![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
13 Cards in this Set
- Front
- Back
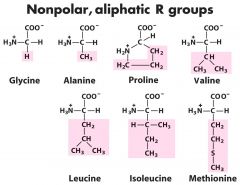
Non-polar groups (7)
|
Gly-G ---- Ala-A ----- Pro-P ------ Val-V -----Leu-L------- Ile-I----- Met-M
|
|
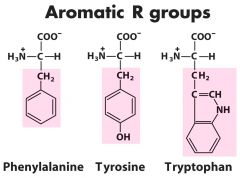
Aromatic R (3) -- tYYYr
|
Phe-F------ Tyr-Y-------Trp-W
|
|
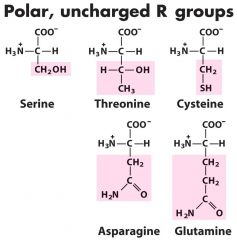
Polar uncharged (5) --- asparagiNNNe
|
Ser-S------Thr-T-------Cys-C------Asn-N------Gln-Q (most abund)
|
|
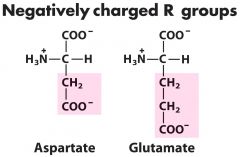
Negative charged -1 (2) --- aspartATE ---- MSG (5C)
|
Asp-D-----Glu-E (neurotransmitter)
|
|
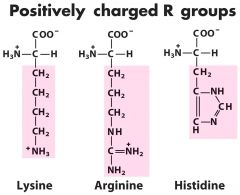
Positive charged +1 (3) -- aRRRg
|
Lys-K------Arg-R------His-H
|
|
|
OH ------------SH --------H2N-C=O----------COO-----------NH3-NH4 --- noncharged----- Arom
|
ST---CM----NQ-----ED-------KRH---- GAPVLIM------FYW
|
|
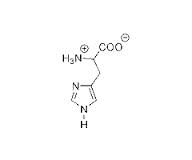
His positive when
|
when pH<6.0 in pronated form --> low pH lots of H add H in
|
|
Sickle cell --- less water soluble --- brittle ---- treatment
|
Glu --> Val on #6 (neg - vs nonpolar) ---> noncharged less polar water is polar ----- crystalize ----- give more O2
|
|
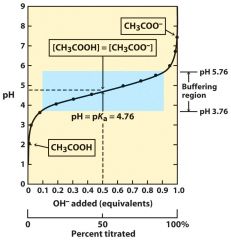
pH=pKa + log (B/A) -- protonation state (acid state) --> base/Acid
|
--> pH determine the drug in its acid form or basic form
|
|
|
What is the pH at which we have 10 times more of the acid form than the basic form for the drug “aspirin”? pKa=3.5
|
pH=pKa + log (B/A) ---- pH = 3.5 + log (1/10) --> pH = 3.5 - 1 = 2.5 --- more acid in stomach ---- need enteric coating
|
|
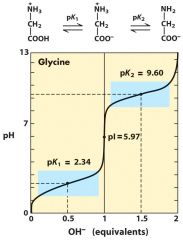
pI and buffer capacity
|
at pI: drug has no charges ---- buffer in 1 pH unit of its pKa
|
|
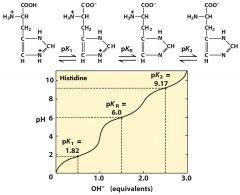
His pI --> use the point of 0 charged
|
pI=(6.0+9.17)/2=7.59 (when it has both + and -)
|
|
|
Protein cross-link Cys (curly hair)
|
A disulfide bridge
|

