![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
45 Cards in this Set
- Front
- Back
|
definition. the maximum velocity of reaction that a particular amount of enzymes could possibly give
|
Vmax
|
|
|
is vmax extensive or intensive?
|
EXtensive. The more the enzyme the more higher the vmax
|
|
|
is Km extensive or intensive
|
INtensive. enzyme concentration does not change Km
|
|
|
what is inversely related to the binding affinity of the enzyme for substrate
|
Km
|
|
|
what does a large Km tell you
|
rxn only works when substrate conc is very high
|
|
|
what does a low Km tell you
|
rxn will function toward vmax even at a very low concentration of substrate
|
|
|
what three things could happen to the ES
|
back to substrates (E+S) or forward to products (E+P)
|
|
|
what is [Etotal]
|
[ES] +[Efree] which simply considers all forms of enzyme going on in a reaction
|
|
|
state the MM equation
|
v = vmax[S]/(Km + [S])
|
|
|
state the Lineweaver Burk equation
|
1/v = km/vmax x 1/[s] + 1/vmax
|
|
|
what is the slope of the lineweaver burk? The y intercept? the x and y axes?
|
slope = km/vmax
y int = 1/vmax y axis = 1/v x axis = 1/[s] |
|
|
what are the units of Km?
|
concentration... same units as [S]
|
|
|
what if Km = S
|
Then v is half velocity of vmax
|
|
|
what are three kinds of inhibitors?
|
competitive
non competitive uncompetitive |
|
|
where do competitive inhibitors bind? non competitive? uncompetitive?
|
same site
can bind to different site on enzyme OR ES complex binds exclusively to ES |
|
|
what changes in the MM or LB with a competitive inhibitor and why?
|
vmax is the same
km apparent increases because it takes reaction longer to get there |
|
|
what changes in the MM or LB with non competitive inhibitors?
|
vmax decreases
Km stays the same |
|
|
what changes in uncompetitive inhibition?
|
kM and vmax
BUT slope stays the same! |
|
|
what reactive groups are typically found in irreversible inhibitors?
|
ser
cys |
|
|
Finish the lecture
|
Finish the lecture!
|
|
|
which inhibitors usually form a covalent bond with some reactive gorup in the enzyme frequently a serine or cys at the active center
|
irreversible
|
|
|
what was our class example of a irreversible inhibitor
|
nerve gas DFP
|
|
|
what other type of inhibition do irreversible inhibitors look like
|
noncompetitive
|
|
|
what happens if you have an irreversilbe inhibitor that gave 50% inhibition and double it again?
|
100% inactivation
|
|
|
how can you overcome an irreversible inhibitor
|
high substrate conc
|
|
|
what is a suicide substrate
|
looks like the normal substrate but when it binds it forms a covalent bond
|
|
|
what three things does the reaction velocity depend on?
|
catalyc speed
availability of substrate concentration of product (rate of reverse reaction) |
|
|
define Km
|
the [S] at which reaction proceeds at 1/2vmax
|
|
|
what are the units of Km
|
sames as the substrate concentration units
|
|
|
T or F. Km depends on [enzyme] concentration
|
FALSE. Km is an intensive constant
|
|
|
Km is inversely relate to the affinity of an enzyme for its substrate, the higher the affinity the _____ the Km
|
lower
|
|
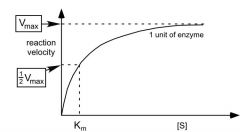
what would .5 units of the enzyme look like?
|
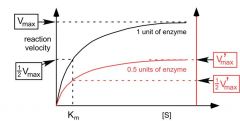
|
|
|
define Km in terms of k1 k2 k3
|
Km = (k2+k3)/k1
|
|
|
state the MM equation
|
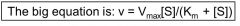
|
|
|
State the LB equation
|

|
|
|
Name two major classes of inhibitors
|
reversible
irreversible |
|
|
Name the three subclasses of reversible inhibitors
|
competitive
non competitive un competitive |
|
|
______ inhibitors react only with the free enzyme binding to the active site
|
competitive
|
|
|
what do the plots for competitive inhibitors look like?
|
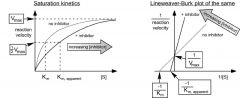
|
|
|
________ inhibitors react with the free enzyme and the enzyme substrate complex. They usually bind to a site on the enzyme surface away from the active site
|
non competitive
|
|
|
what do plots for noncompetitive inhibitors look like?
|
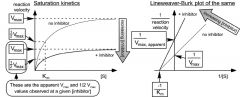
|
|
|
_______ inhibitors only react with the substrate bound form of the enzyme
|
uncompetitive
|
|
|
what do uncompetitive inhibitor plots look like?
|
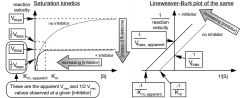
|
|

fill in the chart
|
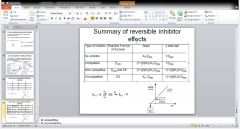
|
|
|
Addition of an irreversible inhibitor will continually lower _____
|
vmax
|

