
Chemical CH3OCH3 is an example of what type of organic compound? |
Ether (R-O-R) Alcohol (ROH) ALDEHYDE (RCHO) KETONE (RCOR) CARBOXYLIC ACID (RCOOH) |
|
|
IUPAC name of CH3CH=CHCH2CH2OH |
3-penten-1-ol |
|
|
What are the hybridizations of ē orbitals between carbons 3 and 4 in the molecule CH2=CHCH2CH2CH3 |
sp3 - sp3 sp3 - single bond sp2 - double bond sp - triple bond |
|
|
Hybridizations of the orbitals between carbons 1 and 2 in the molecule CH2=CHCH2CH2CH3 |
sp2 - sp2 |
|
|
Type/s of bond/s is/are present between carbons 2 and 3 in ē molecule 2-butene |
pi bond and sigma bond single bond = 1 sigma bond double bond = 1 sigma, 1 pi bond triple bond = 1 sigma, 2 pi bonds |
|
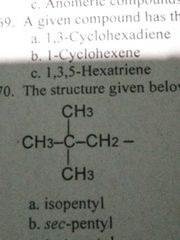
This structure is commonly called: |
Neopentyl |
|
|
Carbon with four different moieties |
Stereocenters |
|
|
Formula for ethers |
ROR |
|
|
CH3CH2COCH2CH3 is a/an: |
Ketone |
|
|
Cahn-Ingold-Prelog system is used for what purpose? |
Determine R or S designation of enantiomers |
|
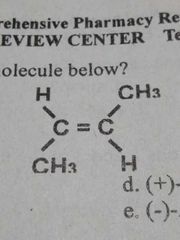
IUPAC name of the given molecule |
trans-2-butene |

