![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
20 Cards in this Set
- Front
- Back
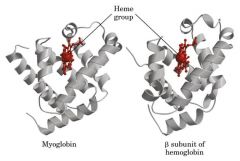
Myoglobin
|
tert histadine structure includes Fe-heme and bind to O2 more tightly Hb
|
|
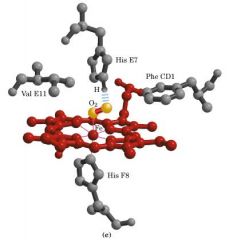
allosteric interactionn (O2 -heme)
|
binding of a molecule to a binding site affects binding properties of another site on the protein.
|
|
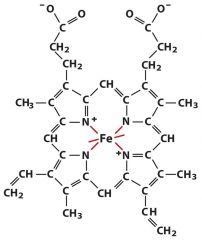
prosthetic group (Fe-heme)
|
non-protein permanently associated with the protein
|
|
CO-Hb in smokers
|
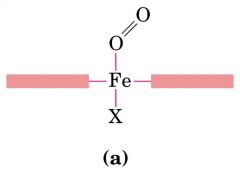
CO2 bind to Fe2+ more tightly than O2 --- CO bind to Fe2+ strongest (straighter) ---- nonsmoker 1% vs 10% of CO in smoker
|
|
|
MetHb (Fe3+)
|
is oxidated from "normal" Fe2+ --- can't bind to O2 and CO2 --- only bind to CN --
|
|
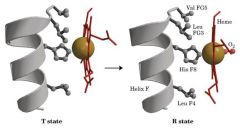
R state relax have more room to hold on ..........
|
Oxygen
|
|
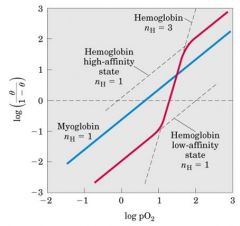
Hb O2 binding cooperativity
|
bind one and then all 4
|
|
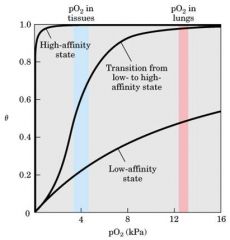
Myosin - Hb Oxygen binding curve
|
Muscle Myo bind more than Hb ----- lung bind the same
|
|
|
BPG is -2 in LOW pH (His is POSitve) will ......
|
bind to His stronger than O2 --- low pH = high CO2
|
|
BPG is -2 in HIGH pH (His is NEUtral) will ......
|
not bind to Hb and O2 will take over
|
|
|
Acidosis vs alkalosis
|
COPD (too much CO2 in blood) vs hyperventilation (too much O2 in blood - high pH - BPD can't kick out O2 for brain to use)
|
|
|
HbF vs HbA
|
HbF bind to O2 stronger (less aff to BPF) than HbA and act like Myoglobin to take O2 from O2
|
|
|
Tay-Sachs diseases
|
Hexo-saminidase-A (accumulate lipid on brain)
|
|
|
Met-Hb (Fe3+) is a "spontaneous" products of .....
|
Fe2+ ---- it does not bind oxygen but Cyanide (which doesn't bind to Hb-Fe2) ------------------ P
|
|
|
Amyl nitrite is used to treat ....... (metHb good side)
|
cyanide poisoning by converting Hb-Fe2 to metHb-Fe3 (Cyanide will release Fe-Cu center in site 4 of ETC and bind to metHbFe3) Nitrite will oxidize (take an e) Fe2 to Fe3
|
|
|
cyanide poisoning
|
cyanide will bind to Fe-Cu center sub unit in Site 4 and prevent Cyt C enzyme from passing e- to O2 (via Fe-Cu) --> can't use O2 ETC is shut down at site 4
|
|
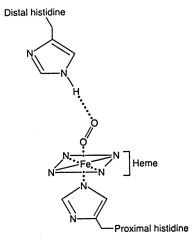
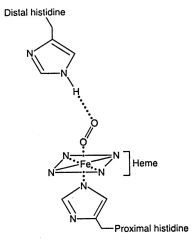
When oxygen binds to a heme-containing protein, the two open coordination bonds of Fe2+ are occupied by:
|
one O2 molecule and one amino acid atom.
|
|

Myoglobin and the subunits of hemoglobin have
|
very similar tertiary structures, but different primary structures
|
|
|
The interactions of ligands with proteins
|
are usually transient - transferable.
|
|
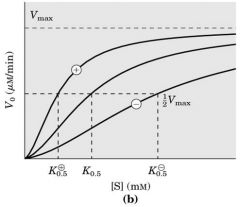
In the binding of oxygen to myoglobin, the relationship between the concentration of oxygen and the fraction of binding sites occupied can best be described as
|
hyperbolic.
|

