![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
556 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
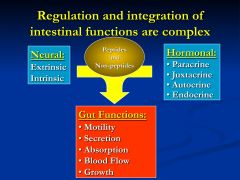
What are the functions of the gut?
What signals regulate and integrate these functions? |
|
|
|
|
What are the two types of enteric nerves?
|
Extrinsic and intrinsic
*extrinsic - from vagal or spinal nerves *intrinsic- part of the digestive tract |
|
|
|
Where are gut hormones derived from?
|
derived from extra and intra- intestinal endocrine cells
|
|
|
|
What are the two types of major regulatory mediators of the gut?
|

Peptides vs. Non-peptides
|
|
|
|
What are the examples of peptide regulating factors?
|
gastrin, secretin, vasoactive intestinal peptide (VIP)
|
|
|
|
What are the examples of the growth and trophic factor regulating factors (peptides)?
|
Insulin, TGF-alpha, and IGF
|
|
|
|
What are the steroid regulatory factors?
|
Vitamin D, Aldosterone, Hydrocortisone
|
|
|
|
What are the amino acid derivate regulatory factors?
|
NO, NE, Epi, Histamine, and Serotonin
|
|
|
|
What are the phospholipid derived regulatory factors?
|
Arachidonic acid and Platelet activating factor (PAF)
|
|
|
|
What are the major families of gut regulatory peptides?
|

Gastrin and Secretin Families are most important
|
|
|
|
How are gut neurotransmitters grouped?
|

Made/Stored/ Released by enteric neurons
|
|
|
|
What effect does cholera toxin have on G-proteins?
|
Cholera toxin inhibits the exchange of GTP for GDP
*so the Gs is persistently active |
|
|
|
What are the two types of guanylate cyclases?
|
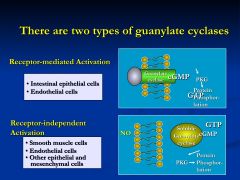
|
|
|
|
Where is the guanylin receptor located?
|
expressed on the luminal membrane of gut epithelial cells
*activated by either guanylin (gut hormone) or heat stable enterotoxin (STalpha- E.coli) |
|
|
|
Once the guanylin receptor is activated what action occurs?
|
The guanylate cyclase converts GTP to cGMP
*this leads to net secretion of water and electrolytes and the development of diarrhea (TRAVELER'S DIARRHEA) |
|
|
|
How does the receptor independent activation of guanylate cyclase occur?
|
NO moves through membrane and acts on the soluble receptor protein
*GTP - cGMP-PKG-Protein phosphorylation = diarrhea |
|
|
|
Do some intestinal growth factors result in activation of other pathways?
|
YES!
EGF, PDGF, and IGF activate membrane tyrosine kinase receptors |
|
|
|
What is the important role of Arachidonic Acid?
|
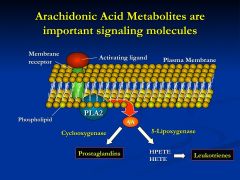
Hormone and neurotransmitter activation of AA metabolism result in the production of prostaglandins and leukotrienes
*which are important intracellular and extracellular signaling molecules |
|
|
|
What mechanisms are part of the extrinsic regulation pathways?
|
endocrine
paracrine autocrine juxtacrine neurocrine |
|
|
|
Define endocrine pathway?
|
regulatory hormones are secreted into the blood stream and delivered to distant targets
*multiple tissues and cell types can be regulated simultaneously *ex: gastrin |
|
|
|
Define the paracrine pathway?
|
hormones stored in secretory granules at the basal pole are released and stimulate receptors on the cells immediately adjacent
*this is how mucosal endocrine cells that are interspersed between intestinal epithelial cells exert their regulatory effects |
|
|
|
Define the autocrine pathway?
|
its release stimulates/autoregulates its on behavior and function
|
|
|
|
Describe the juxtacrine pathway?
|
regulatory agents that affect many types of cells that are in close proximity
*histamine secreted by lamina propria - affects mucosal, blood flow, and smooth muscle |
|
|
|
Explain the neurocrine pathway?
|
utilizes the enteric neurons
synapse directly with the lining cells of the gut to activate specific receptors of target tissues *specificity |
|
|
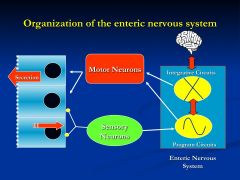
Organization of the Enteric nervous system?
|
Enteric nervous system is the gut's own nervous system
*produces a very patterned and predictable response *sensory neurons will set into the circuitry of events to produce an effect that is integrated with other motor neurons *this system does receive some signals from the brain |
|
|
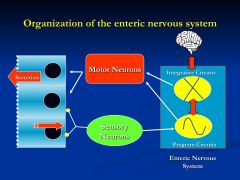
Organization of the Enteric nervous system?
|
Enteric nervous system is the gut's own nervous system
*produces a very patterned and predictable response *sensory neurons will set into the circuitry of events to produce an effect that is integrated with other motor neurons *this system does receive some signals from the brain |
|
|
|
What two aspects are integrated for intestinal function?
|
Enteric Neurons and Endocrine cells of the gastrointestinal mucosa
|
|
|
|
Where can endocrine and enterochromaffin cells be found?
|
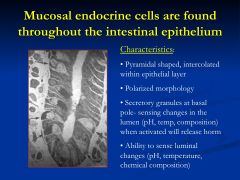
these cells are found intercalated among epithelial cells
*secretions are paracrine in fashion |
|
|
|
Is the distribution of most mucosal endocrine cells random?
|
No! the region specific expression is relevant to their primary physiological actions
|
|
|
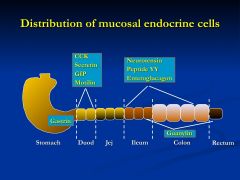
What is this slide showing?
|
How regional specificity is very important
*Gastrin- distal 1/3 of the stomach *Duod and prox jej - CCk, Secretin, GIP, and Motilin *Ileum and colon - neurotensin, peptide YY, enteroglucagon *Colon - Guanylin |
|
|
|
Describe the digestion of a meal?
|
1. Stomach expands and this activates the baroreceptors - leading to the secretion of gastrin
2.endocrine gastin (luminal nutrients) enter circulation and is carried back to the proximal 2/3 of the stomach which stimulates the secretion of gastric acid (hydrochloric) - pepsin also now secreted -increases motility 3) Gastric chyme stimulates the duodenal release of GIP, CCK, and secretin 4)CCK and secretin stimulate the pancreatic and biliary secretions - which are very important for normal digestion 5) GIP, CCK, and secretin feedback inhibit gastric secretions *GIP also slows gastric emptying - efficient digestion of all nutrients |
|
|

What does this patient have?
|
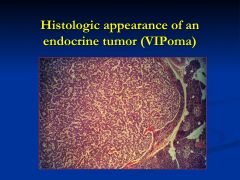
rare endocrine tumor
- makes are secretes larges amounts of gut peptides: VIP, motilin, and neurotensin |
|
|
|
What does this patient have?
|
rare endocrine tumor
- makes are secretes larges amounts of gut peptides: VIP, motilin, and neurotensin |
|
|
|
Why is facial flushing and hypotension seen with VIPoma?
|
VIP causes vasodilation and one of its manifestations is facial flushing and hypotension
|
|
|
|
What does VIP normally do?
|
it regulates numerous intestinal functions: motility, intestinal water and electrolyte transport, and blood flow
*potent secretagogues - this action is stimulated by adenylate cyclase and increased cAMP |
|
|
|
What is the mechanism of action for VIP?
|
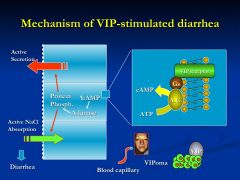
VIP stimulates VIP receptors
activating adenylate cyclase *leading to active water secretions *inhibits absorption *activate vasodilation |
|
|
|
Why does hypokalemia occur with VIPoma patients?
|
stool losses and the chronic effects of aldosterone, that is secreted in response to hypovolemia.
*Aldosterone increases renal absorption of Na at the expense of increased K excretion |
|
|
|
How would you treat VIPoma?
|
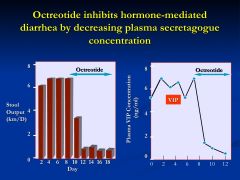
with somatostatin analog = Octreotide
*inhibition of hormone release is the major mechanism this drug uses |
|
|
|
What are the effects of somatostatin on the GI tract?
|

|
|
|

What does this patient have?
|
Celiac Sprue
|
|
|
|
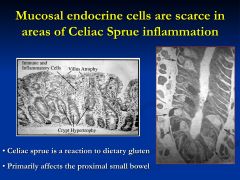
What is normally secreted where Celiac sprue has its major effects?
|
affects the duodenum and proximal jejunum
*where secretin and cck is made * aren't have the release of pancreatic and biliary secretions |
|
|
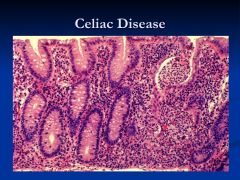
what are the histological characteristics of celiac disease?
|
*small intestine mucosal inflammation
*villus atrophy (shortening or absence) * crypt hypertrophy * lamina propria packed with immune cells |
|
|
|
What comprises the innervation to the gut?
|
The extrinsic sympathetic and parasympathetic fibers & the enteric nervous system
|
|
|
|
What are the parasympathetic fibers to the gut?
|
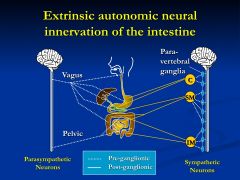
vagus and pelvic nerves
*pelvic supply distal colon and rectum *synapse with post-ganglionic parasympathetics or enteric neurons located in intestinal wall plexus |
|
|
|
What are the sympathetics to the GI tract?
|
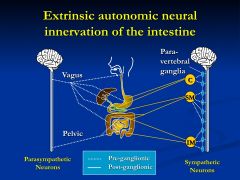
pre-ganglionic efferent fibers from spinal cord synapse at paraveterbral ganglia and post ganglionic project into intestine
|
|
|
|
Where is the enteric nervous system (ENS) found?
|
Intrinsic enteric neurons are found in the gut wall located in two major plexuses ( Auerbach's and Meissner's)
|
|
|
|
Where is Auerbach's plexus is located where?
|
found between the circular and longitudinal muscle layer
|
|
|
|
Where is the Meissner's plexus found?
|
located in the submucosa
|
|
|
|
What does the diversity of ENS neurons reflect?
|
The wide range of the physiological functions of the GI tract
|
|
|
|
Propulsive motor contractions of the intestine require the coordination of?
|
Excitatory and inhibitory enteric neurons
|
|
|
|
What function do the interneurons in the ENS provide?
|
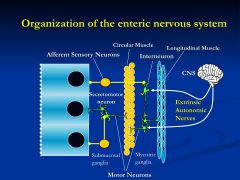
interneuronal connections that permit complex and integrated responses involving many digestive processes
|
|
|
|
Why are the functions of the guy hardwired?
|
Because timing is everything in the gut
|
|
|
|
Describe the events of Peristalsis?
|
1) intestinal contents distend the lumen -stimulating intramural baroreceptors
2) activating excitatroy motor neurons = contraction *relaxation of longitudinal and contraction of circular muscle 3) at the same time there are inhibitory neurons in the segment immediately distal to the contracting muscle *prepares the receiving segment for luminal contents **events NEED to be coordinated |
|
|
|
Explain the events of Peristalsis?
|
1) the distention of the lumen by intestinal contents stimulates intramural baroreceptors
2) these receptors activate excitatory motor neurons that cause contraction (relaxation of longitudinal and contraction of circular) 3) at the same time, inhibitory neurons in the segment immediately distal to the contracting segment are relaxed **this prepares the the receiving segment to take on the luminal contents |
|
|
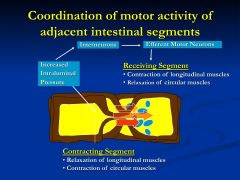
what is this slide showing?
|
Steps of Peristalsis
|
|
|
|
Where is smooth muscle and striated muscle found within the gut?
|
Skeletal muscle is found in the upper third of the esophagus and the external anal sphincter
**everywhere else is smooth muscle |
|
|
|
How can the smooth muscle of the gut contract as a single unit?
|

* especially in circular smooth muscle cells, as annular contraction requires simultaneous activation of all muscle fibers in that region
|
|
|
|
Describe the longitudinal muscle of the GI tract?
|
oriented with their axis running along the length of the bowel
* in colon, they are organized into a separate thick cords = teniae coli |
|
|
|
What are the two types of electrical activity seen in the intestinal smooth muscle?
|
1) BER
2) spike potnential |
|
|
|
What are BER's and where are they found?
|
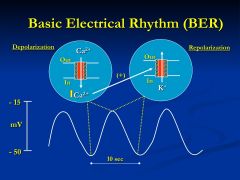
Basic electrical rhythm = BER
found throughout except in esophagus and proximal stomach |
|
|
|
Where does the BER arise from?
|
from specialized "pacemaker" cells throughout the GI tract
*BER frequency is different in different parts of the gut |
|
|
|
What is the relationship between contraction and BER
|
They serve as setting a certain beat and under certain circumstances where contraction is activated then you will see the contraction with BER
* BER =10 so the most contractions you can have is 10 the least is 0 but never more than 10 |
|
|
|
What greatly affects the appearance and frequency of spike potentials?
|
hormonal and neurotransmitter signals
** greater number of spike potentials = greater the degree of muscular contraction |
|
|
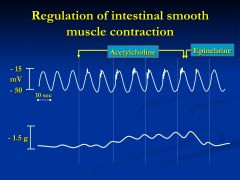
What is this slide showing?
|
muscarinic receptor agonist = Ac is added which provides increased spike potentials at the top of the depol phase - increase in muscle contraction
VS. when Epi is associated then see disappearance of spike potentials - decreasing muscle contraction **Neither agent affects BER frequency |
|
|
|
Regulation of intestinal motor function
*excitatory agents |

increase cytosolic calcium
|
|
|
|
Regulation of intestinal motor function *inhibitory
|
-agents increase cyclic nucleotide levels in smooth muscle cells cause relaxation
|
|
|
|
Regulation of intestinal motor function
*inhibitory |

- agents that increase cyclic nucleotide levels in smooth muscle cells cause relaxation
|
|
|
|
Describe the Motility pattern of segmentation?
|
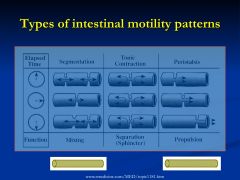
Non-propulsive, annular contractions of the circular muscle layer, found in small and large intestine
*important for mixing intestinal chyme with digestive juices (squeezing) - puts food into a state that it can be effectively absorbed |
|
|
|
Describe the motility pattern of Tonic Contractions?
|
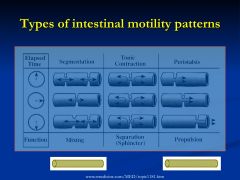
Zones of increased and continuous pressure that function as sphincters
*important for seperating and regulating intestinal flow into various compartments *relaxation allows gated passage of luminal contents to another compartment |
|
|
|
Describe the motility pattern of peristalsis?
|
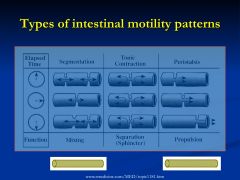
highly integrated, complex motor pattern characterized by sequential annular contraction of gut segments
*producing a sweeping propulsive wave |
|
|
|
Primary Esophageal Peristalsis
|
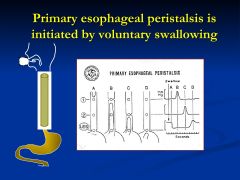
Propulsive, sequential, annular contractions that follow a swallow (2-4 cm/sec)
*The LES relaxes to let food enter - the peristaltic wave stops at the LES and doesn't continue into the stomach |
|
|
|
Explain Secondary Peristalsis
|

Propulsive wavelike contractions that are initiated in the absence of swallowing and by increased luminal pressure
*serves to clear retained food and fluid or remove refluxed gastric contents from the esophagus |
|
|
|
What are the two important roles of the LES?
|
allowing food to enter the stomach and preventing the reflux of gastric contents
|
|
|
|
What is reflux esophagitis?
|
inflammation and structure formation that results from inadequate sphincter pressure
|
|
|
|
What is achalasia?
|
arises when the LES pressure is abnormally high, preventing entry of food into the stomach and causing dilation of the proximal stomach
|
|
|
|
What are the regulators of LES pressure and function?
|

|
|
|
|
What are the three major functions of the stomach?
|
1) reservoir for food
2) mixes the meal with gastric secretions to initiate digestion 3) gated release of small amounts of gastric chyme into the small intestine for further digestion and absorption |
|
|
|
Describe the Reservoir function of the stomach?
|
Upper 1/3 of the stomach serves as a reservoir
**Receptive Relaxation - allows it to accommodate large gastric volumes w/o a large increase in pressure |
|
|
|
Which nerve regulates receptive relaxation of the upper 1/3 of the stomach?
|
Vagal nerve
-vagotomy - reduces distensibiltiy and you feel full quicker |
|
|
|
Describe the mixing function of the stomach?
|
distal 2/3 of the stomach
- mixes gastric contents with digestive juices (through peristalsis) **regulated by primary pacemaker |
|
|
|
What and where is the primary pacemaker of the stomach?
|
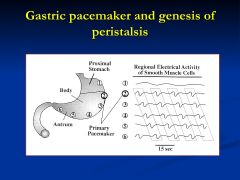
located in the mid-portion of the greater curvature of the stomach
- phase lag in distal segments of BER in the stomach - this allows waves to being in upper regions and propagate down |
|
|
|
Describe the grinding and gated release of gastric contents within the stomach?
|
Gastric peristalsis through the antrum coupled with increased pyloric sphincter tone
- large food particles reflex back into gastric lumen to undergo additional breakdown - so only small amount enter intestine at one time |
|
|
|
What is dumping syndrome?
|
When gastric emptying is rapid and inappropriate
*characterized by nausea, sweating, abdominal cramps, and weakness |
|
|
|
What is Gastroparesis?
|
condition associated with severe diabetic patients who have autonomic neuropathy
* caused by impaired or absent gastric motility and delayed emptying of the stomach * will experience post-prandial (after meal) bloating, nausea, and early satiety |
|
|
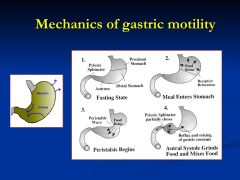
What is the second step?
|
2) Receptive relaxation or the upper 1.3 of the stomach allows food to stored wtihout significant increase in intra-gastric pressure
|
|
|
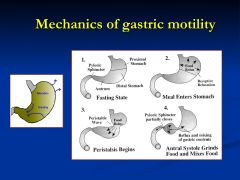
Explain the 3rd step
|
Boluses of food are mixed with gastric juices and moved towards the antrum
|
|
|
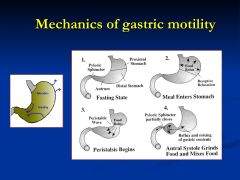
Explain step 4
|
As peristalsis sweeps through the antrum (antral systole) the pyloric sphincter is maintained in a partially open state, only allowing liquefied contents to enter the small intestine. Larger food particles reflux for further grinding and mixing
|
|
|
|
What is the purpose to small intestinal motility?
|
major site of digestion and absorption of nutrients
* motility patterns found here are primarily for mixing and propulsion of luminal contents |
|
|
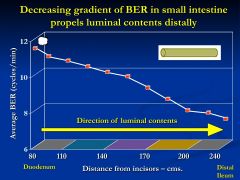
what is this slide showing?
|
Decreasing gradient of BER frequencies promotes distal movement of intestinal contents
|
|
|
|
What determines the max rate of segmentation of that region?
|
BER
|
|
|
|
What are agents will increase spike potentials = increasing likelihood of contraction?
|
Ach
Bombesin CCK |
|
|
|
What are the inhibitory agents of spike potential and therefore contractions?
|
Alpha-adrenergics
NO VIP glucagons |
|
|
|
What are Migrating Motor Complexes(MMC)?
|
A motor pattern only seen during fasting or inter-digestive periods that producing a wavelike contraction running the entire length of the bowel
|
|
|
|
Why are MMC thought to be important?
|
sweeping the lumen free of content and preparing the small intestine for receipt of the next meal
|
|
|

|
Good Job
|
|
|

|
Has 3 different phases
In fasting state- phase 3 *MMC disappears during meal and reappears after a meal |
|
|

What is this showing?
|
Gastrin or CCK initiate the initially phases of MMC
*hormones from the early phase of digestion *this is showing the possibly that gastrin and CCK are the main reason MMC's stop with meals |
|
|
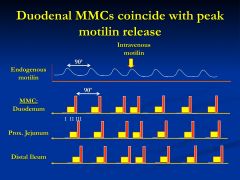
|
Motilin will reset the MMC
- 90 min phase is shortened and then reset - Initiation may be hormone-dependent |
|
|
|
How much does the colon receive per day?
Consisting of what? |
0.5-2.5 L /day
* undigested and porrly absorbed food residue, water, electrolytes |
|
|
|
What does the colon need to reduce the load to so diarrhea doesn't result?
|
200 gm/day
|
|
|

|
THe movements of the colon are slow and irregular and aimed at increasing contact time between luminal fluid and the absorptive mucosal surface
|
|
|
|
What are teniae coli?
|
are the longitudinal muscles that are organized into three seperate bands
*change the length of the colon |
|
|
|
What are haustrations?
|
Segmentation of the colon
*segmentation is the major motor activity of the colon |
|
|
|
Are rectal motility and anal sphincter function distinct from colonic motility?
|
Yes
|
|
|
|
How does the rectum function as a storage site
|
Receptive relaxation
- allows accommodation of large stools |
|
|
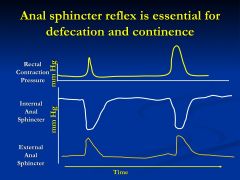
|
when rectal contraction pressure increases (intraluminal baroreceptors)
- the internal sphincter relaxes and the external sphincter contracts |
|
|
|
What is Valsalva maneuver?
|
individual initiates defecation by voluntarily decreasing the tone of the external anal sphincter and increasing intraabdominal pressure by forcing air against the closed glottis
|
|
|

|
-compromised LES function
- autonomic neuropathy can also affect gastric emptying b/c the stomach becomes flaccid and antral peristalsis is lost |
|
|
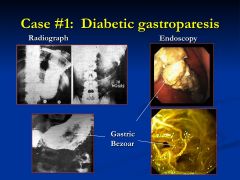
What is diabetic gastroparesis?
|
Gastroparesis can result in the formation of a bezor = conglomerated mass of undigested vegetable and fruit fiber
|
|
|
|
Diabetes induced autonomic neuropathy can have similar effects as what?
|
The surgical interruption of the vagus nerve
|
|
|
|
What symptoms can autonomic neuropathy lead to?
|
intestinal stasis
- overgrowth of bacteria --nutrient malabsorption and diarrhea severe constipation or diarrhea loss of anal sphincter tone |
|
|
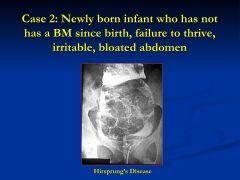
What is this caused by?
|
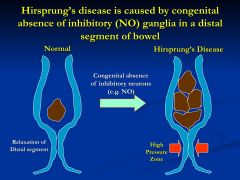
|
|
|
|
What is Achalasia?
|
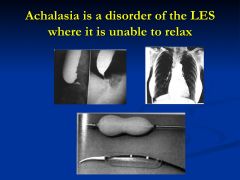
neuromuscular that affects LES
failure of LES to relax -decrease NO and VIP signaling |
|
|
|
What is coffee ground emesis indicative of?
|
blood digested in HCl and emesis means throw up
|
|
|
|
What is tarry stool indicative of?
|
bleeding in the upper GI tract
|
|
|
|
What does orthostatic mean?
|
bp drops upon standing
- due to the loss of blood |
|
|
|
Why is the secretion of intrinsic factor in the stomach so important?
|
it is required for the absorption of vitamin B12
|
|
|
|
What are the divisions of the stomach?
|
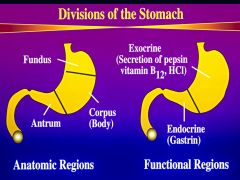
endocrine region is in the antral mucosa = gastrin
endocrine - fundus and body = HCl, pepsin, and intrinsic factor |
|
|
|
What is one main characteristic of the fundus?
|
Receptive relaxation
|
|
|
|
What is the basic unit of the stomach?
|
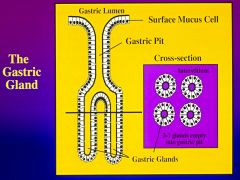
|
|
|
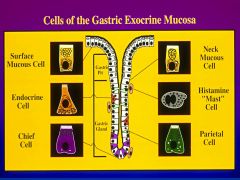
|
*Surface mucus cells- simple columnar = make a secrete mucus and alkaline fluid for protecting the stomach
* Gastric Pits- conduits for delivering the secretions (3-7 glands) - 50% of total luminal area * Gastric gland - many different cell types- germinal cell proliferation in this area |
|
|
|
Parietal cells- where are they found and what do they produce?
|
produce HCl acid and intrinsic factor
found in the upper gland |
|
|
|
Mucous cells- where are they and what do they produce?
|
mucus neck cells are found in the upper gland and throughout the gland
make mucus and bicarbonate * protect stomach against auto digestion *mucus also acts as a lubricant |
|
|
|
Chief cells- where are they and what do they do?
|
They are found near the base and make and secrete pepsinogen
|
|
|
|
Mast cells- where are they and what do they do?
|
basal side of the epithlium
*regulates gastric acid and pepsin secretions *secrete histamine (juxtacrine) that affects gastric glands |
|
|
|
Endocrine glands - where are they and what do they do?
|
are between chief and parietal cells
* play a major role in regulating chief and parietal cell secretory function *secrete bioactive amines and peptides that stimulate the other cells through a paracrine action |
|
|
|
What are two specialized endocrine cells?
|
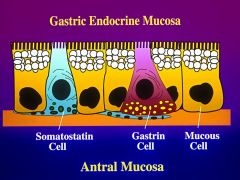
G-cells are gastrin containing - regulate gastric acid
basal side contains secretory granules and apical side is narrow * secreted from the endocrine portion of the stomach and reaches the exocrine portion of the stomach in this fashion D cells- make and secrete somatostatin dendritic like membranes - contacting adjacent cells - rapid delivery of somatostatin in paracrine fashion |
|
|
|
describe the morphological change parietal cells undergo?
|
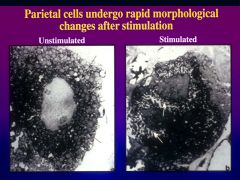
resting- tubovesiclular bodies and large mitochondria are present - canaliculi are lined by short stubby microvilli
activation - rapid transformation - prepare for acid secretion tubovesicular bodies fuse with the canulicular or lumenal membrane and increase surface area 6-10 fold **rapid insertion of proton pumps |
|
|
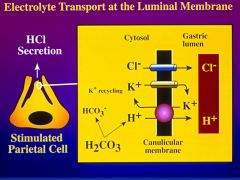
Describe all that is going on in this slide?
|
this is an electroneutral membrane transporter
to move H+ in K+ is moved out = requires ATP - this pump is only active after the tubovesicular body has inserted into the canalicular membrane * H+ is generated from carbonic acid due to the activity of carbonic anhydrase *K+ needs to me on the side of the lumen to be pumped out so it moves down its conc. gradient into the lumen * Cl helps maintain electroneutrality and also produces HCl on lumenal side |
|
|
|
Regulation of K+ and Cl channels on the canulicular membrane may help determine what?
|
May determine rates of acid secretion
|
|
|
|
How do Proton Pump inhibitors work?
|
weal acids that are selectively taken up and concentrated in the acidic canaliculi or gastric parietal cells
- then converted to sulfonamides that react with the thiol groups *bind to the alpha subunit of the proton pump and block its function |
|
|
|
What are the three major activators of acid secretion for the parietal cell?
|
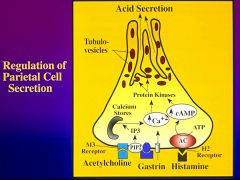
Gastrin, Histamine, and Acetylcholine
|
|
|
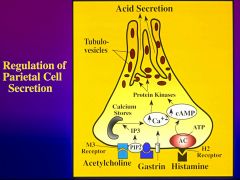
describe the histamine receptor
|
Histamine stimulates the H2-type receptor and activates adenylate cyclase
*increases cAMP- dependent protein kinase = initiating acid secretion |
|
|
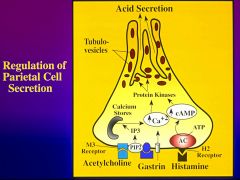
Describe the gastrin receptor
|
gastrin stimulates the gastrin receptor which are coupled to the PIP2 hydrolysis
Increase in cytosolic Ca++ (IP3) activate Ca -dependent protein kinases synergistic with cAMP pathways |
|
|
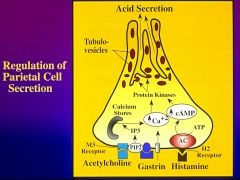
Describe the Ach receptor
|
Ach stimulates a type 3 muscurinic receptor (M3) which are also coupled to PIP2 hydrolysis - increasing cytosolic Ca
|
|
|
|
What accompanies gastric acid secretion?
|
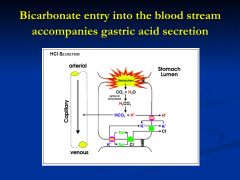
generating a proton for acid secretion through carbonic anhydrase also forms bicarbonate which moves into the blood stream
|
|
|
|
What are the other important pathways regulating gastric acid secretion?
|
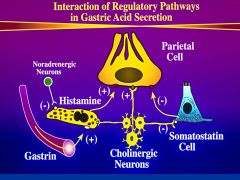
1) gastrin secreted by antral G cells
2) Ac from cholinergic neurons - stimulate mast cells - secretion of histamine 3) Somatostatin is the major paracrine agent that inhibits gastric acid secretion |
|
|
|
PNH: Clinical Features
|
(1) Chronic intravascular hemolysis causing chronic paroxysmal hemoglobinuria, elevated LDH;
(2) Normochromic, normocytic anemia (unless IDA is also present); (3) Pancytopenia; (4) Thrombosis of venous systems can occur - eg. of the hepatic veins (Budd-Chiari syndrome); (5) May evolve into aplastic anemia, myelodysplasia, myelofibrosis, and acute leukemia; (6) Abdominal, back, and musculoskeletal pain |
|
|
|
The Gastric Acid secretion can be divided into how many phases?
|
3 phases
-cephalic -gastric -intestinal |
|
|
|
Describe the cephalic phase of gastric secretion?
|
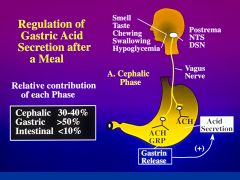
evoked by a stimuli from the central nervous system - can be initiated by sight, smell, and taste of food
|
|
|
|
What are the three important areas of the brainstem that relay messages to the stomach?
|
Postrema
nucleus tractus solitarii (NTS) dorsal motor nucleus (DMN) *preganglionic vagal nerves relay message to the enteric neurons - stimulating gastrin realeasing peptide and ACh -= release gastrin from antral G-cells |
|
|
|
Describe the gastric phase of gastric acid secretions?
|
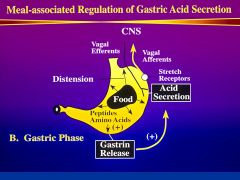
accounts for 50% of total acid
starts when fluid or food enter stomach * as distention increases - gastric output increases (antrum) - response mediated by vagal nerve fibers and local factors (gastrin) |
|
|
|
The presence of {peptides and amino acids} or {carbohydrates and fats} stimulate further gastric acid secretions?
|
peptides and amino acids further stimulate gastric acid secretions
|
|
|
|
Describe the intestinal phase of gastric acid secretion?
|
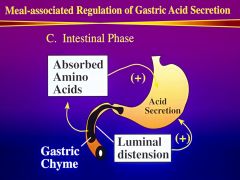
least important phase <10%
stimulated by luminal distention and absorption of amino acids |
|
|
|
What can the cephalic phase of gastric acid secretion be inhibited by?
|
intracerebral-ventricular injection of agents like:
-bombesin -neurotensin -cotricotropin-releasing factor (CRF) * important modulators |
|
|
|
What can the gastric phase of gastric acid secretion be inhibited by
|
increased luminal acidity (especially in the antrum)
- mediated by enteric neurons and increased release of somatostatin by D cells |
|
|
|
What can the intestinal phase of gastric secretion by inhibited by?
|
luminal presence of acid
hyperosmolar solutions luminal fat * stimulates: GIP, neurotensin,somatostatin, secretin, VIP,, PYY |
|
|
|
What is the most important digestive enzyme secreted by gastric chief cells?
|
Pepsinogen
|
|
|
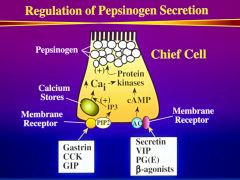
Regulation of pepsinogen secretion
|
pepsiogen secretion parallels gastric acid secretion since many of the regulating agents are the same
|
|
|
|
What does an increase in cAMP do for the chief cell?
|
stimulates the release of pepsinogen but also increases pepsinogen synthesis
(VIP, secretin, PGE, Beta-antagonist) |
|
|
|
How is luminal pepsiogen activated?
|
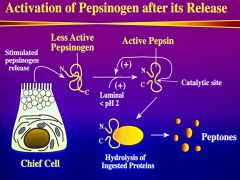
pH of 2 - active
*acid is required for acitivation conformational change - opens catalytic site *pepsin has a positive feedback effect |
|
|
|
What does pepsin do?
|
it hydrolyzes ingested proteins into peptons - which are potent signals for gastrin and CCK
*pepsin has a high affinity for collagen (works more on meat than vegetables) |
|
|
|
What function does mucus serve in the stomach?
|
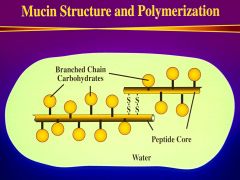
Protects against autodigestion
*Mucin- glycoproteins that are heavily glycosylated - linked through disulfide bonds *forms a gel (80% water) |
|
|
|
Purpose of bicarbonate secretion?
|
lining cells and creating buffer zone
* dependent on carbonic anhydrase *stimulated by vagal nerves and E-series prostaglandins |
|
|
|
What is the mucus gel buffer zone?
|
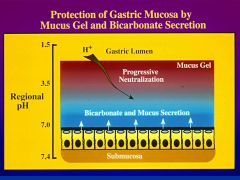
mucus gel forms a semi-impermeant layer that protects underlying cells from the acidic lumen and retains bicarbonate secreted from underlying epithelial cells (alkaline)
|
|
|
|
Factors affecting the buffer zone...leading to peptic ulcer development?
|
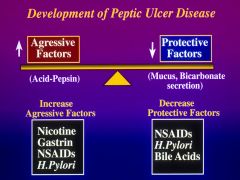
Balance between aggressive and protective factors
|
|
|
|
What is used to treat peptic ulcer disease?
|
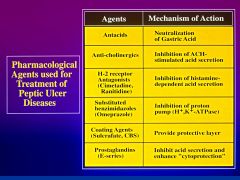
|
|
|
|
How do proton pumps work?
|

Made as a pro-drug then collect and aggregate where pH is less than 3
customized drugs that will accumulate in the lumen *acid converts them to active form - sulfinamide- which bind the thiol groups of the pump and inactivate it |
|
|
|
What is H. pylori?
|
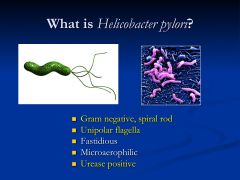
most common cause of duodenal ulcer disease
*can survive in harsh acid milieu due to its expression of urease, an enzyme that makes NH3 that can buffer protons ** stimulates a mucosal inflammatory response, stimulates acid secretion, and compromises barrier function |
|
|
|
What does NSAIDs do to the GI tract?
|
aspirin and motrin cause GI bleeding
*NSAIDs also stimulate acid secretion |
|
|
|
What is Gastrinomas?
*Clinically described as Zollinger-Ellison syndrome |
endocrine tumors that secrete gastrin and cause hypergastrinemia
* consequences: hyper-secretion of acid and pepsin *leading to diarrhea and recurrent peptic ulcer disease |
|
|
|
Composition of the pancreas?
|
exocrine and endocrine function
80% of the total volume is due to glandular tissue endocrine cells only account for 2% |
|
|
|
What is the primary functional unit of the pancreas?
|
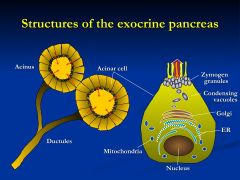
Acinus - composed of contiguous, pyramid-shaped glandular cells with apex facing the lumen
|
|
|
|
Are acini polarized cells?
|
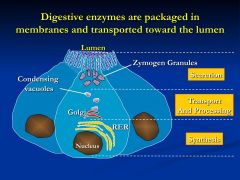
Yes. They have distinct functional and structural differences in the apical and basolateral domains
*have developed RER and Golgi for synthesis and storage of digestive proteins *Zymogen or storage granules are found in the apical or luminal pole- released into the ductule by exocytosis - then membrane vesicles are recycled |
|
|
|
What do the acinar cells produce?
|
digestive enzyme proteins that are eventually transported through the ductular system into the duodenum to be mixed with intestinal chyme
|
|
|
|
How is acinar cell secretion regulated?
|
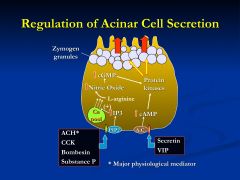
ACh, CCK, Bombesin, Sub P act on PIP2 increasing Ca
*cholinergic muscarinic receptors appear to be the dominant pathway for Ca-mediated Secretin and VIP - A.C - increasing cAMP |
|
|
|
When calcium is released what does it trigger?
|
Ca stimulates nitric oxide synthase (NOS), leading to the formation of NO
* NO activates guanylate cyclase activity to increase cellular cGMP levels which release zymogen granules |
|
|
|
Which is more powerful for pancreatic secretions: cAMP or Ca++?
|
Ca+ mediated agonist is more importnat than cAMP mediated agonists in regulating acinar cell functions
|
|
|
|
Types of Pancreatic enzymes?
|

most digestive enzymes are made and stored in their inactive proenzyme form
*once secreted into duodenal they are converted to their active forms |
|
|
|
What is the master pancreatic enzyme?
|
Trypsin- essential for hydrolytic activation of other proenzymes in the duodenal lumen
|
|
|
|
Where do exopeptidases and carboxypeptidases cleave?
|
They cleave proteins fro their ends
|
|
|
|
Proteolytic enzymes make up ___% of all the protein found in pancreatic juice?
|
80%
|
|
|
|
What are the lipolytic enzyme and what do they do?
|
involved in lipid digestion
-lipase -phospholipase -carboxyesterase |
|
|
|
What do amylolytic enzymes work on?
|
spillting 1-4 glycosidic linkages
- important for carbohydrate digestion |
|
|
|
What do nucleases do?
|
digest nucleic acids
|
|
|
|
What is the purpose of trypsin inhibitor?
|
provides extra protection to acinar cells in the event active trypsin forms within zymogen granules - this would have devastating events
|
|
|
|
How does the pancreas protect itself from pancreatic enzymes?
|
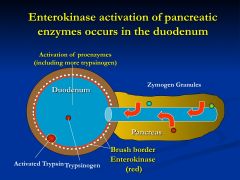
1)made in pro-enzyme
2) packaged into zymogen granules 3) co-packing with trypsin inhibitor 4) geographical (pancreas-duct- duodenum) |
|
|
|
Trypsinogen is activated how?
|
Enterokinase (enteropeptidase) is a brush border enzyme that converts trypsinogen to trypsin
|
|
|
|
What does trypsin do?
|
activates other proenzyme
|
|
|
|
What is the difference between ductular and acinar secretions?
|
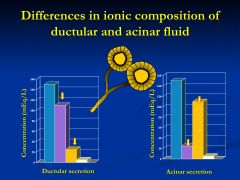
ductular secretion is HCO3-rich
whereas acinar fluid has a plasma like electrolyte composition |
|
|
|
Why are ductular secretions high in HCO3?
|
important for neutralizing gastric acid entering the duodenum
|
|
|
|
What is the purpose of acinar fluid?
|
essential for transporting secreted enzymes into the ductular system
|
|
|
|
What determines the final concentration of pancreatic juices?
|

|
|
|
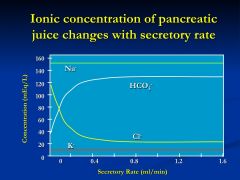
explain this slide?
|
- at low flow rates, pancreatic juice is mostly NaCl with small K and HCO3
- as rates increase, an inverse relationship between Cl and HCO3 - juice becomes progressively more alkaline * K and Na concentrations are independent of secretory rate |
|
|
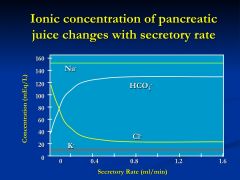
explain this slide?
|
- at low flow rates, pancreatic juice is mostly NaCl with small K and HCO3
- as rates increase, an inverse relationship between Cl and HCO3 - juice becomes progressively more alkaline * K and Na concentrations are independent of secretory rate |
|
|
|
What is the major regulator of ductular fluid secretion?
|
Secretin
- made by endocrine cells and is a potent stimulus of pancreatic bicarbonate secretion |
|
|
|
What are the phases of pancreatic function?
|
1) cephalic - 50%
2) gastric - 10% 3) intestinal - most important |
|
|
|
Describe the cephalic phase of pancreatic function?
|
initiated by the CNS
-activation of pancreatic functions occurs by stimuli from efferent vagal fibers -promotes secretion of enzymes and bicarbonate |
|
|
|
Describe the gastric phase of pancreatic function?
|
least important phase - 10%
* initiated by stomach distention and by the presence of aa and peptides ** have little effect on bicarbonate secretion |
|
|
|
Describe the intestinal phase of pancreatic secretion
|
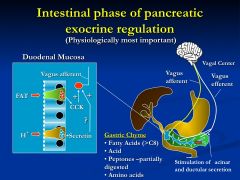
initiated by gastric chyme into the intestinal lumen (especially fat acids)
*fatty acids and H+ stimulate mucosal endocrine cells secretion of CCK and secretin (juxtacrine action) -- activates cholinergic enteric neurons |
|
|

|
bicarbonate release is proportional to acid load
* this process is tightly regulated by secretin - also has an inhibitory effect on gastric acid secretion and motility - allowing time for digestion **pH of the duodenum is neutral |
|
|
|
How is pancreatic function returned to normal?
|
Through feedback inhibition of pancreatic function
* presence of active pancreatic protease inhibit pancreatic enzyme secretion |
|
|
|
What are the advantages of maintaining duodenal pH near neutral?
|

|
|
|

|

|
|
|

|
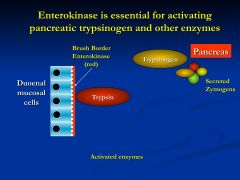
enterokinase is absent so the pancreatic enzymes are low because they are all still in their zymogen form
* enterokinase converts trypsinogen to trypsin which further activates the zymogens |
|
|

|
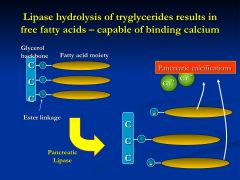
Chronic pancreatitis
|
|
|
|
What are the causes of chronic pancreatitis?
|

|
|
|
|
What is the pathophysiology of chronic pancreatitis?
|

|
|
|

|
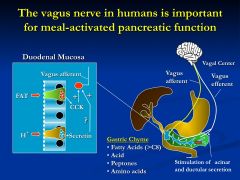
vagal nerve is essental for mediating CCK-stimulated acinar-cell zymogen secretions (secretin - bicarbonate)
* CCK is released by endocrine cells of the duodenal mucosa in response to luminal fat then CCK stimulates afferent vagal fibers that initiate the majority of stimulated acinar cell secretions |
|
|
|
What the three situations in which amylase will be in the urine?
|
1) duct obstruction
2) rupture 3) pancreatic destruction |
|
|
|
What can one suspect when amylase>> creatine in the urine?
|
Pancreatitis
- used as a diagnostic tool |
|
|
|
How does fluid absorption differ throughout the small intestine?
|
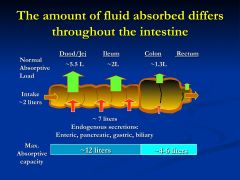
2L by mouth
7L by secretions = 9 L /day * most fluid is removed by Duod/Jej/Ileum 8.8 L gets absorbed of the 9 L that enters *just 1% off = then you will have symptoms |
|
|
|
Describe the permeability throughout the stomach?
|
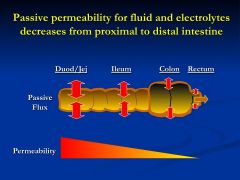
* small bowel must be "leakier" to accommodate rapid changes in water and electrolyte fluxes after a meal
*permeability of the colon and rectum must be less leaky to prevent back flux of absorbed water in electrolytes - property required for solidification of stool |
|
|
|
What are defensins?
|
They are secreted by paneth cells
* have antimicrobial properties |
|
|
|
What are enterocytes responsible for?
|
They are at the top of the villi and do the most secretion
|
|
|
|
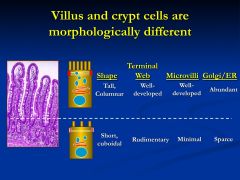
What are the functional differences between crypt cells and villi cells?
|
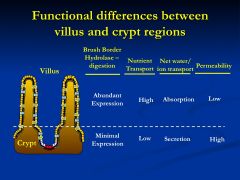
Crypt - short, immature, cubodial, rudimentary microvilli, and terminal web -- and tight junction complexes
* Permeability high to accommodate secretions Villi cells- tall columnar, mature cells, well developed terminal web,tight junctions *solubility low to protect against back flow |
|
|
|
Appreciate the difference
|
|
|
|
What role do blood vessels and lymphatics play for the intestine?
|
intestinal water and electrolyte transport
- absorption - carry away nutrients, water, electrolytes -secretion - deliver fluid and electrolytes |
|
|
|
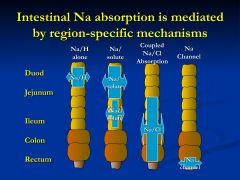
In the proximal small intestine how is Na absorbed?
|
Na-H exchangers function independently in the proximal small intestine
|
|
|
|
In the Ileum how is Na absorbed?
|
Na-H exchange is coupled with anion exchanger (solute and Cl)
|
|
|
|
In the Colon how is Na absorbed?
|
through the help of an Na-H exchanger (Cl)
|
|
|
|
How is Na absorbed in the Rectum?
|
Na-H exchanger coupled to Cl and also through an Na channel
|
|
|
|
Summary
|
|
|
|
Where is Chloride absorbed at?
|
throughout the GI tract
passively -coupled Na and Cl absorption Duodenum- ileum - HCO3 dependent Cl absorption - distal ileum to rectum |
|
|
|
Potassium transport throughout the small intestine occurs how?
|
most is absorbed passively
** in the colon however active K secretion and absorption are observed |
|
|

|
Na is high on the luminal side right after a meal
- uses a low Na gradient in exchange for a proton *Amiloride (diuretic) sensitive luminal membrance transporter that mediates the exchange of one proton with on Na ion - Na, K-ATPase extrudes the Na out the other side |
|
|
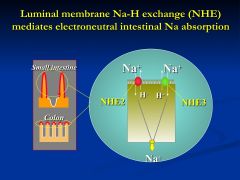
Why have two exchangers that do the same thing?
|
THe NHE3 is the fine tuning of exchange because it is more tightly regulated that NHE2
* these exchangers are NOT in the crypts |
|
|
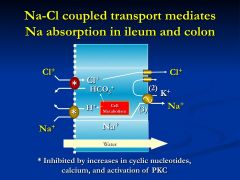
|
Na-H coupled to Cl (anion exchanger)
Cl is also absrobed - net effect - absorbing both Na and Cl - bicarbonate being extruded into the intestinal lumen |
|
|
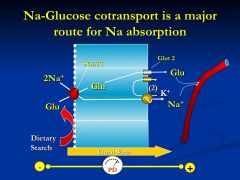
|
*SGLT1 is an Na-Glucose co-transporter
* Glucose extis the cell by Glut2, whereas Na is pumped out by the Na pump Effective way of getting Na and glucose into the blood stream - moving 2+ charges so the blood side is more positive than the lumen - NaCl absorbed in the presence of glucose ** In contrast to other Na absorptive pathways, glucose and aa stimulated Na absorption are NOT affected by enterotoxins such a cholera toxin or enterotoxin |
|
|
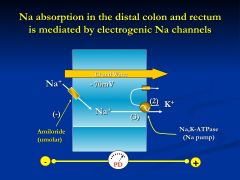
|
powerful pump
- pump out sodium...water will follow and then the luminal contents will solidify |
|
|
|
What is the rationale behind using sugar or aminoacid-based oral rehydration solutions for treatment of cholera or traveler's diarrhea?
|
Glucose and Na absorption though SGLT1 isn't affected by enterotoxins (cholera or ST-alpha)
* use sugar or aminoacid-based oral rehydration solutions **rice also very effective |
|
|

what is intestinal secretion driven by?
|
secretion of Cl or HCO3
- Cl enters the cell via basolateral membrane Na/K/2Cl - Cl enter lumen through CFTR - opening of these channels is done by protein kinases ** active Cl secretion in the gut is regulated by numerous neurotransmitters and gut peptides |
|
|
|
What are the two divisions of regulatory mediators of intestinal water and electrolyte transport?
|
2 groups:
1) secretagogues 2) proabsorptive agents |
|
|
|
How doe secretagogues agents work?
|
they stimulate net intestinal secretion by activating active anion secretion and inhibiting NaCl absorption
- Serotonin, CCK, Gastrin, Guanylin, arachiodnate metabolites (NO), stimulate enteric neurons, Blood (VIP, Calcitonin) |
|
|
|
How do Pro-absorptive agents work?
|
somatostatin, catecholamines, and adrenal steroid hormones
|
|
|
|
Can the intestinal mucosa change?
|
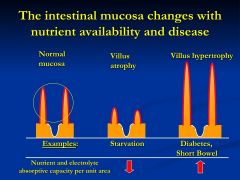
*starvation- there is nothing there to absorb so don't put energy into maintaining villi
* diabetes (short bowel)- elongation- body doesn't think that glucose is there so they hypertrophy |
|
|
|
Acute regulation of intestinal water and transport makes rapid changes by?
|
cellular regulations of transport protein activity and NOT by altered expression of their abundance
*when signals cease the transport rates return to normal |
|
|
|
Do pro-absorptiev agents inhibit the release of secretagogues?
|
Yes.
Ex: adrenergic agents stimulate pre-synaptic alpha-2 adrenergic receptors INHIBITING Ach release for enteric neurons |
|
|
|
By what mechanism do pro-secretory agents work?
|
increase cyclic nucleotide or cytosolic Ca
*Adenylate cyclase- VIP and prostaglandin E2 = increase cAMP *PIP2- Ach and serotonin = increases in Ca |
|
|
|
How does Guanylin work?
|
secreted into the intestinal lumen
binds surface receptors that have guanylate cyclase activity * increase in cGMP = net secretion - increased anion secretion and inhibiting NaCl absorption |
|
|
|
By what mechanism does enterotoxin of E.Coli?
|
binds to the guanylin receptor
* causing profuse diarrhea that is common for traveler's diarrhea |
|
|
|
What are three examples of chronic regulation so intestinal water and electrolyte transport?
|
"intestinal adaptation"
1) massive small bowel resection or disease 2) plasma volume depletion 3) SCFA |
|
|
|
What effect does massive small bowel resection or disease have?
|
increase in villus width and height are adaptive responses = increase in absorptive capacity
** increase in enterocyte Na absorptive capacity due to increased gene expression of Na-H exchangers |
|
|
|
What is the adaptation in plasma volume depletion?
|
intestine increases its ability to absorb fluid and electrolytes
**increased plasma levels of aldosterone - increases expression of luminal membrane Na-H exchangers (NHE3)- proximal colon * increase in Na channel and Na pump in distal colon and rectum * GAPDH is unchanged ***allows the increased extraction of water and electrolytes |
|
|
|
What is colonic CHO metabolized to?
|
CO2, SCFA,H2 (gas)
|
|
|
|
What are colonocytes?
|
are mature, non-dividing epithelial cells forming the colonic epithelium. These columnar epithelial cells belong to one of several epithelial cell lineages in the gastrointestinal tract, which differ from each other in morphology and function (
|
|
|
|
What are the effects of SCFA on intestinal mucosal function?
|
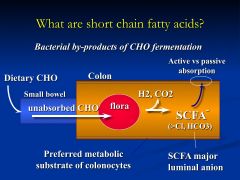
1) preferred metabolic fuel soruce for coloncytes
2) promote mucosal proliferation and differentiation 3) stimulate intestinal Na absorption 4) chronic upregulation of eletroneutral Na absorption in colonic mucosa |
|
|
|
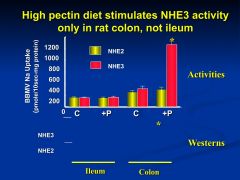
high fiber diet (pectin)
* marked increase in the protein expression of colonic NHE3 ** mediated by SCFA- stimulated increases in NHE3 gene expression *only occurs in the colon because that is where the bacteria are located |
|
|
|
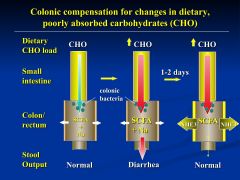
when you start changing dietary habits
- dump a larger load on bacteria and overwhelms them - but continue diet and then the bacteria will accomodate * possibly due to increase transporters like NHE3 to accommodate the uptake of more fluid |
|
|
|
what are the major pathophysicological mechanisms causing diarrhea?
|

|
|
|

|
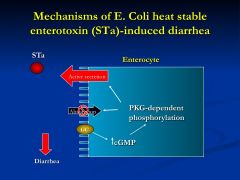
STalpha binds to guanylate cyclase - increase in cGMP = active secretion of anions and inhibiting uptake of Na and Cl
** no mucosal inflammation of destruction |
|
|
|
Do increased propulsive motor contractions cause diarrhea?
|
Not by themselves but usually associated with diarheally cases
* reduce the contact time between luminal fluid and the absorptive surface area (can cause nutrient malabsorption) |
|
|

|
Cholera binds GM1- ganglioside receptor of the enterocyte
* inserts its alpha sub-unti into the cell * irreversibly activating adenylate cyclase through Gs reg. subunit - increases in cAMP causes net secretion by inhibiting NaCl absorption and stimulates active anion secretion |
|
|
|
Is nutrient dependent Na absorption affected by cholera toxin
|
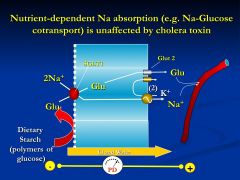
No
|
|
|
|
What are the solutions to helping cholera toxin?
|

low bicarbonate due to secretion
- can use oral replacement therapy so that numbers go back to normal *BUT you can't do anything to help the diarrhea you might actually make it worse - need cells to renew so wait 5 days |
|
|
|
What occurs in cystic fibrosis?
|
CF patients are unable to secrete chloride through CFTR
- leads to very viscous secretions * can lead to mucus obstruction in intestinal lining = meconium ileus * can also see blocking of pancreatic ducts * pulmonary infections are common |
|
|
|
What occurs during diabetic diarrhea?
|
uncommon
* diarrhea, anal incontinence, and autonomic neuropathy due to: diabetes- associated destruction of noradrenergic autonomic nerves to the gut *nerves have a pro-absorptive role Treatment: alpha-2- adrenergic receptor agonist, clonidine, Octreotide |
|
|
|
SUMMARY
|
|
|
|
What does salivary amylase break down?
|
starch
|
|
|
|
What does lipase break down?
|
fat
|
|
|
|
What does HCl acid and pepsin break down?
|
protein
|
|
|
|
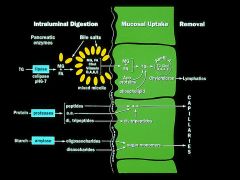
What are the three phases of the events in the small intestine?
|
1)intraluminal digestion
2)mucosal absorption 3) nutrient delivery |
|
|
|
What does digestion consist of?
|
characterized by pancreatic enzyme secretion and the release of bile salts that are critical for micellar solubilization of lipid
* Key events - release of CCK and secretin - stimulate bile salt and pancreatic secretion |
|
|
|
When is CCK released from the proximal small intestine?
|
in response to amino acids and fatty acids in the lumen
*CCK acts indirectly through the release of Ach which act on M3 receptors in the pancreas |
|
|
|
What does CCK do?
|
1) stimulates pancreatic enzyme secretion (amylase, lipase, and protease) from pancreatic acinar cells
2) causes contraction of the gallbladder 3) relaxation of Sphincter of Odi |
|
|
|
What does secretin do?
|
stimulates bicarbonate secretion from pancreatic duct cells
*neutralization of the acid |
|
|
|
Main source of calories in the diet?
|
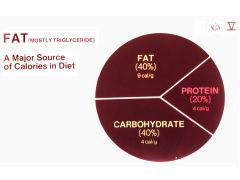
|
|
|
|
CCK stimulates secretion of which enzymes?
|
protease is the only one secreted in an inactive form
|
|
|
|
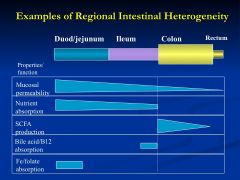
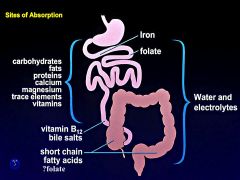
Nutrients are absorbed the entire length of the small intestine
*exception of folate and iron which are just absorbed in the proximal duodenum and jejunum *biles salts and cobalamin can only be absorbed in the distal ileum *Ca can be absorbed throughout at high concentrations |
|
|
|
What factors affect or influence the efficiency of nutrient uptake in the mucosa?
|
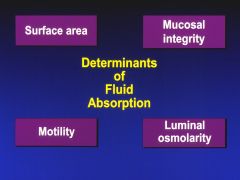
1) number of villus absorptive cells present
2) presence of brush border hyrolases 3) specific nutrient transport proteins on the brush border membrane 4) transit time (motility) - determines the amount of time of contact |
|
|
|
length of small intestine in child and adult
|
|
|
|
|
How is the surface area of the small intestine amplified?
|

-folded on itself
-off each fold there are villi - on each villi there are microvilli (brush border) |
|
|
|
Explain how absorption of water and electrolytes are coupled to nutrient absorption?
|
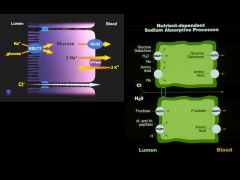
Sodium-glucose ligand transporter 1 (SGLT1)
- Na and glucose move into the cell - glucose then transported across the basolateral membrane into paracellular space * net result: water and electrolyte absorption |
|
|
|
How much energy do dietary carbohydrates provide?
|
|
|
|
|
How much energy do dietary carbohydrates provide
|

about half of an american's diet
|
|
|
|
Definition of Dietary fiber?
|
dietary fibers are mainly constituents of plant cell walls that resist digestion by enzymes of the human GI tract
|
|
|
|
How does digestion of starches occur?
|

Through alpha amylases (salivary amylase and pancreatic amylase)
|
|
|
|
What are the products of alpha-amylase digestion?
|
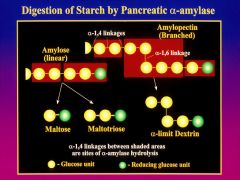
1) maltose
2) maltotriose 3) branched alpha-limit dextrin 4) oligosacchardies |
|
|
|
What must happen to oligosaccharides before digestion?
|
must be digested to sugar monomers before they are absorbed
*hydrolyzed by brush border enzymes *activity of oligosaccharides is greatest in the proximal small intestine |
|
|
|
digestion of alpha-limit dextrins (branched)?
|
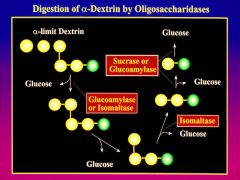
removal of glucose from non-reducing end by GLUCOAMYLASE
ISOMALTASE cleaves the alpha 1-6 linkage * digestion of maltotriose and maltose to glucose by SUCRASE OR GLUCOAMYLASE |
|
|
|
Absorption of glucose or galactose occurs how?
|
Through SGLT1 transporter
- along with 2 Na+ ions = secondary active transport |
|
|
|
Under high luminal glucose conditions which other mechanism is at work for absorption of monosacchardies?
|
Glut2 insertion in the apical membrane = regulated by Ca, intestinal taste receptors,insulin, glucagon-like peptide 2, and stress
* |
|
|
|
Under high luminal glucose conditions which other mechanism is at work for absorption of monosacchardies?
|
Glut2 insertion in the apical membrane = regulated by Ca, intestinal taste receptors,insulin, glucagon-like peptide 2, and stress
* |
|
|
|
Glut5 vs. Glut2
|
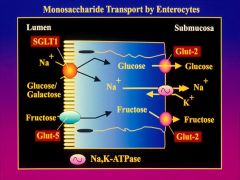
monosaccharide transporter for fructose by facilitated diffusion on lumen side
* Glut 2 does (glucose, galatose, and fructose) |
|
|
|
What are the sites of sugar absorption?
|
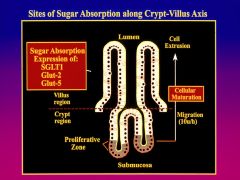
transporters are regulated along the longitudinal axis (highest in jejunum and lowest in ileum) and horizontal axis
|
|
|
|
Acquired lactase deficiency?
|
expression of lactase beings in late gestation
and then declines after weaning down to only 10% *decreased synthesis of the enzyme **acquired lactase deficiency develop abdominal gas/bloating, cramps, and diarrhea * unabsorbed lactose draws water into the lumen *lactose - SCFA- methane, CO2, and H2 |
|
|
|
Congenital glucose- galactose malabsorption?
|
Rare autosomal recessive disease (missence mutation in SGLT1)
* glucose isn't being taken up with 2 Na+ *increased concentrations in the lumen will increase amount of water that stays in the lumen *Fructose is well tolerated and can be feed to infants with this disorder |
|
|
|
|
Fiber that gets down to the colon where bacteria can digest it
*generate short chain fatty acids and gas *bacteria help salvage these nutrients |
|
|
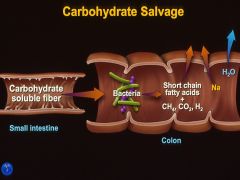
|
fiber that isn't soluble gets down to the colon
*bacteria can digest it * generate SCFA,CO2, H2 *bacteria salvage these nutrients |
|
|
|
Lactase is inefficient
if you aren't absorbing the SCFA that the bacteria make then the pH of your stool with decrease |
|
|
|
Sources of proteins?
|

1) the diet
2) endogenous proteins in digestive secretions 3) proteins present in exfoliated intestinal cells |
|
|
|
If gastric pepsins are absent, is protein digestion and absorption affected?
|
* No, gastric pepsin only accounts for 15% or dietary proteins
* pepsins are irreversibly denatured in mildly alkaline fluid, and thus little peptic digestion occurs past the duodenum |
|
|

|

|
|
|
|
How are pepsins made?
|
synthesized in gastric chief cells as inactive pro-enzymes
-signal sequence directs the protein to golgi - condensed into secretory granules - secretion of granules by exocytosis occurs through increases in cAMP or Ca - cleavage of 44 aa at N-terminus in acidic environment = active |
|
|

endopeptidase and exopeptidases?
|
endo- trypsin, chymotrypsin, elastase
exo- carboxypeptideases A and B - remove aa from carboxy-terminus |
|
|
|
After pancreatic proteases, how is further digestion of oligopeptides occur?
|
through brush border membrane peptidases
-20 different (1 of 4 groups_ *endopeptidases * aminopeptidases * carbozypeptidases * dipeptidase |
|
|
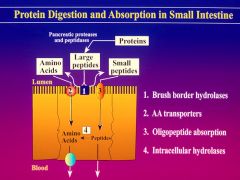
|
The brush border peptidases are mainly active against peptides of four or more aa to produce free amino acids as well as di and tri-peptides
** either small peptides of aa can be absorbed by enterocytes * single membrane transport system with broad specificity |
|
|
|
The jejunum, transport of ____ is the most significant
|
transport of di and tri-peptides is more significant than free aa
|
|
|
|
The ileum is more active in the uptake of ____ than the jejunum?
|
more active in the uptake of single amino acids
- free aa are transported across the brush border |
|
|
|
Intracellular peptidase and transport across the basolateral membrane occurs how?
|
enterocyte contains at least four intracellular peptidases that are involved in digestion of di and tri- peptides
* 90% or more of the products of protein digestion appear in the portal blood as free aa * 5 classes of transporters on basolateral side - 3 Na independent and 2 Na dependent |
|
|
|
How do acidic aa move across the basolateral side of the enterocyte?
|
no known basolateral transporters for acidic aa (glutamine, glutamate, aspartate)
** shunted into the pathways of energy metabolism |
|
|
|
What is Hartnup's disease?
|
rare autosomal recessive disorder that involves defective renal and intestinal transport of neutral aa
** malabsorbed tryptophan is metabolized by gut bacteria to various indoles and excreted in feces and urine * Tryptophan is a precursor for Niacin |
|
|
|
What is Cystinuria?
|
defect in the dibasic brush border aa transporter in the small intestine and renal proximal tubule
|
|
|
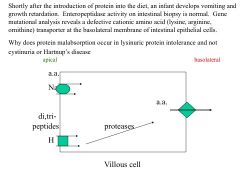
|
in lysinuric protein intolerance, a rare autosomal recessive defect in the transporter present in the basolateral membrane of intestinal villus and renal tubular epithelial cells
*unlike cystinuria or hartnup - babies with lysinuric intolerance have severe growth retardation and failure to thrive **in the prior two disease the defect can be bypassed * but in lysinuric, the transport across the basolateral membrane can not occur- and therefore are never absorbed |
|
|
|
What is the primary lipid consumed in the diet?
|
Long chain triglycerides and smaller amount of cholesterol, phospholipids, and fat-soluble vitamins.
|
|
|
|
Where does the digestion of lipids begin?
|
In the stomach with gastric lipase
* hydrolyzes the 3-position fatty acid |
|
|
|
What produces gastric lipase? Characteristics of gastric lipase?
|
chief cells in the fundus of the stomach
* its secretion is stimulated by cholinergic agents (gastrin and CCK) * active over a broad range of pH (2-6) *resistant to pepsin *inhibited by bile salts |
|
|
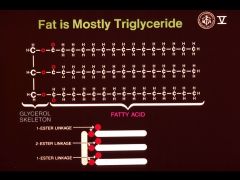
|
most dietary triglycerides are composed of long chain fatty acids (C14-22) saturated or unsaturated
* polyunsaturated are preferentially located in the 2 position of the glycerol backbone |
|
|
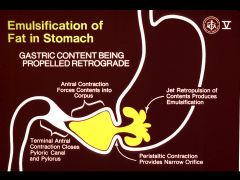
|
The churning action of gastric peristalsis against the closed pylorus results in the formation of oil emulsions containing triglycerides in its core
|
|
|
|
Gastric lipase is responsible for what percentage of total intraluminal lipid digestion?
|
20-30%
*fat emulsion is then emptied into the duodenum |
|
|
|
Fat in the duodenum strongly inhibits what action?
|
gastric emptying
*this is to ensure that fat is not emptied more rapidly than the duodenum can accommodate |
|
|

|
Summary
|
|
|
|
Properties of Gastric Lipases?
|

|
|
|
|
Gastric acid in duodenum stimulates secretion of what?
|
Secretin - peptide hormone from neuroendocrine cells
*secretin stimulates secretions of bicarbonate rich fluid by pancreatic ductular cells |
|
|
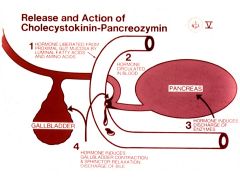
|
1) hormone secreted from gut when fatty acids and aa are present 2) hormone circulates in blood
3) CCK induces discharge of enzyme 4) induces gallbladder contraction and sphincter relaxation |
|
|
|
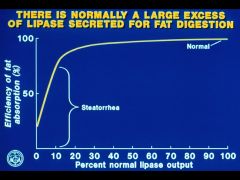
How much lipase is secreted for fat digestion?
|
|
|
|
|
What is the product after pancreatic lipase has worked on the triglyceride?
|
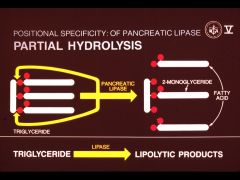
3 fatty acids
Now these products are soluble in micelles where triglycerides are not |
|
|
|
Tegmentum:
|
The intermediate level of the midbrain containing nuclei for cranial nerves and part of the reticular formation
|
|
|
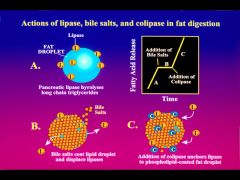
What is this showing?
|
Bile salts with lecithin - coat and stabilize the fat emulsion
*inhibits pancreatic lipase activity * therefore, pancreatic colipase is required to displace the bile salts **colipase also serves as an anchor the lipase then you can have cleavage of the triglyceride |
|
|
|
Properties of Pancreatic lipase
|

|
|
|
|
What are bile-salt activated lipases? What function do they have?
|
ex: phopholipase A2 and cholesterol esterase
*require bile salts for their activity * will catalyze hydrolysis of many ester bonds (unlike pacreatic lipases) **bile salts induce conformational changes in the enzyme that provides access to bulky substrates |
|
|
|
Why doesn't human milk which contains lipases, catalyze hydrolysis of lipids in the breast tissue
|
There is a bile-salt requirement for human milk
|
|
|
|
In adult, pancreatic lipase or bile-salt activated lipase has more of an effect?
|
pancreatic lipase 10-60x more lipolytic activity
*majority of triglyceride digestion * in neonates, pancreatic lipase is immature compensated with gastric lipase and milk bile salt activated lipase |
|
|
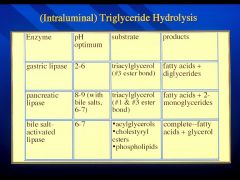
|
Summary
|
|
|

|
suggests pancreatic insufficiency
* fat malabsorption due to decreased lipase secretion = impaired lipolysis * unabsorbed triglycerdie emulsion causes large bulky stools (don't stimulate fluid secretion) * pancreatic insufficiency usually due to CF or hereditary pancreatitis 2) oral pancreatic enzymes are denatured at acid pH - lack bicarbonate secretion all of the enzymes are being destroyed by acidic environment *improve by blocking acid secretion H2 or PPI |
|
|
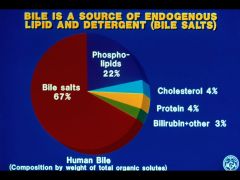
|
Major component of bile
*above critical concentration- bile salts form micelles |
|
|

|
mixed micelles are multi-molecular aggregates of varying size depending on the ratio between bile salts and lipolytic product
|
|
|
|
What is the rate limited step of lipid uptake?
|
the diffusion of mixed micelles across this unstirred water layer
*pH gradient exists across this layer- the fluid is in immediate contact with the brush border is about 1 pH more acidic *lower pH may enhance absorption (as protonated FA are more soluble than ionized forms) |
|
|
|
How are fatty acids and 2 monoglycerides taken into the enterocyte?
|
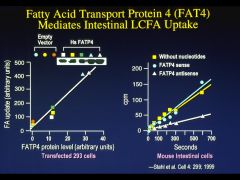
Through the microvillus membrane fatty acid transporter FATP4
the mechanism for 2 monoglycerides is probably by simple diffusion across brush border- high lipid solubility |
|
|

|
Summary
|
|
|
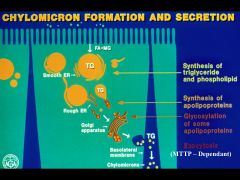
|
chylomicrons undergo further processing in the golgi
followed by fusion into secretory vesicles and exocytosis at the basolateral membrane of enterocyte expels into the extracellular space |
|
|
|
What are the two families of cytosolic fatty acid-binding proteins?
|
Fabpi- specific for long chain
Fabpl- broader specificity |
|
|
|
What do lipids need to be bound to intracellular Fabps?
|
prevents lipids form forming lipid droplets in the cytosol and directs them to the SER for triglyceride resynthesis
|
|
|
|
What happens when the free fatty acids reach SER?
|
the fatty acids and 2 monoglycerides are re-esterified to form triglycerides
*large lipid droplets are formed at the apex of the enterocyte following a fatty meal |
|
|
|
Lipid droplets are converted into chylomicrons by what?
|
Phospholipids and apolipoproteins A,B, and C
|
|
|
|
What are the two pathways for Triglyceride synthesis?
|
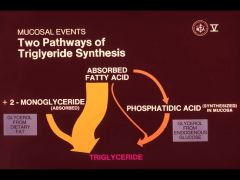
|
|
|
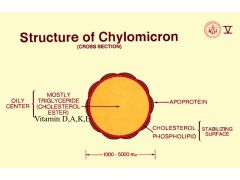
|
Chylomicrons are spherical and vary greatly in size
-90% of their mass is accounted for by triglycerides - too large to transverse the mucosal capillaries -leave the intestine in fenestrated lacteals and ultimately delivered to the venous circulation by the thoracic duct |
|
|
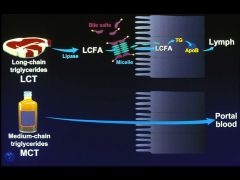
|
MCT (6-12C) - small amt of diet
*digestion and absorption of MCTs differ significantly MCTs are: 1) hydrolyzed more rapidly 2) more H2O soluble 3) don't need bile salts to be taken up by enterocyte 4) directly released *not resynthesized * no chylomicrons |
|
|
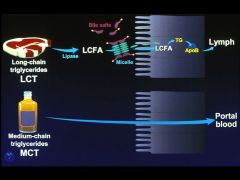
|
MCT (6-12C) - small amt of diet
*digestion and absorption of MCTs differ significantly MCTs are: 1) hydrolyzed more rapidly 2) more H2O soluble 3) don't need bile salts to be taken up by enterocyte 4) directly released *not resynthesized * no chylomicrons |
|
|
|
Pros and Cons of MCT?
|
Pro: useful in people with digestion and absorption issues
- nutritional supplement in patients with GI disorders Con: MCT do not contain essential polyunsatruated fatty acids, which are the precursors for prostaglandins, leukotrienes, etc. |
|
|
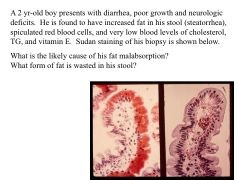
|
these finds suggest failure or transport of chylomicrons from the basolateral membrane of villus cells
* decreased lipids in the blood = cause lipid membrane abnormalities (RBC) **ABETALIPOPROTEINEMIA |
|
|
|
What is abetalipoproteinemia?
|
rare autosomal disease due to failure in secretion of chylomicrons in the intestine
*mutation in gene for microsomal triglyceride transfer protein (MTTP) - protein required fro the assembly and secretion of apo-B containing lip-proteins *triglycerides are lost in the stool as the enterocytes are sloughed as part of normal turnover |
|
|
|
Why is bile needed?
|
the physicochemical properties of bile salts make micellar solubilization of lipid possible in the aqueous environment of both bile and intestinal fluid
|
|
|
|
How much bile is secreted daily?
|
1L/day in adults
|
|
|
|
Composition of hepatic bile?
|
97.5% water / bile salts/ mucin / lipid
|
|
|
|
How are bile salts synthesized?
|
synthesized from cholesterol
|
|
|
|
What are the two bile salts directly formed from cholesterol?
|
The primary bile salts:
* sodium cholate * sodium chenodeoxycholate **synthesis is regulated by feedback inhibition |
|
|
|
What is the rate limiting step of bile salt synthesis?
|
at the site of microsomal enzyme 7alpha-hydroxylase
= rate limiting in the synthesis of bile salts from cholesterol |
|
|
|
Bile salts returning to the liver can also have an inhibiting effect...how?
|
act at the rate-limiting enzyme in the cholesterol synthesis, HMG-CoA reductase
|
|
|
|
How much bile salts are made daily?
|
.3-.6 / day
but this can increase 5-10x in the case of bile salt depletion |
|
|
l'espoir (m.)
|
hope
|
|
|
|
honteux (-euse)
|
shameful
|
|
|
|
What purpose does cholyl amidase serve?
|
bacterial enzyme i the terminal ileum that can cleave the extremely strong amide conjugation
|
|
|

|
Conjugation of bile salts lowers the pKa
* allowing bile salts to remain in solution at normal duodeanl-jejunal pH (6.5) - will not precipitate out |
|
|
|
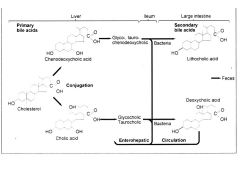
Enterohepatic circulation?
|
conserves bile salts within the intestine by the action of the enterohepatic circulation
|
|
|
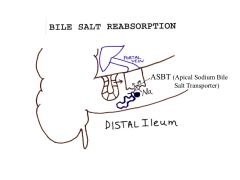
|
Normal conditions: bile salts are efficiently reabsorbed by the apical sodium-dependent bile transporter (ABST) located in the ileum
|
|
|
|
Why is there a little jejunal absorption of bile salts?
|
Due to the glycine conjugates being at a higher pKa
*this allows a little jejunal absorption by passive nonionic diffusion |
|
|
|
How much of the intraluminal concentration of bile salts is reabsrobed?
|
90% is re-absorbed
*but by recycling 5-10x /day the physiological pool of bile salts becomes much greater |
|
|
|
2 ways that bile salts are attacked in the intestinal lumen?
|
1) deconjugation (cholyl amidases)
2) dehydroxylation - to produce the following secondary bile salts |
|
|
|
What are the secondary bile salts?
|
1) sodium deoxycholate
* from cholate conjugates 2) sodium lithocholate *from chenodeoxycholate conjugates |
|
|
|
After a single enterohepatic circulation how many bile acids are no longer conjugated?
|
10-20% of the portal venous bile acids are no longer conjugated after a single circulation
|
|
|
|
Where are bile salts re-conjugated?
|
The bile salts are reconjugated after being taken up by the liver and then re-excreted into the bile
|
|
|
|
Do secondary bile salts (like primary) undergo enterohepatic circulation?
|
Yes!
* significant amounts of deoxycholate are reabsorbed * but only trace amounts of lithocholate appear in bile |
|
|
|
What is the normal distribution of bile salts?
|
40% cholate
40% chenodeoxycholate 20% deoxycholate 13% lithocholate |
|
|
|
What are the 4 things that normal bile salt kinetics depend on:
|
1) hepatic synthesis
2) secretion into bile 3) appropriate intestinal mileu 4) intact enterohepatic circulation |
|
|
|
Bile salts act as a detergent to do what function?
|
solubilize fats for their efficient absorption
|
|
|
|
Bile salt synthesis is tightly regulated... why?
|
Because bile salts are cytotoxic at high concentrations
*want to prevent liver damage |
|
|
|
What are nuclear hormone receptors?
|
transcription factors that are activated by ligands
*nuclear receptor FXR forms a heterodimer with RXR to increase transcription of bile salt metabolism |
|
|
|
FXR up-regulates expression of bile salt transporters on what membrane of the cell?
|
Both the basolateral and the canalicular hepatocyte membranes
|
|
|
|
When to micelles form?
|
When hydrophilic properties predominate
ex: bile salts |
|
|
|
When do bilaminar sheets of crystals form?
|
when hydrophobic and hydrophilic are about equal
|
|
|
|
When do hydrophobic properties form?
|
in a molecule that is insoluble
ex: cholesterol |
|
|
|
What happens when the critical micellar concentration is exceeded?
|
micelles form when the concentrations of bile salts exceed this threshold
|
|
|
|
Mixed micelles?
|
larger lipid-soluble core than a pure bile salt solution
*solubilization of lipid in the inner lipid core for him *formation of mixed micelles is crucial to solubilizing cholesterol in the bile and dietary fat |
|
|
|
What actions can biliary ductules preform?
|
capable of both reabsorption and secretion
|
|
|
|
What is the function of the gallbladder?
|
mucosa of the normal gallbladder is capable of concentrating bile salts, pigments, and lipid constituents
|
|
|
|
Sources of Intraluminal cholesterol?
|
1) diet
2) intestinal turnover 3) bile |
|
|
|
Explain intraluminal digestion of cholesterol?
|
hydrolysis of cholesterol esters by cholesterol esterase
* then solubilized into micelles *uptake across the brush border * reseterfication of enterocytes with fatty acids |
|
|
|
What are the two functions mentioned for phospholipids?
|
1) cell membranes
2) chylomicron assembly |
|
|
|
Intraluminal digestion of phospholipids?
|
hydrolysis by phopholipase A2 to yield a LCFA and lysolecithin
*lysolecithin- water soluble and rapidly absorbed by diffusion *LCFA are solubilized in micelles |
|
|
|
What are the fat-soluble vitamins?
|
E, D, A, K
|
|
|
|
Biochemical functions of fat-soluble vitamins?
|

|
|
|
|
What are the food sources for fat-soluble vitamins?
|

|
|
|
|
Describe the intraluminal digestion of Vitamin A?
|
retinyl esters are hydrolyed by pancreatic esterase to retinols and carotenoids
|
|
|

|
summary
|
|
|
|
Vitamin D is absorbed how?
|
-solubilized in micelles
-uptake across mucosa is by passive diffusion -packaged into chylomicrons -exocytosis into the liver and kidney |
|
|
|
Where does D3 interaction with Vitamin D?
|
1, 25 (OH)2D3 interaction with a vitamin D receptor
|
|
|
|
What is the intraluminal digestion of Vitamin E?
|
*de-esterfication of pancreatic esterase and solubilized inot micelles
* passie diffusion into enterocytes, re-esterfied, into chylomicrons, and delivered to circulation via lymphatics |
|
|
|
Absorption of Vitamin K?
|
phylloquinones from the diet are solubilized into micelles and taken up into enterocytes by passive diffusion
|
|
|
|
summary
|
|
|

|
summary
|
|
|
|
summary
|
|
|

|
summary
|
|
|
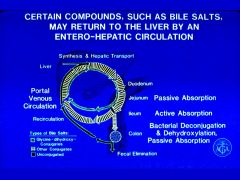
|
summary
|
|
|
|
What are the water-soluble vitamins?
|

|
|
|
|
Function of water soluble vitamins?
|
function as coenzymes
|
|
|
|
What are the minerals that are absorbed in the small intestine?
|
Calcium, phosphorus, magnesium, and sulfur
|
|
|
|
What are the trace elements that are necessary for biochemical processes?
|
Calcium
Phosphorus Magnesium *provide structural support (skeleton) |
|
|
|
Function of Vitamins and Minerals
|

|
|
|
|
What are the exceptions to absolute dietary vitamin requirements?
|

|
|
|
|
What is cobalamin?
|
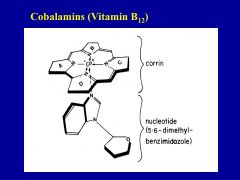
has two portions
nucleotide and porphyrin-like corrin ring- contains cobalt at center |
|
|
|
What are some of the ligands that are attached to the cobalt in the corrin ring?
|
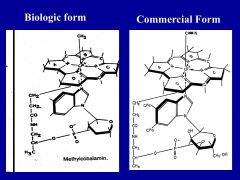
1) methyl- biological
2) 5-deoxyadenosyl group 3) cyanide group- commercial |
|
|
|
Is Cobalamin conserved?
|
Yes. Cbl is excreted into the bile and then reabsorbed
Daily losses: 1-3 ug *Due to large stores in the liver reabsorption in the intestine it could take 2-12 years for a Cbl deficiency to be detected |
|
|
|
Foods containing B12
|

|
|
|
|
Cobalamin-Binding Protein: Intrinsic factor
|
Source: Partietal cells
Required for: uptake of Cbl by ileum |
|
|
|
Cobalamin-Binding Protein: R binders
|
Source: Many cells, all secretions
Required for: Unknown |
|
|
|
Cobalamin-Binding Protein: Cubulin
|
Source: ileal cells, endocytic receptor
Required for: Uptake of Cbl-IF |
|
|
|
Cobalain binding Protein: IF Aminoless
|
Source: endocytic receptor
Required for: complex by ileum |
|
|
|
Cobalain binding Protein: Transcobalamin II
|
Source: liver, intestinal mucosa, macrophages
Required for: Transport of Cbl out of gut and into cells |
|
|
|
Once cobalamins are liberated from bonds in proteins, the Cbl is bound to one of two different proteins...what are they? How do they differ?
|

at lower pH (stomach) more Cbl is attached to R-proteins
but once the Cbl enter the small intestine- R proteins are degraded by proteases freeing Cbl to bind to IF |
|
|
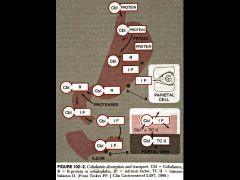
|
Summary
|
|
|
|
once the IF- Cbl complex has formed where does it travel?
what occurs? |
To the distal ileum
- where it is exposed to specific endocytic receptors (cubulin and aminonless) which are expressed at the apical surface of ileal cells *absorb IF-Cbl |
|
|
|
What is Imerslund-Grasbeck Syndrome?
|
Syndrome seen in Finnish population that supports the role of cubilin in vitamin B12 absorption
|
|
|
|
What happens to Cbl-IF once internalized?
|
IF is degraded
*then Cbl is complexed with transcobalamin II in the portal circulation |
|
|
|
What does TCII do?
|
Transports cobalamins to tissues such as bone marrow and the nervous system
|
|
|
|
What is cobalamin essential for?
|
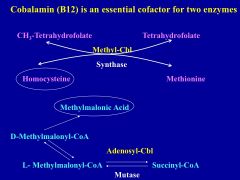
- synthesis of succinyl CoA
- methionine (aa, purine, pyrimidine) - conversion of homocystenine to methionine ** deficiencies can disrupt DNA synthesis ***leading to megoblastic anemia and neuropsychiatric disturbances |
|
|
|
Common causes of Cobalamin deficiency?
|
1) chronic atrophic gastritis
2) ileal disease (Crohn's, tropical sprue, ileal resection 3) bacterial overgrowth *lack or abnormal IF * decrease surface receptors for Cbl-IF complex * lack or abnormal TCII |
|
|
|
What purpose does folate serve in the body?
|
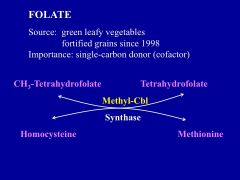
* bacteria in the colon can also produce folate
* acts to transfer 1 carbon unit for a number of enzymatic reactions involved in DNA, RNA, and protein synthesis |
|
|
|
Symptoms of folate deficiency?
|
megaloblastic anemia and neural tube defects in pregnancy
|
|
|
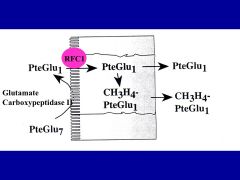
|
*folate taken in by diet is predominately in the polyglutamate form
*glutamate carrboxypeptidase II cleaves the molecule at the brush border to produce monoglutamate *uptake by folate carrier (RFC1) *in enterocyte, it is methylated and reduced *transported across basolateral membrane to the portal circulation and liver |
|
|
|
What are the two forms that iron is present as?
|
The iron heme- 10%
nonheme (ionic form) -90% * intestinal absorption of heme iron is more efficient |
|
|
|
What is the mechanism for absorption of heme iron?
|
* at duodenal pH, heme is split from globin
* absorbed as metalloporphyrin into enterocytes * once in cell iron is released from heme by heme oxygenase *Iron is transported bound to transferrin in the plasma *delivered to tissues for use or storage |
|
|
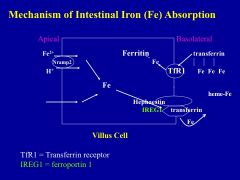
|
* Nonheme iron mostly like Fe3+
* complexed with aa, sugars, asorbic acid to keep Fe in solution at duodenal pH * Fe3+ converted to Fe2+ @ brush border (ferrireductase) * Nramp2 allows absorption into enterocyte * transported across cell to basolateral side where it crosses through IREG1 - ferroxidase hephaestin required for export * bound to transferrin in serum ** TfR1 is the receptor that allows Fe uptake into cells |
|
|
|
Regulation of Iron absorption is tightly regulated... how?
|
*decrease Fe in diet the Nramp2 receptor is unregulated
* increase Fe in diet the Nramp2 receptors is downregulated ** iron absorption is overall regulated by the liver |
|
|
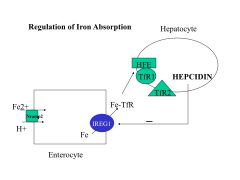
Role of Hepcidin on IREG1
|
*Hepcidin - secreted by hepatocytes, inhibits iron absorption in intestine
* directly interacts with IREG1 at membrane *interaction results in the internalization and degradation of IREG1 = reducing iron export |
|
|
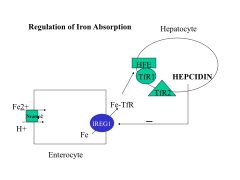
Three molecules that regulate hepatic hepcidin?
|
HFE
transferrin receptor 2 hemojuvelin *HFE/TfR1 and TfR2 - sense changes in blood transferrin concentrations |
** low blood concentrations of diferric transferrin result in decreased hepcidin release from liver and increased Fe uptake
|
|
|
Where is calcium absorbed?
|
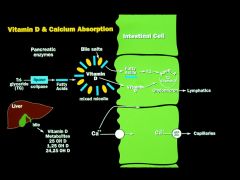
throughout small intestine at high levels - passively paracellular route
low levels - active saturable energy driven transcellular route (need Vit D3) *in duodenum |
|
|
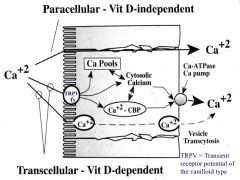
Transcellular calcium transport involves 3 steps:
|
1) entry into the cell via calcium channel (TRPV6 - expression Vit D dependent) - present on brush border
2)Translocation through enterocyte bound to calbindin D9K 3) extrusion at the basolateral membrane against an electrochemical gradient via a calcium ATPase and Na/Ca exchanger *ATPase stimulated by Vit D3 |
|
|
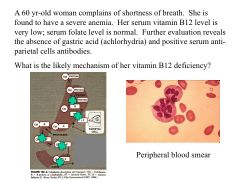
|
Autoimmune inflammation of the stomach is a rather common cause of vit. B12 deficiency in older women
*inflammation results in parietal cell damage and loss of acid production - loss of IF *can't cleave B12 from food due to decreased acid *later the loss of IF - further failure to absorb |
|
|

|
Review
|
|
|

|
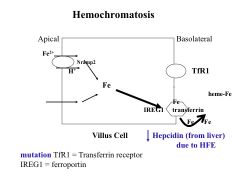
high % saturation in the blood due to primary hemachromatosis
- iron overload due to increased absorption of iron Treatment: phlebotomy and iron-chelation therapy |
|
|
|
round ligament contained in the falciform ligament
*right lobe (caudate and quadrate as well) * surrounded by fibrous Glisson's capsule which is richly innervated |
|
|
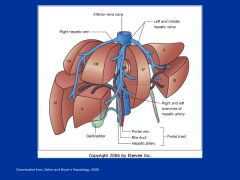
|
Segmentation of the liver into 8 parts
*each with its distinct blood supply |
|
|
|
What is the vascular anatomy
|
*Oxygenated blood is supplied by the right and left branches of the hepatic artery (from celiac)
*Oxygen poor blood is supplied by the right and left branches of the portal vein draining to celiac, SMA, IMA **bile ducts supplied solely by hepatic artery |
|
|
|
Describe the vascular anatomy
|
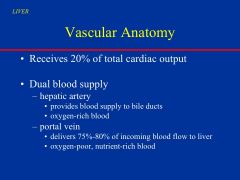
portal vein drains the GI tract of blood
*vascular drainage through hepatic veins to the IVC **dual blood supply- very resistant to anoxia |
|
|
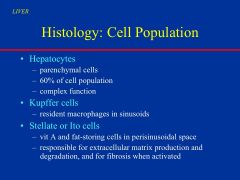
What are the cells types that compose the liver?
|

*kupffer cells-comprise the liver's immune defense - clears dead cells and debris
|
|
|
|
what is a liver lobule
|
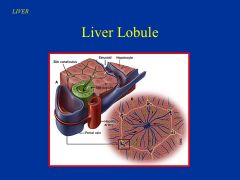
histologic and functional unit of the liver
-hexagonal with portal triads (artery, vein, bile duct) - continuous system of communicating hepatocytes - sinusoids - fenestrated capillary lining and no bm - separate these columns of parenchymal cells |
|
|
|
What is the space of Disse?
|
the space between the hepatocytes and the sinusoidal endothelial cells
|
|
|

|
portal triad is at center with the terminal hepatic venules at the periphery
* Zone 1 - cells closest- first to receive oxygenated blood and nutrient rich blood = last to develop hypoxia - first to regenerate Zone 3 - last to receive blood, most sensitive to toxic or vasculature injury |
|
|
|
What are the main activities of Zone 1 cells?
|
glucose synthesis and release
oxidative energy metaboism aa utilization bile acid and biliruben excretion ammonia detoxification |
|
|
|
What are the main activities of Zone 3 cells?
|
hepatocyte are most active in glucose uptake and utilization and biotransformation
|
|
|
|
Trace blood flow through liver starting at hepatic artery or portal vein?
|
-hepatic artery and portal vein in zone 1 along sinusoids
-drains into terminal hepatic venules - terminal vein - terminal vein unites with hepatic vein -IVC |
|
|
|
Trace the flow of bile?
|
-bile canaliculi (smallest branches in the biliary tree) from zone 3 to zone 1 of the acinus
- enters small terminal bile ductules - empty into small bile duct located in portal triad |
|
|
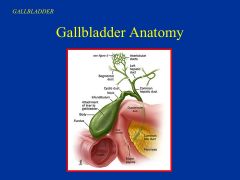
|
under right lobe of liver
30-50 ml of bile -blood supply for cystic artery -common hepatic duct to common bile duct |
|
|
|
Describe normal hepatic blood flow?
|
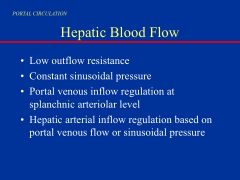
- liver has no active role in regulating portal inflow = passive recipient of blood
- wide range of portal blood flow without effect on pressure * decrease in portal venous flow or sinusoidal pressure = reflex in the increase of hepatic arterial flow |
|
|
|
Why is radius the most important determinate of resistance?
|
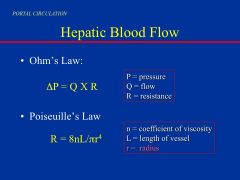
|
|
|
|
What is normal portal venous wedge pressure and how do you obtain this value?
|
5-8 mmHg
*measure wedge hepatic venous pressure -- internal jugular or femoral vein |
|
|
|
What is the hepatic venous pressure gradient?
|
when comparing the pressure difference between the wedge hepatic venous pressure and the free hepatic venous pressure (IVC)
**should only be a 1-4 mm Hg difference when normal ** not normal when ascites (large amounts of intraabdominal fluid) is present |
|
|
|
What are the pressure measurements for PVP and HVPG?
|

|
|
|
|
Define Portal Hypertension?
|
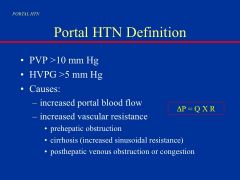
|
|
|
|
In cirrhosis, how does the relationship between flow and pressure change?
|
small increases in portal venous inflow may be associated with significant increases in portal vein pressure
|
|
|
|
Common causes for portal hepatic disease and their classifications
|

intrahepatic has further classifications that can be based on the wedge hepatic venous pressure
|
|
|
|
Prehepatic Causes
|
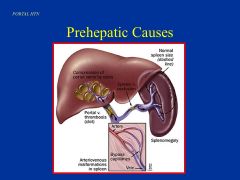
|
|
|
|
Intrahepatic cause
|
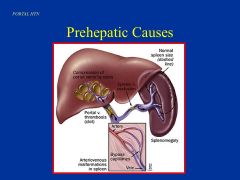
|
|
|
|
What are the characteristics of Cirrhosis?
|

|
|
|
|
Posthepatic Causes
|
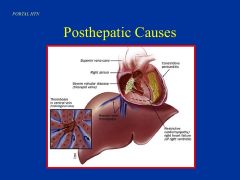
|
|
|
|
What is portosystemic collaterals?
|
collateral blood vessel from between the portal system and the systemic vasculature du to an increase in portal pressure
*seen in: esophagus, stomach, abdomen,periumblical area, and rectum |
|
|
|
Example of Caput medusa (periumblicial collateral) formation
|
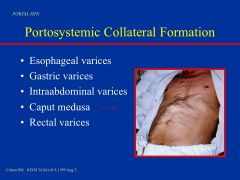
|
|
|
|
Is portal HTN a common complication of cirrhosis?
|
YES! Cirrhosis - process of diffuse fibrosis following hepatocyte destruction and nodular regeneration - leading to distorted liver architecture
|
|
|
|
What are some cases where cirrhosis might result?
|
chronic viral hepatitis
alcohol inherited metabolic disease biliary disease autoimmune disorders drugs and toxins |
|
|
|
The two major path-physiologic consequences of cirrhosis?
|
1) Portal HTN
2) hepatic failure |
|
|
|
What is splenomegaly and hypersplenism?
|

spelnomegaly- increased splenic vein pressure
hypersplenism- sequesteration |
|
|
|
When do varcies form?
when do you have a bleeding risk? |
Varices from when portal pressure is at least 10 mm Hg
Bleeding is likely when the portal pressure is at least 12 mm Hg |
|
|
|
What is bleeding for a variex governed by?
|
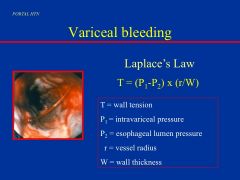
|
|
|
|
How are bleeding esophageal varices treated?
|
1) sclerotherapy
2) banding |
|
|
|
How can an acute reduction in portal pressures be achieved?
|
1) vasopressin
2) octerotide *via splanchnic arteriolar constriction |
|
|
|
How can chronic reduction in portal pressure be achieved?
|
Beta- adrenergic bockers
* another approch is to divert blood flow away from the portal system into systemic venous system by the surgical creation of shunts = transjugular intrahepatic portosystemic shunting (TIPS) |
|
|
|
What is hepatic encephalopathy
|

entry of ammonia and other toxins normally cleared by the liver but are reaching the systemic circulation instead due to build up of collateral arteries - this is how it reaches the brain
*causes astrocyte swelling and neurologic dysfunction |
|
|
|
Toxins implicated in hepatic encephalopathy
|
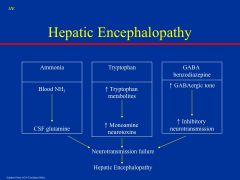
|
|
|
|
What are the stages of hepatic encephalopathy?
|
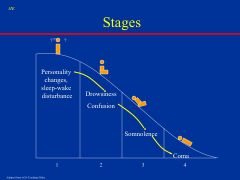
|
|
|
|
What is ascites?
|
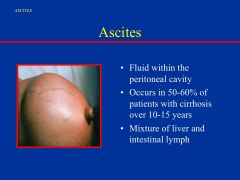
- increased in splanchnic vessels due to portal HTN results in increased fluid formation
|
|
|
|
What is hypoalbuminemia?
|
Also seen in with cirrhosis, results in decreased plasma oncotic pressure which favors an extravascular shift of fluid = Acties formation
|
|
|
|
Pathophysiology of cirrhosis?
|

|
|
|
|
Pathophysiology of cirrhosis: the two main pahways- increased intrahepatic resistance and decrease arteriolar resistance (vasodilation)
|
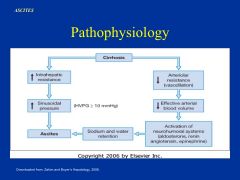
increased production of NO - vasodilation
*leads to decreased effective plasma volume *activation of compensatory mechanisms ** increased in renin-angio and sympathetic nervous system leads to increased renal resistance and decreased glomerular filtration rate * kidney compensates by retaining Na and H20 = ascites |
|
|
|
What is hematemesis?
|
vomiting of blood
|
|
|
|
How is portal hypertension managed?
|
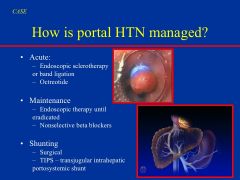
|
|
|
|
Where does plasma protein synthesis occur?
Which proteins are made? |
most proteins are made in the liver
*albumin *glycoproteins |
|
|
|
Where are the plasma proteins synthesized? What are the main plasma proteins?
|

Liver
*albumin *glycoproteins -- liver also produces most of the coagulation factors |
|
|
|
What is albumin's half- life?
|
~3 weeks
|
|
|
|
What is typically produced from the breakdown of aa?
|

Ammonia is also produced by bacterial degradation of urea (producing CO2 and NH3 - diffuse into portal circulation)
|
|
|
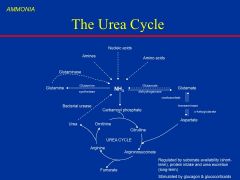
What is the urea cycle regulated by?
|
regulated by intracellular substrate levels on a short term basis and the amount of protein intake and rate of urea excretion on a long term basis
|
|
|
|
What does hyperammonia causes what?
And what can lead to hyperammonia? |
Causes: neurological dysfunction
*inherited defects in the urea-cycle enzymes, hepatic dysfunction, and shunting of portal blood to the systemic circulation can lead to hyperammonemia |
|
|
|
Monosaccharide Metabolism
|
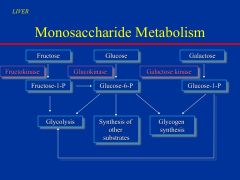
|
|
|
|
Metabolic pathways for hepatic utilization of glucose, fructose, and galactose
|
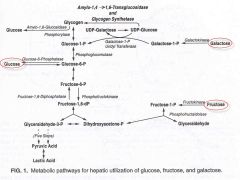
|
|
|
|
Under aerobic conditions, the liver uses what as its main source of energy?
|
Fatty acids (instead of carbohydrates)
* the rate of glycolysis will be low |
|
|
|
What is glycogen synthesis
|

|
|
|
|
What is glycogenolysis
|

|
|
|
|
What is gluconeogenesis?
|

- recycling alanine from muscles
- Cori cycle ( lack of glucose) |
|
|
|
Describe the intergration of the glucose- alanine cycle and the cori cycle?
|
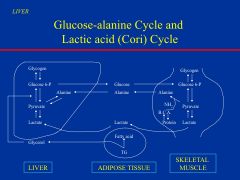
|
|
|
|
What is lipogenesis?
|
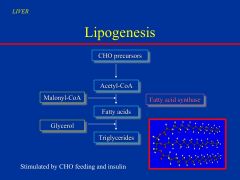
synthesis of fatty acids from carbohydrate precursor in the liver by their conversion to acetyl coenzyme a by the cytosolic fatty acid synthase complex
|
|
|
|
Characteristics of lipoproteins
|
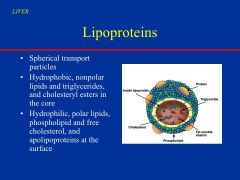
|
|
|
|
Triglycerides synthesized by the liver are secreted by...
|

* portion of VLDL is converted to LDL
*VLDL and LDL are either stored as adipose tissue or used for energy production by muscle |
|
|
|
What is significant of good levels of HDL?
|
HDL cleans up periphery by bring triglycerides to the liver = decrease risk of heart disease
* increase LDL and VLDL not a very good thing because you are producing triglycerides and placing them on the vessels |
|
|
|
When does lipolysis occur?
|
during fasting or starvation, or in diabetes mellitus with insulin deficiency when carbs are in short supply
*then lipolysis of triglycerdies stored in the adipose tissue occurs |
|
|
|
What is ketogenesis? and what purpose do ketones serve?
|
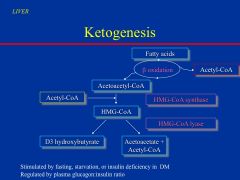
- ketones can be used as alternative energy source by muscle and brain
|
|
|
|
The liver is also a major site for cholesterol synthesis from...
|

*cholesterol important for:
- cell membrane - steroids - precursor for bile acid synthesis |
|
|
|
What are events that up-regulate cholesterol synthesis?
|
*hepatocyte depletion of cholesterol, depletion of the bile acid pool
*hormones: thyroid hormone, cortiocosteroids, adn glucagon |
|
|
|
what are the things that down-regulate cholesterol synthesis?
|
events that increase cellular cholesterol
- uptake of dietary cholesterol |
|
|
|
What is the target enzyme for many cholesterol lowering drugs?
|
HMG-CoA Reductase
|
|
|
|
What are the three metabolic conditions in humans?
|

|
|
|
|
What occurs in the fed state?
|
dietary glucose is absorbed from the intestines and is readily available as an energy source to all tissues (especially brain and RBC)
muscle: stored as glycogen liver: stored as glycogen *dietary fat absrobed in the intestine is transported as triglycerides (incorporate in chylomicrons) to liver and peripheral tissues |
|
|
|
What occurs in the post -absorptive state?
|

*liver maintains blood glucose level by glucogenolysis
* aa released by muscle are used for gluconeogenesis * adipose tissue starts releasing fatty acids which are in turn oxidized by the live and muscle to produce energy |
|
|
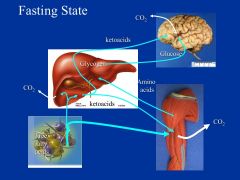
Fasting state- what occurs?
|
liver continues to supply lucose to RBC and brain
*hepatic stores of glycogen become depleted within 48 hrs * gluconeogenesis from aa * as fasting continues, fatty acids released from adipose tissue are oxidized to ketones |
|
|
|
What is the liver's biotransformation?
|
The liver is responsible for the biotransformation and clearance of endogenous and exogenous toxic substances
* 2 different groups: phase I and phase II |
|
|
|
What are the two different groups for biotransformation?
|

* phase I - make substances more polar and water-solublefor bile or urine excretion
- can lead to detox or more toxic intermediates * phase II- conjugation of molecule to a drug - enhances solubility and detoxifies compound -- exception is methylation which can detox but compounds become less soluble |
|
|
|
What all can biotransformation led to?
|

*Hepatocellular Injury occurs when the biotransformation leads to the formation of more toxic or carcinogenic metabolite
|
|
|
|
Explain Acetamionphen's Biotransformation?
|

conjugated with sulfate and glucoronic acid - each render it harmless
* BUT 5-15% is converted to reactive intermediate, NAPQI * NAPQI will react with sulfhydryl groups in proteins and cause damage |
*with normal doses most of the NAPQI can add glutathione making Mercapturic acid and be eliminated by the kidneys
*BUT if you overdose this pathway becomes saturated and glutathione stores become depleted *NAPQI accumulates leading to cell injury, live failure, death |
|
|
What is the function, source, absorption, and transport of Vitamin A?
|

|
|
|
|
What is the function, source, absorption, and transport for Vitamin A?
|

* retinol is a fat soluble vitamin
* once in enterocyte taken up by hepatocyte where it is stored as retinyl palmitate in Ito cel * Vit A overdose can lead to toxicity and damage of cells |
|
|
|
What is the storage, mobilization, and circulation of Vitamin A?
|

|
|
|
|
What is the function, sources, and absorption of Vitamin D?
|

* produced in skin as cholecalciferol in the presence of UV
* ergocalciferol can be absorbed from plants in the presence of bile salts * majority incorporated into chylomicrons and secreted into intestinal lymphatics * other portion is directly absorbed into portal circulation |
|
|
|
Describe the synthesis of Vitamin D?
|

* liver does not significantly store vitamin D
* most of the regulation for vitamin D occurs primarily at the level of the kidney |
|
|
|
What is the function, sources, absorption, and transport of Vitamin K?
|

*produced in intestinal bacteria
* once in chylomicron it is attached to a VLDL and secreted for distribution throughout the body ** the liver synthesizes the vitamin K dependent clotting factors |
|
|
|
Describe copper metabolism?
|
* 10% of copper ingested is through the duodenal enterocyte
* bound to albumin and carried to portal circulation * taken up by copper transporter CTR1 *ATP7B - regulates excreted bile * transform copper into ceruloplasmin |
*copper is an important coenzyme and component of proteins
* there are copper dependent catalytic enzymes yet excess copper can be toxic |
|
|
Wilson's Disease?
|
* the main excretory pathway of copper is through bile
* mutation of ATP7B transporter leads to a reduction in conversion of apoceruloplamin to ceruplasmin, which carries 90% of circulating copper to peripheral tissues * failure to excrete copper into bile will lead to accumulation of copper in hepatocytes = liver injury and cirrhosis |
|
|
|
How can a reduction in copper lead to anemia?
|
Copper helps maintain the integrity of blood cells
so if you decrease copper you will carry less oxygen |
|
|
|
Where is the major iron repository?
|
The Liver
|
|
|
|
What regulates the efflux of iron from macrophages and absorption of iron into the intestinal mucosa?
|
Hepcidin
|
|
|
|
What level is iron absorption mainly regulated at?
|
duodenal enterocyte
- enterocyte produces TFR-1 and HFE protein in cytoplasm - HFE and beta-microglobulin bind with TFR-1 on basolateral surface - sensor for transferrin iron in blood and allows entry into enterocyte - move to the apical surface together where iron absorption acutally occurs ** iron within the crypt enterocyte determines expression of DMT-1 and ferroportin proteins which are responsible for iron absorption on the apical surface |
|
|
|
*Ferric form - ferrous form (duodenal cytochrome B)
* absorption into enterocyte (DMT-1) *iron exported to portal circulation by ferroportin-1 * then converted back to ferric form so it can bind to apotransferrin (by hephaestin) * Diferric transferrin circulates in portal circulation - reaches hepatocyte * enters via TFR-1 or TFR-2 * amount of iron in the cell regulates expression of hecidin |
|
|
|
What is hepcidin? What does it do?
|
a peptide messenger that conveys the need for iron absorption to the enterocytes
** a high hepcidin level results when there is increased amounts of intracellular iron to effect a reduction in iron absorption |
|
|

|

|
|
|

|

|
The Manifestations of the patient's liver dysfunction?
* hypoglycemia *hypoalbuminemia * coagulopahty - prolonged prothrombin time * hepatic encephalopathy |
|
|
What is bilirubin?
|
It is the degradation product of heme
*85% is derived from senescent RBC *15% derived from free heme |
|
|
|
How is bilirubin transported to the liver?
|
bound to serum albumin
* undergoes conjugation in the hepatocytes and excretion into the bile via the bile canaliculus |
|
|
|
How much bilirubin is produced daily?
|

|
|
|
|
describe the metabolism of bilirubin?
|
*heme - biliverdin (heme oxygenase in spleen)
*biliverdin - bilirubin (biliverdin reductase) *bilirubin enter circulation bound to albumin (makes it more soluble) *facilitated diffusion into liver *bound to ligandin and Z protein and then undergoes conjugation * conjugated bilirubin into bile canalicular membrane by MRP2 (active) *becomes a component of bile |
|
|
|
What are the two different forms of bilirubin?
|

* unconjugated can inhibit synthesis of RNA and proteins and metabolism of carbohydrate
|
|
|
|
What is Kernicterus?
|
When babies are switching for fetal hemoglobin to adult hemoglobin they have to deal with unconjugated bilirubin - causing jaundice and brain damage
|
|
|
|
What is the chemical difference between conjugated and unconjugated bilirubin?
|
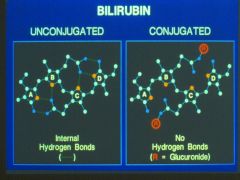
|
|
|
|
Once conjugated bilirubin reaches the intestines what is it converted to?
|

*urobilinogens by bacteria
* then eliminated in stool and by kidneys ** small fraction may be reabsorbed and re-excreted in bile |
|
|
|
Why is urine yellow?
|
Due to urobilin which is the oxidation product of urobiliogens
|
|
|
|
What would be your first thought it you couldn't detect urobilinogen in the urine or stool?
|
complete bile duct obstruction
|
|
|
|
When is urobilinogen production increased?
|
During liver disease or in over production of bilirubin
|
|
|
|
What is the pathway for bilirubin in a fetus?
|
Conjugated bilirubin excreted by the liver into the intestinal lumen is deconjugated by an intestinal Beta-glucoronidase. reabsorbed, and ultimately cleared by the placenta
|
|
|
|
What is breast milk jaundice?
|

* develop unconjugated hyperbilirubinemia due to breast feeding
- - beta-glucorinidase activity still present |
|
|
|
What can cause unconjugated Hyperbilirubinemia?
|

|
|
|
|
What is Gilbert's syndrome?
|

This is type II- UDP-glucoronosyl transferase
- partial transferase activity |
|
|
|
What is type I Crigler-Najjar Syndrome?
|
Type I UDP-glucoronosyl transferase
* resulting in severe hyperbilirubinemia * death |
|
|
|
What are the defects in conjugated hyperbilirubinemia?
|

* Dubin- Johnson is intermittent mild jaundice - MRP2 can't transport bilirubin across canalicular
*accumulation of dark pigment in the hepatic lysosomes |
|
|
|
What occurs in cholestasis and
hepatitis? |
regurgitation of bilirubin into the circulation due to failure of excretion from the liver
|
|
|
|
Therapy for Unconjugated Hyperbilirubinemia?
|

|
|
|
|
How does phototherapy work?
|
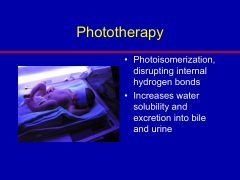
|
|
|
|
What is the function of bile?
|

|
|
|
|
What is the composition of bile?
|

|
|
|
|
What allows bile acids to act like detergents?
|
they have a hydrophilic and hydrophobic component
*facilitate intestinal absorption |
|
|
|
How are the primary bile acids made?
|

|
|
|
|
What are the canalicular transporter proteins involved in bile formation and transport
|
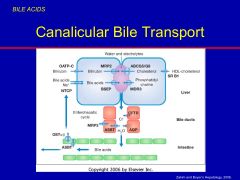
* NTCP (Na dependent)- uptakes bile from the blood into hepatocyte (basolateral side)
* OATP (Na independent) * bile salt transporters: BSEP, ABCG5/G8, MDR3, and MRP2 ** once in hepatocyte have to move against large concentration gradient (active- BSEP-bile salts) * MRP2 - bilirubin |
|
|
|
What occurs after canalicular secretion?
|

* bile undergoes alkalinization and dilution
* the ductular cells absorb and secrete fluid and electrolytes * bile ductular secretion and flow is stimulated by secretin |
|
|
|
What stimulates ductular flow?
|

|
|
|
|
Describe concentration and storage of bile?
|

*gallbladder concentrates bile 5-10 fold
* after a meal, CCK promotes gallbladder contraction |
|
|
|
Special requirement ACMS cards (SR) are for what?
|

ALSE, Avionics Support, Ground Support & Public Audit
|
PG 85-00-10 pg 5-1
|
|
|
Route for Bile
|
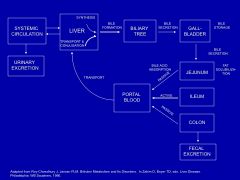
* this is the only route were cholesterol is eliminated from the body
*liver compensates for this loss by increasing cholesterol synthesis |
|
|
|
Primary bile salts are converted by intestinal bacteria to secondary bile acids
|
*sulfolithocholic acid - is poorly absorbed and rapidly lost from the enterohepatic circulation
|
|
|
|
How do the primary bile salts become secondary bile salts?
|
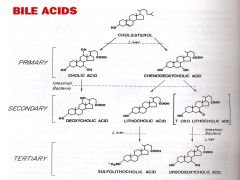
* Sulfolithocholic acid is poorly absrobed and rapidly lost from the enterohepatic circulation
|
|
|
|
What are the disorders of bile acid metabolism?
|

|
|
|
|
大どおり
|

おおどおり
|
boulevard (Oh Oh Dori)
|
|
|
How does cholestasis manifest?
|

|
|
|
|
What is choledochal cyst?
|
Manifests due to a back up of bilirubin into the blood
- patient gets jaundice - accumulation of bile acids in the skin causes histamine release and pruritus - lipoprotein metabolism is deranged and hyperlipidemia occurs which may result in cutaneous xanthomata - fat soluble vitamin malabsorption |
|
|
|
What happens when one has ileal inflammatory disease or ileal resection
|

* bile acids aren't reabsorbed like they should be
* so when then they enter the colon and cause a secretory diarrhea * increased bile acid loss so hepatic synthesis is increased to compensate * if can't fully compensate then you might have malabsorption of bat soluble vitamins |
|
|
|
True or False:
Na+ ions are 'LESS' favorable with cell permeability in the 'CELL MEMBRANE' of a 'NEURON' than K+ ions? (Na+ IONS MOVE 'INWARD' AND SLOWER THAN K+ IONS MOVE 'OUTWARD) |
True
|
|
|

|

|
|
|

|

|
|
|
|
How was the diagnosis of primary sclerosing cholangitis made?
|
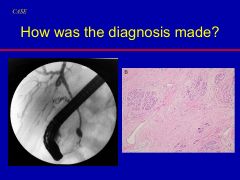
|
|
|
|
What are the other complications of the primary sclerosing cholangitis?
|

|
|
|
|
What is Charot's triad?
|
fever
upper quadrant abdominal pain jaundice |
|

